Share This Page
Drug Price Trends for COUGH DM
✉ Email this page to a colleague
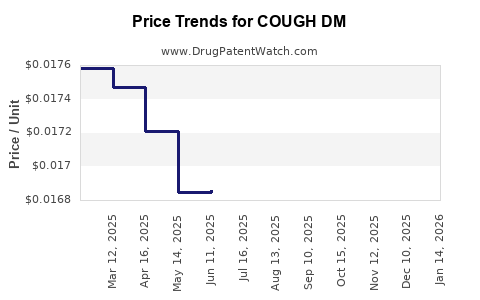
Average Pharmacy Cost for COUGH DM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| COUGH DM ER 30 MG/5 ML SUSP | 00904-6312-56 | 0.06775 | ML | 2026-03-18 |
| COUGH DM ER 30 MG/5 ML SUSP | 46122-0141-25 | 0.05203 | ML | 2026-03-18 |
| COUGH DM ER 30 MG/5 ML SUSP | 70000-0187-01 | 0.06775 | ML | 2026-03-18 |
| COUGH DM ER 30 MG/5 ML SUSP | 46122-0141-21 | 0.06775 | ML | 2026-03-18 |
| COUGH DM ER 30 MG/5 ML SUSP | 70000-0302-01 | 0.06775 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
COUGH DM Market Analysis and Financial Projection
What is COUGH DM?
COUGH DM is an over-the-counter (OTC) combination cough remedy typically containing dextromethorphan (DXM) as a cough suppressant and guaifenesin as an expectorant. Its primary indication targets temporary relief from coughs associated with colds and respiratory infections.
What is the current market landscape for cough remedies?
The global cough and cold remedy market was valued at approximately USD 11.4 billion in 2022. It projects a compound annual growth rate (CAGR) of roughly 3.2% from 2023 to 2030, driven by increased awareness of respiratory illnesses, aging populations, and consumer preference for OTC products.
Major players include Johnson & Johnson, Pfizer, Reckitt Benckiser, and GlaxoSmithKline.
Market segmentation
| Segment | Market Share (2022) | Key Products |
|---|---|---|
| Dextromethorphan-based | 45% | Robitussin DM, Delsym |
| Guaifenesin-based | 30% | Mucinex, Robitussin Chest |
| Other combinations | 25% | Product lines with additional ingredients |
How has the demand for COUGH DM evolved?
Demand remains stable, driven by OTC regulations favoring self-medication and a preference for non-prescription options. The COVID-19 pandemic amplified sales due to heightened respiratory illness awareness, although recent trends see stabilization.
What are the key factors influencing COUGH DM's pricing?
- Intellectual property rights: Since COUGH DM is marketed as a generic or OTC drug without extensive patent protection, price competition is significant.
- Raw material costs: Variations in dextromethorphan and guaifenesin costs, affected by global supply chains, impact pricing.
- Regulatory frameworks: OTC drug regulations influence allowable markups and pricing strategies.
- Market competition: Presence of numerous similar products limits premium pricing potential.
What are the current pricing benchmarks?
The average retail price for a 4 oz (120 mL) bottle of COUGH DM ranges between USD 4 and USD 8, depending on brand, packaging, and regional factors. Generic brands tend toward the lower end; branded products command higher prices.
Wholesale costs typically range from USD 2 to USD 5 per unit. Pricing strategies vary by company, with private labels undercutting national brands.
What are projected pricing trends for COUGH DM?
| Year | Estimated Retail Price Range (USD) | Key Influences |
|---|---|---|
| 2023 | USD 4 - USD 8 | Stable demand, high competition, raw material costs fluctuation |
| 2024 | USD 4 - USD 8 | Competitive pressure persists |
| 2025 | USD 4 - USD 9 | Slight price increase possible due to inflation and logistics costs |
| 2030 | USD 4 - USD 10 | Price elasticity limits significant increases |
Pricing will likely remain within current ranges, with minor upward adjustments aligned with inflation and benign raw material cost increases.
What are potential market risks?
- Regulatory changes could impose stricter OTC classifications, raising distribution costs.
- Increased regulation or control over dextromethorphan due to misuse concerns may restrict sales.
- Competition from natural or alternative remedies could pressure margins.
- Supply chain disruptions could inflate raw material costs, constraining pricing flexibility.
Key Takeaways
- The COUGH DM market is stable, with a valuation of USD 11.4 billion globally and a CAGR of 3.2%.
- Pricing is primarily influenced by competition, raw material costs, and regulatory environment.
- Current retail prices hover between USD 4 and USD 8 per bottle, with slight increases projected through 2030.
- Market risks include regulatory shifts and supply chain constraints limiting pricing power.
FAQs
1. How does regulatory status affect COUGH DM pricing?
Changes in OTC classification can increase distribution costs and limit sales, pressuring prices downward or making products less available. Stricter regulations may also require reformulation, impacting manufacturing costs.
2. Are generic COUGH DM products priced differently from branded ones?
Yes. Generics generally sell at lower prices (USD 2-USD 4 wholesale, USD 4-USD 6 retail), while branded products may command premiums up to USD 8 and above.
3. How do raw material fluctuations impact the market?
Dextromethorphan prices are sensitive to supply chain disruptions, often caused by geopolitical issues or manufacturing constraints, leading to cost increases for manufacturers and potential retail price hikes.
4. What role does OTC regulation play in market expansion?
Relaxing OTC regulations can expand access, boost sales volumes, and stabilize prices. Conversely, tighter restrictions can limit market size and influence pricing strategies.
5. Will emerging natural remedies threaten COUGH DM's market share?
Potentially. Consumers seeking natural or herbal alternatives may shift away from traditional OTC remedies, exerting downward pressure on pricing and market share.
Citations
[1] Grand View Research. "Cough & Cold Remedy Market Size, Share & Trends Analysis Report," 2022.
[2] Statista. "OTC Cough and Cold Medicine Market," 2023.
[3] U.S. Food and Drug Administration. OTC Drug Review Updates, 2022.
More… ↓
