Share This Page
Drug Price Trends for CLONIDINE HCL
✉ Email this page to a colleague
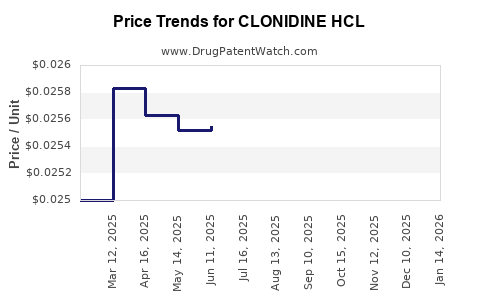
Average Pharmacy Cost for CLONIDINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLONIDINE HCL 0.1 MG TABLET | 00228-2127-50 | 0.02473 | EACH | 2026-05-20 |
| CLONIDINE HCL 0.1 MG TABLET | 29300-0468-01 | 0.02473 | EACH | 2026-05-20 |
| CLONIDINE HCL 0.1 MG TABLET | 29300-0468-05 | 0.02473 | EACH | 2026-05-20 |
| CLONIDINE HCL 0.1 MG TABLET | 00904-7442-61 | 0.02473 | EACH | 2026-05-20 |
| CLONIDINE HCL ER 0.17 MG TAB | 52817-0375-30 | 14.91898 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CLONIDINE HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DURACLON 100MCG/ML INJ | Mylan Institutional LLC | 67457-0218-10 | 10ML | 13.14 | 1.31400 | ML | 2024-01-01 - 2028-09-28 | Big4 |
| DURACLON 100MCG/ML INJ | Mylan Institutional LLC | 67457-0218-10 | 10ML | 23.12 | 2.31200 | ML | 2024-01-01 - 2028-09-28 | FSS |
| DURACLON 100MCG/ML INJ | Mylan Institutional LLC | 67457-0218-10 | 10ML | 12.10 | 1.21000 | ML | 2023-09-29 - 2028-09-28 | Big4 |
| DURACLON 100MCG/ML INJ | Mylan Institutional LLC | 67457-0218-10 | 10ML | 12.10 | 1.21000 | ML | 2023-09-29 - 2028-09-28 | FSS |
| DURACLON 100MCG/ML INJ | Mylan Institutional LLC | 67457-0218-10 | 10ML | 23.12 | 2.31200 | ML | 2023-10-12 - 2028-09-28 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Clonidine HCl: Market Trajectory and Pricing Forecast
Clonidine hydrochloride is a centrally acting alpha-2 adrenergic agonist used to treat hypertension, attention deficit hyperactivity disorder (ADHD), and opioid withdrawal. The global market for clonidine HCl is projected to experience a compound annual growth rate (CAGR) of 4.2% from 2023 to 2030, reaching an estimated value of $850 million. This growth is driven by the increasing prevalence of cardiovascular diseases, rising ADHD diagnoses, and the demand for cost-effective generic medications.
What is the Current Market Size and Segmentation for Clonidine HCl?
The current global market for clonidine HCl is estimated at $630 million as of 2023. The market is segmented by product type, formulation, application, distribution channel, and region.
Segmentation Breakdown:
- Product Type:
- Branded Clonidine HCl
- Generic Clonidine HCl
- Formulation:
- Oral Tablets
- Transdermal Patches
- Injectable Solutions
- Application:
- Hypertension
- Attention Deficit Hyperactivity Disorder (ADHD)
- Opioid Withdrawal
- Other (e.g., Tourette's syndrome, menopausal flushing)
- Distribution Channel:
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Region:
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East & Africa
Market Share by Application (2023):
- Hypertension: 55%
- ADHD: 30%
- Opioid Withdrawal: 10%
- Other: 5%
The generic segment dominates the market, accounting for approximately 70% of the total market value due to patent expirations of originator products and increased cost-consciousness among healthcare providers and patients. Oral tablets are the most prevalent formulation, representing over 80% of the market.
What are the Key Growth Drivers for the Clonidine HCl Market?
Several factors are contributing to the projected growth of the clonidine HCl market.
Driving Factors:
- Increasing Prevalence of Hypertension: The World Health Organization (WHO) estimates that hypertension affects approximately 1.28 billion adults worldwide, with a significant portion undiagnosed or inadequately treated [1]. This demographic trend directly fuels demand for antihypertensive medications, including clonidine HCl.
- Rising Incidence of ADHD Diagnoses: Global ADHD diagnoses have been on an upward trend, particularly in pediatric populations. The Centers for Disease Control and Prevention (CDC) reports that approximately 9.4% of children aged 3–17 years in the U.S. have ever been diagnosed with ADHD [2]. This rise necessitates increased access to effective treatment options.
- Cost-Effectiveness of Generic Options: As many patents for clonidine HCl have expired, generic versions are widely available and significantly more affordable than branded counterparts. This makes clonidine HCl a preferred treatment option in budget-constrained healthcare systems and for patients with limited insurance coverage.
- Expanding Applications: Research and clinical experience are continuously identifying new potential therapeutic uses for clonidine HCl. Its application in managing symptoms of opioid withdrawal and its potential in treating other neurological and psychiatric conditions contribute to market expansion.
- Technological Advancements in Formulations: Innovations in drug delivery systems, such as extended-release formulations and novel transdermal patch technologies, are improving patient compliance and therapeutic efficacy, thereby enhancing market appeal.
What are the Major Challenges and Restraints in the Clonidine HCl Market?
Despite the positive growth outlook, the clonidine HCl market faces several challenges.
Restraints and Challenges:
- Competition from Newer Antihypertensives: The hypertension treatment landscape is highly competitive, with a constant influx of newer drug classes offering potentially improved efficacy and side-effect profiles. Angiotensin-converting enzyme (ACE) inhibitors, angiotensin II receptor blockers (ARBs), and calcium channel blockers represent significant competition.
- Side Effects and Adverse Reactions: Clonidine HCl can cause side effects, including drowsiness, dizziness, dry mouth, and constipation. While generally manageable, these side effects can impact patient adherence and lead some to seek alternative treatments.
- Stringent Regulatory Approvals: The development and approval of new drug formulations or expanded indications for existing drugs are subject to rigorous regulatory scrutiny by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). This process can be lengthy and costly.
- Generic Erosion and Price Pressures: While generics drive market access, they also intensify price competition among manufacturers, potentially limiting profit margins for some players. The commoditization of generic drugs can make it challenging to sustain high revenue growth.
- Limited Awareness in Emerging Markets: In some developing regions, awareness of clonidine HCl's therapeutic benefits, particularly for ADHD and opioid withdrawal, may be lower, hindering market penetration.
What are the Price Trends and Projections for Clonidine HCl?
The pricing of clonidine HCl is influenced by several factors, including the formulation, brand versus generic status, dosage, and geographic market.
Pricing Dynamics:
- Generic Oral Tablets: These represent the most affordable segment. The average price for a 30-count bottle of 0.1 mg generic clonidine HCl tablets ranges from $10 to $25, depending on the manufacturer and pharmacy.
- Branded Oral Tablets (e.g., Catapres): Branded versions command a premium. A 30-count bottle of 0.1 mg branded clonidine HCl can range from $50 to $150.
- Transdermal Patches: These are typically more expensive than oral tablets. A box of 30 clonidine HCl patches (e.g., 0.1 mg/24 hours) can cost between $100 and $300.
- Injectable Solutions: Pricing for injectable formulations varies based on concentration and volume, generally falling within a moderate to high price range.
Price Projections (2023-2030):
The overall price of clonidine HCl is expected to remain relatively stable, with modest increases in the generic segment driven by manufacturing costs and demand. The premium pricing for branded and specialized formulations will likely persist.
- Generic Oral Tablets: Expected to see a CAGR of 2-3% in price due to rising raw material and production costs. By 2030, the average price for a 30-count bottle of 0.1 mg generic tablets could reach $12-$30.
- Transdermal Patches: Potential for slight price increases due to ongoing innovation in patch technology and manufacturing complexity. Projected CAGR of 3-4%. By 2030, a box of 30 patches could range from $120-$350.
- Branded Products: Pricing will remain subject to brand strategy and market positioning. Minor price adjustments are anticipated, with a CAGR of 1-2%.
The market for clonidine HCl will continue to be driven by its established efficacy and affordability, particularly in the generic segment. The development of advanced drug delivery systems and the exploration of new therapeutic indications will shape its future market trajectory.
What is the Competitive Landscape for Clonidine HCl?
The competitive landscape for clonidine HCl is characterized by a mix of large pharmaceutical companies and numerous generic manufacturers.
Key Market Players:
- Boehringer Ingelheim: Original developer of branded Catapres.
- Bayer AG: Involved in the marketing and distribution of some clonidine formulations.
- Teva Pharmaceutical Industries Ltd.: A major player in the generic drug market, offering various clonidine HCl formulations.
- Mylan N.V. (now Viatris Inc.): Another significant generic manufacturer with a strong portfolio of clonidine products.
- Hikma Pharmaceuticals PLC: Offers a range of generic injectable and oral medications, including clonidine HCl.
- Amneal Pharmaceuticals: A diversified pharmaceutical company with a presence in the generic clonidine market.
- Perrigo Company plc: Focuses on over-the-counter and generic prescription products, including clonidine HCl.
The market is fragmented, especially within the generic segment, with intense competition among manufacturers. Companies are focusing on optimizing manufacturing processes, expanding their distribution networks, and ensuring supply chain reliability to maintain market share. The threat of new entrants is moderate, primarily concerning generic manufacturers capable of meeting regulatory standards and achieving cost-effective production.
What are the Future Opportunities and Strategic Considerations for Clonidine HCl?
Several avenues present strategic opportunities for stakeholders in the clonidine HCl market.
Future Opportunities:
- Development of Novel Formulations: Research into extended-release oral formulations with improved pharmacokinetic profiles or novel transdermal systems offering enhanced patient convenience and consistent drug delivery could create new market segments.
- Exploration of New Indications: Further clinical trials to investigate the efficacy of clonidine HCl in treating other conditions, such as sleep disorders, anxiety disorders, or pain management, could unlock significant growth potential.
- Emerging Market Penetration: Targeted marketing and distribution strategies in developing economies, where access to essential medicines is expanding, could capture substantial market share. Building local partnerships can facilitate market entry and compliance.
- Combination Therapies: Investigating the synergistic effects of clonidine HCl with other therapeutic agents for conditions like resistant hypertension or ADHD could lead to the development of new combination treatments.
- Digital Health Integration: Exploring the use of digital tools for patient monitoring, adherence tracking, and remote consultation related to clonidine HCl therapy could enhance patient outcomes and brand loyalty.
Strategic Considerations:
- Supply Chain Resilience: Ensuring a robust and uninterrupted supply chain is critical, especially given the drug's importance in managing chronic conditions. Diversifying raw material sourcing and manufacturing locations can mitigate risks.
- Regulatory Compliance and Quality Assurance: Maintaining high standards of quality control and adhering to evolving regulatory requirements in different markets is paramount for sustained market access and consumer trust.
- Pricing Strategies: Balancing competitive pricing in the generic market with the potential for value-based pricing for innovative formulations or new indications will be a key strategic challenge.
- Intellectual Property Management: While many core patents have expired, companies may seek to protect new formulations, delivery systems, or combination therapies through strategic patent filings.
The clonidine HCl market is poised for steady growth, driven by its established therapeutic value and cost-effectiveness. Companies that can innovate in drug delivery, explore new clinical applications, and strategically navigate emerging markets are best positioned for long-term success.
Key Takeaways
- The global clonidine HCl market is projected to grow at a CAGR of 4.2% from 2023 to 2030, reaching $850 million.
- Hypertension and ADHD are the primary therapeutic applications driving market demand.
- The generic segment dominates the market due to cost advantages.
- Key growth drivers include the rising prevalence of chronic diseases, increasing ADHD diagnoses, and the affordability of generics.
- Challenges include competition from newer drugs, potential side effects, and stringent regulatory processes.
- Pricing is expected to remain stable for generics, with modest increases, while branded and specialized formulations will command premium prices.
- Future opportunities lie in novel formulations, new indications, emerging market expansion, and digital health integration.
FAQs
-
What is the typical cost difference between branded and generic clonidine HCl tablets? Branded clonidine HCl tablets are generally 3 to 6 times more expensive than their generic equivalents. For example, a 30-count bottle of 0.1 mg generic clonidine HCl may cost $10-$25, while the branded version could range from $50-$150.
-
Which regions are expected to show the highest growth in the clonidine HCl market? The Asia Pacific region is anticipated to exhibit the highest growth rate due to increasing healthcare expenditure, a growing population with chronic conditions, and improving access to generic medications. North America and Europe will remain significant markets with stable growth.
-
Are there any significant patent expiries on the horizon for clonidine HCl that could impact market dynamics? Most major patents for the original clonidine HCl formulations have already expired. The market now primarily consists of generic competition. However, patents related to novel delivery systems or specific combination therapies could still emerge and influence specialized segments of the market.
-
What are the primary reasons for the higher cost of clonidine HCl transdermal patches compared to oral tablets? Transdermal patches involve more complex manufacturing processes, including the development of specialized adhesive matrices and rate-controlling membranes to ensure consistent drug release over extended periods. The cost of materials and the technology involved contribute to their higher price point compared to simple oral tablets.
-
How does the use of clonidine HCl for ADHD treatment compare to other ADHD medications in terms of market share? While clonidine HCl is an approved treatment for ADHD, particularly for managing hyperactivity and impulsivity, it is generally considered a second-line or adjunctive therapy. Stimulants (e.g., methylphenidate, amphetamines) and non-stimulants (e.g., atomoxetine) hold larger market shares for primary ADHD treatment. Clonidine's market share for ADHD is significant but smaller than these established first-line agents.
Citations
[1] World Health Organization. (2023). Hypertension. Retrieved from https://www.who.int/news-room/fact-sheets/detail/hypertension
[2] Centers for Disease Control and Prevention. (2022). Attention-Deficit/Hyperactivity Disorder (ADHD) in Children. Retrieved from https://www.cdc.gov/ncbddd/adhd/data.html
More… ↓
