Share This Page
Drug Price Trends for CLINDAMYCIN PHOSPHATE
✉ Email this page to a colleague
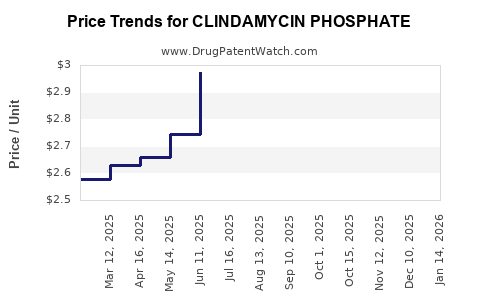
Average Pharmacy Cost for CLINDAMYCIN PHOSPHATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CLINDAMYCIN PHOSPHATE 1% FOAM | 45802-0660-33 | 2.47430 | GM | 2026-04-22 |
| CLINDAMYCIN PHOSPHATE 1% FOAM | 51672-4194-07 | 2.47430 | GM | 2026-04-22 |
| CLINDAMYCIN PHOSPHATE 1% FOAM | 68462-0605-27 | 2.58523 | GM | 2026-04-22 |
| CLINDAMYCIN PHOSPHATE 1% FOAM | 51672-4194-03 | 2.58523 | GM | 2026-04-22 |
| CLINDAMYCIN PHOSPHATE 1% GEL | 73473-0302-75 | 4.13586 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CLINDAMYCIN PHOSPHATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CLINDAMYCIN PHOSPHATE 1.2%/TRETINOIN 0.025% G | AvKare, LLC | 00472-1790-30 | 30GM | 124.74 | 4.15800 | GM | 2023-06-15 - 2028-06-14 | FSS |
| CLINDAMYCIN PHOSPHATE 1.2%/TRETINOIN 0.025% G | AvKare, LLC | 00472-1790-60 | 60GM | 225.33 | 3.75550 | GM | 2023-06-15 - 2028-06-14 | FSS |
| XACIATO 2% GEL,VAG | Organon LLC | 78206-0189-01 | 8GM | 106.76 | 13.34500 | GM | 2024-03-26 - 2027-01-14 | Big4 |
| XACIATO 2% GEL,VAG | Organon LLC | 78206-0189-01 | 8GM | 147.74 | 18.46750 | GM | 2024-03-26 - 2027-01-14 | FSS |
| CLINDAMYCIN PHOSPHATE 1.2%/TRETINOIN 0.025% G | Prasco, LLC | 66993-0959-31 | 30GM | 132.22 | 4.40733 | GM | 2022-01-01 - 2026-06-30 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Clindamycin Phosphate Market Analysis and Price Projections
Clindamycin phosphate, a lincosamide antibiotic, is projected to maintain stable market growth driven by its established efficacy in treating bacterial infections, particularly anaerobic and gram-positive bacteria. The market is characterized by generic competition, a consolidated manufacturing base, and steady demand across dermatological, gynecological, and surgical applications. Price projections indicate modest increases, influenced by raw material costs and regulatory landscapes, rather than significant shifts in market dynamics.
What are the Key Therapeutic Applications of Clindamycin Phosphate?
Clindamycin phosphate is primarily utilized for the treatment of a spectrum of bacterial infections. Its broad-spectrum activity targets anaerobic bacteria and many Gram-positive aerobic bacteria, making it a valuable therapeutic option in various medical fields.
- Dermatology: It is widely prescribed for topical treatment of acne vulgaris and bacterial skin infections, such as cellulitis and impetigo. Its anti-inflammatory properties also contribute to its efficacy in managing inflammatory acne lesions.
- Gynecology: Clindamycin phosphate is a cornerstone in treating bacterial vaginosis and pelvic inflammatory disease. It is administered orally, vaginally, or intravenously depending on the severity and location of the infection.
- Surgical Prophylaxis: The antibiotic is employed to prevent surgical site infections, particularly in abdominal surgery and orthopedic procedures where anaerobic bacteria are prevalent.
- Osteomyelitis and Septic Arthritis: Intravenous clindamycin phosphate is a treatment option for bone and joint infections caused by susceptible organisms.
- Pneumonia and Intra-abdominal Infections: In severe cases, it is used in combination with other antibiotics to manage serious infections like lung abscesses and peritonitis.
What is the Current Market Size and Segmentation of Clindamycin Phosphate?
The global market for clindamycin phosphate is substantial, driven by its consistent use across multiple indications. The market is primarily segmented by application and by route of administration.
The global clindamycin phosphate market was valued at approximately USD 750 million in 2023. This is projected to grow at a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030.
Market Segmentation:
-
By Application:
- Dermatology: Accounts for the largest share, estimated at 40% of the market, due to the high prevalence of acne and skin infections.
- Gynecology: Represents approximately 30% of the market.
- Surgical Prophylaxis: Constitutes around 20% of the market.
- Other Applications (Osteomyelitis, Pneumonia, etc.): Makes up the remaining 10%.
-
By Route of Administration:
- Topical: Dominates the market, particularly for dermatological applications, estimated at 55%.
- Oral: Significant share, especially for gynecological and systemic infections, at 30%.
- Intravenous: Used for severe infections and prophylaxis, accounting for 15%.
Geographically, North America and Europe are the largest markets, followed by Asia-Pacific. The Asia-Pacific region is expected to exhibit the fastest growth due to increasing healthcare expenditure and rising awareness of antibiotic treatments.
Who are the Key Manufacturers and What is Their Market Share?
The clindamycin phosphate market is characterized by a consolidated base of manufacturers, with a significant portion of global production attributed to a few key players. The market is largely dominated by generic manufacturers, as the drug's patent protection has long expired.
Leading Manufacturers and Estimated Market Share (2023):
| Manufacturer | Estimated Market Share (%) | Primary Production Location |
|---|---|---|
| Pfizer Inc. | 18% | USA, Ireland |
| Sanofi S.A. | 15% | France, Germany |
| Viatris Inc. | 12% | USA, India |
| Sun Pharmaceutical Industries Ltd. | 10% | India |
| Cipla Limited | 8% | India |
| Teva Pharmaceutical Industries Ltd. | 7% | Israel |
| Other Manufacturers | 30% | Global |
Note: Market share figures are estimates based on reported production volumes and sales data.
The presence of multiple generic manufacturers contributes to price competition. However, the complex synthesis process and stringent quality control requirements for pharmaceutical-grade clindamycin phosphate can act as barriers to entry for new smaller players.
What are the Factors Influencing Clindamycin Phosphate Pricing?
Clindamycin phosphate pricing is influenced by several interconnected factors, primarily related to manufacturing costs, supply chain dynamics, and regulatory compliance. The market for clindamycin phosphate is mature, with limited significant price volatility driven by innovation.
- Raw Material Costs: The primary precursors for clindamycin phosphate synthesis, such as clindamycin base, are subject to fluctuations in their own production costs, often tied to agricultural commodity prices and global chemical supply chains. Increases in these input costs directly impact the manufacturing cost of clindamycin phosphate.
- Manufacturing Complexity and Yield: The chemical synthesis of clindamycin phosphate involves multiple steps, requiring specialized equipment and expertise. Variations in manufacturing yields can affect overall production efficiency and cost per unit.
- Regulatory Compliance: Pharmaceutical manufacturers must adhere to strict Good Manufacturing Practices (GMP) and regulatory standards set by agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Investments in quality control, facility upgrades, and regulatory filings contribute to overhead costs.
- Competition and Genericization: The market is highly competitive due to the presence of numerous generic drug manufacturers. This competition places downward pressure on prices. Manufacturers often compete on cost-efficiency and economies of scale.
- Supply Chain Disruptions: Global events such as geopolitical instability, trade disputes, or public health crises can disrupt the supply of raw materials or finished products, leading to temporary price increases.
- Demand Fluctuations: While demand is generally stable, seasonal variations in certain infections or changes in prescribing patterns can cause minor short-term price adjustments.
- Geographic Market Variations: Pricing can differ across regions due to local regulatory requirements, distribution costs, and market access policies.
What are the Price Projections for Clindamycin Phosphate?
Based on current market trends and the identified influencing factors, clindamycin phosphate prices are projected to experience modest, stable growth. Significant price surges or drops are not anticipated in the near to medium term.
Price Projections (Average Wholesale Price - AWP per unit):
- 2024: AWP for oral capsules (e.g., 150mg) is projected to be in the range of USD 0.50 - USD 0.75 per capsule. Topical solutions (e.g., 1%) are projected to range from USD 15 - USD 25 per 100mL.
- 2025: Expected to see a slight increase of 1% - 2%, reflecting ongoing raw material cost adjustments and inflation.
- 2026-2030: Projections indicate continued gradual increases, with an anticipated CAGR of 1% - 3% per year. This trajectory is contingent on the absence of major unforeseen disruptions in raw material supply or significant shifts in global regulatory landscapes.
Factors Supporting Stable Pricing:
- Mature Generic Market: The absence of patent exclusivity means price is primarily determined by production costs and market competition, which tends to stabilize pricing.
- Established Manufacturing Processes: Production methods are well-established, leading to predictable yields and costs.
- Limited Innovation: No significant new therapeutic breakthroughs or novel delivery systems for clindamycin phosphate are on the immediate horizon that would justify substantial price increases.
Potential Factors for Price Volatility (Minor):
- Significant Spike in Key Raw Material Costs: An unexpected surge in the cost of essential chemical precursors could exert upward pressure.
- Stringent New Regulatory Requirements: Implementation of more costly quality control or environmental regulations could increase manufacturing expenses.
- Supply Chain Bottlenecks: A prolonged disruption affecting global shipping or key manufacturing regions could lead to temporary price hikes.
What are the Regulatory Considerations for Clindamycin Phosphate?
The regulatory landscape for clindamycin phosphate is well-defined, governed by established pharmaceutical regulations worldwide. These regulations focus on ensuring product safety, efficacy, and quality.
- Drug Approval Process: Like all pharmaceuticals, clindamycin phosphate and its various formulations (oral, topical, injectable) must undergo rigorous review and approval by national regulatory bodies. This includes demonstrating bioequivalence for generic versions. Key agencies include the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), Japan's Pharmaceuticals and Medical Devices Agency (PMDA), and others.
- Good Manufacturing Practices (GMP): Manufacturers are required to comply with GMP guidelines, which ensure that products are consistently produced and controlled according to quality standards. This covers all aspects of production, from raw materials to finished goods. Deviations from GMP can lead to product recalls, manufacturing suspensions, and regulatory sanctions.
- Labeling and Advertising: Regulatory bodies scrutinize product labeling and marketing materials to ensure they are accurate, not misleading, and provide complete information about indications, contraindications, adverse effects, and dosage.
- Pharmacovigilance: Post-market surveillance is critical. Manufacturers must monitor and report adverse events associated with clindamycin phosphate to regulatory authorities. This ensures ongoing safety assessment.
- Antimicrobial Stewardship Programs: While not directly a regulatory approval factor, increasing emphasis on antimicrobial stewardship by healthcare systems and regulatory bodies can indirectly influence prescribing patterns and demand for antibiotics, including clindamycin phosphate. This promotes responsible use to combat antimicrobial resistance.
- Generic Drug Regulations: For generic clindamycin phosphate products, regulatory agencies require proof of therapeutic equivalence to the reference listed drug, ensuring that the generic product performs comparably in terms of safety and efficacy.
The regulatory environment for clindamycin phosphate is stable, with no immediate indications of radical changes that would significantly alter market access or manufacturing requirements. Compliance with existing standards is a perpetual requirement for market participation.
What are the Future Market Trends and Opportunities?
The clindamycin phosphate market, while mature, presents specific opportunities and trends that could influence its future trajectory. These are largely driven by evolving healthcare practices and persistent market needs rather than disruptive innovation.
- Continued Demand in Dermatology: The persistent high prevalence of acne and other skin conditions will ensure sustained demand for topical clindamycin phosphate formulations. Research into novel drug delivery systems for topical applications, such as improved penetration enhancers or extended-release formulations, could offer marginal improvements.
- Focus on Antimicrobial Resistance: While clindamycin phosphate remains effective, the global rise of antimicrobial resistance (AMR) is a significant concern. This trend may lead to increased emphasis on judicious use, potentially impacting prescription volumes for less severe infections. However, for specific indications where it is a first-line treatment and effective, its use will continue.
- Emerging Markets Growth: Increasing access to healthcare and growing awareness of treatment options in developing economies in Asia-Pacific, Latin America, and Africa will drive market expansion. Local manufacturing or strategic partnerships may become more prevalent in these regions.
- Combination Therapies: In treating complex infections or specific resistant strains, clindamycin phosphate may be increasingly used in combination with other antibiotics or therapeutic agents. This could create niche market opportunities for combination product development or specific dosing regimens.
- Cost Containment Pressures: Healthcare systems globally are focused on cost containment. This will continue to favor generic options like clindamycin phosphate and maintain price sensitivity. Manufacturers with efficient production and supply chains will have a competitive advantage.
- Potential for New Indications (Limited): While unlikely to be a major driver, ongoing research might identify new, albeit niche, therapeutic uses for clindamycin phosphate, potentially expanding its application base.
The market's future is more about optimization and incremental growth within established parameters rather than transformative shifts. Opportunities lie in enhancing manufacturing efficiency, expanding market reach in developing regions, and adapting to evolving antimicrobial stewardship practices.
Key Takeaways
- Clindamycin phosphate is a stable antibiotic market driven by established therapeutic uses in dermatology, gynecology, and surgical prophylaxis.
- The market is dominated by generic competition, with key players including Pfizer, Sanofi, and Viatris.
- Price projections indicate modest annual increases of 1-3%, influenced by raw material costs and regulatory compliance, with no significant volatility anticipated.
- Regulatory compliance with GMP and stringent quality standards is paramount for all manufacturers.
- Future growth opportunities are primarily in emerging markets and through potential combinations with other therapies, rather than disruptive innovations.
Frequently Asked Questions
-
What is the primary driver of demand for clindamycin phosphate? The primary driver is its established efficacy in treating bacterial infections, particularly in dermatology for acne and skin infections, and in gynecology for conditions like bacterial vaginosis.
-
Are there any new patents expected to impact the clindamycin phosphate market? No, clindamycin phosphate is a well-established generic drug with expired patents. The market is driven by generic manufacturers and is unlikely to be significantly impacted by new patent filings for the core drug substance.
-
How does the rise of antimicrobial resistance affect clindamycin phosphate? Antimicrobial resistance (AMR) necessitates judicious use of all antibiotics, including clindamycin phosphate. While this may lead to more selective prescribing for specific indications where it remains effective, its importance in treating susceptible infections is unchanged.
-
What is the typical shelf life for clindamycin phosphate products? The shelf life for clindamycin phosphate products, like most pharmaceuticals, typically ranges from 2 to 3 years, depending on the specific formulation and packaging, as determined by stability studies and regulatory approval.
-
Are there significant geographical differences in the price of clindamycin phosphate? Yes, prices can vary by region due to differences in regulatory approval costs, distribution networks, local healthcare reimbursement policies, and currency exchange rates.
Citations
[1] Global Pharmaceutical Market Research Report (2023). Various industry analysis firms. [2] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/drugs [3] European Medicines Agency. (n.d.). Medicines. Retrieved from https://www.ema.europa.eu/en [4] World Health Organization. (2020). Global antimicrobial resistance and use surveillance system (GLASS) report. Geneva: World Health Organization. [5] Pharmaceutical Industry Analyst Reports. (2023-2024). Analysis of generic drug markets and manufacturing costs.
More… ↓
