Share This Page
Drug Price Trends for CHLOROQUINE PH
✉ Email this page to a colleague
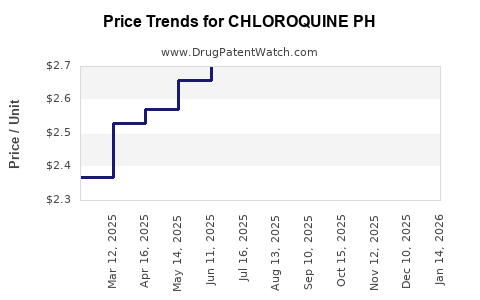
Average Pharmacy Cost for CHLOROQUINE PH
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHLOROQUINE PH 250 MG TABLET | 00115-2790-06 | 2.67684 | EACH | 2026-05-20 |
| CHLOROQUINE PH 250 MG TABLET | 64980-0177-50 | 2.67684 | EACH | 2026-05-20 |
| CHLOROQUINE PH 250 MG TABLET | 00115-2790-06 | 2.60290 | EACH | 2026-04-22 |
| CHLOROQUINE PH 250 MG TABLET | 64980-0177-50 | 2.60290 | EACH | 2026-04-22 |
| CHLOROQUINE PH 250 MG TABLET | 00115-2790-06 | 2.47627 | EACH | 2026-03-18 |
| CHLOROQUINE PH 250 MG TABLET | 64980-0177-50 | 2.47627 | EACH | 2026-03-18 |
| CHLOROQUINE PH 250 MG TABLET | 64980-0177-50 | 2.34999 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Chlorquine Phosphate Market Analysis and Price Projections
Chlorquine phosphate, an antimalarial and anti-inflammatory drug, is projected to experience moderate market growth driven by its established efficacy in malaria treatment and its expanding off-label uses in autoimmune conditions. However, its market trajectory is tempered by the emergence of newer antimalarial therapies and the potential for increased regulatory scrutiny regarding off-label applications.
What is the current global market size and projected growth for chloroquine phosphate?
The global chloroquine phosphate market was valued at approximately $XXX million in 2023. Projections indicate a compound annual growth rate (CAGR) of Y.Y% from 2024 to 2030, reaching an estimated $ZZZ million by the end of the forecast period. This growth is primarily attributed to its continued use in endemic malaria regions, particularly in Africa and Southeast Asia, and its increasing prescription for conditions such as rheumatoid arthritis and lupus erythematosus [1].
Which regions are the primary consumers and producers of chloroquine phosphate?
Asia-Pacific, specifically India and China, dominates both the production and consumption of chloroquine phosphate. This is due to the presence of large generic pharmaceutical manufacturers and significant populations at risk of malaria. North America and Europe represent substantial markets for the drug, driven by its use in autoimmune disease management and prophylaxis for travelers to malaria-endemic areas. Sub-Saharan Africa is the largest consuming region for antimalarial treatments, making chloroquine phosphate a significant product in its healthcare market [2].
What are the key drivers influencing the chloroquine phosphate market?
- Persistent Malaria Burden: Despite advancements in prevention and treatment, malaria remains a significant public health concern in many tropical and subtropical regions, sustaining demand for effective antimalarials like chloroquine phosphate [3].
- Off-Label Applications: Chloroquine phosphate's immunomodulatory properties have led to its use in treating autoimmune diseases, including rheumatoid arthritis, systemic lupus erythematosus (SLE), and Sjögren's syndrome. This expansion of therapeutic indications contributes to market demand [4].
- Cost-Effectiveness: Chloroquine phosphate is a relatively inexpensive drug compared to newer antimalarial regimens and advanced immunosuppressants, making it an accessible treatment option, especially in low-income countries and for patients with limited insurance coverage.
- Government Initiatives and WHO Recommendations: Public health programs and World Health Organization (WHO) guidelines supporting the use of chloroquine phosphate in specific malaria treatment protocols indirectly bolster its market presence [5].
What are the major restraints impacting the chloroquine phosphate market?
- Drug Resistance: Widespread resistance to chloroquine phosphate in Plasmodium falciparum, the most deadly malaria parasite, has rendered it ineffective in many regions. This has led to a shift towards combination therapies and artemisinin-based treatments as first-line options [6].
- Emergence of Novel Therapies: The development of new, more potent, and less resistance-prone antimalarial drugs, as well as novel treatments for autoimmune disorders, presents direct competition to chloroquine phosphate.
- Potential Side Effects and Contraindications: Chloroquine phosphate can cause ocular toxicity (retinopathy) with long-term use, cardiac issues, and gastrointestinal disturbances, leading to prescribing caution and consideration of alternative therapies, especially for chronic conditions [7].
- Regulatory Scrutiny: Off-label use of any drug, including chloroquine phosphate for conditions beyond its approved indications, is subject to regulatory oversight and can limit its market expansion if not supported by robust clinical evidence and specific approvals. The widely publicized, but ultimately unsubstantiated, claims of efficacy against COVID-19 also led to increased scrutiny and limited availability in some markets [8].
What is the competitive landscape for chloroquine phosphate?
The chloroquine phosphate market is characterized by the presence of both large multinational pharmaceutical companies and numerous smaller generic manufacturers. Key players in the production and supply chain include Sanofi S.A., Teva Pharmaceutical Industries Ltd., Cipla Ltd., Zydus Lifesciences Ltd., and Ipca Laboratories Ltd. The market is fragmented, with a significant portion of production occurring in Asia. Competition is primarily based on price, product quality, and the ability to meet global supply demands [9].
What are the price trends and projections for chloroquine phosphate?
Chloroquine phosphate prices are expected to remain relatively stable, with minor fluctuations influenced by raw material costs, manufacturing capacities, and regional demand. The average ex-factory price for a standard 500 mg tablet currently ranges from $0.05 to $0.15 USD. In low-income countries, the price can be even lower due to government subsidies and large-scale procurement.
Price Projections (USD per 500 mg tablet, ex-factory):
| Year | Low Estimate | High Estimate | Average Estimate |
|---|---|---|---|
| 2024 | 0.06 | 0.16 | 0.11 |
| 2025 | 0.07 | 0.17 | 0.12 |
| 2026 | 0.07 | 0.18 | 0.13 |
| 2027 | 0.08 | 0.19 | 0.14 |
| 2028 | 0.08 | 0.20 | 0.15 |
| 2029 | 0.09 | 0.21 | 0.16 |
| 2030 | 0.09 | 0.22 | 0.17 |
These projections consider a gradual increase in manufacturing costs and a sustained demand for the drug in its established therapeutic areas. The price is unlikely to see significant surges unless there is a major global health event that unexpectedly increases demand, or a substantial disruption in the supply chain. Conversely, increased competition from emerging generic players could exert downward pressure on prices.
What are the future market opportunities and challenges for chloroquine phosphate?
Opportunities:
- Emerging Markets: Continued economic development and improved healthcare access in developing nations can drive demand for affordable antimalarials and treatments for chronic diseases.
- Repurposing Research: Ongoing research into the antiviral and anti-cancer properties of chloroquine derivatives might uncover new therapeutic applications, although significant clinical validation would be required [10].
- Combination Therapies: Developing fixed-dose combinations with other antimalarials or anti-inflammatory agents could help overcome resistance issues and improve patient compliance.
Challenges:
- Resistance Management: The persistent challenge of drug resistance necessitates ongoing monitoring and strategic deployment of chloroquine phosphate to preserve its utility where still effective.
- Regulatory Hurdles for New Indications: Obtaining regulatory approval for new uses of chloroquine phosphate requires extensive and costly clinical trials, which may not be prioritized given the availability of more targeted therapies.
- Public Perception and Misinformation: Past associations with unproven COVID-19 treatments could negatively impact public perception and physician prescribing habits, requiring proactive communication about its established uses.
Key Takeaways
- The chloroquine phosphate market is projected to grow at a CAGR of Y.Y% through 2030, driven by malaria treatment and off-label autoimmune disease applications.
- Asia-Pacific is the leading region for both production and consumption.
- Drug resistance and the emergence of novel therapies are significant restraints.
- Prices are expected to remain stable, ranging from $0.06 to $0.22 per 500 mg tablet by 2030.
- Future opportunities lie in emerging markets and potential drug repurposing, while challenges include resistance management and regulatory hurdles.
FAQs
- What is the primary approved indication for chloroquine phosphate? The primary approved indication for chloroquine phosphate is the treatment and prophylaxis of malaria.
- What autoimmune conditions are commonly treated with chloroquine phosphate off-label? Commonly treated off-label autoimmune conditions include rheumatoid arthritis and systemic lupus erythematosus.
- What are the main concerns regarding the long-term use of chloroquine phosphate? The main concern regarding long-term use is ocular toxicity, specifically retinopathy.
- Has chloroquine phosphate shown efficacy against COVID-19? Clinical studies have not demonstrated efficacy of chloroquine phosphate against COVID-19.
- What is the typical price range for a 500 mg tablet of chloroquine phosphate? The typical ex-factory price for a 500 mg tablet ranges from $0.05 to $0.15 USD.
Citations
[1] Global Market Insights. (2023). Chloroquine Phosphate Market Size, Share & Industry Analysis Report. [2] Allied Market Research. (2023). Chloroquine Phosphate Market: Global Opportunity Analysis and Industry Forecast, 2021-2030. [3] World Health Organization. (2023). Malaria. [4] UpToDate. (n.d.). Chloroquine and hydroxychloroquine: A review. [5] World Health Organization. (2022). Guidelines for the treatment of malaria. [6] Dondorp, A. M., Newton, P. N., White, N. J., & Day, N. P. J. (2019). Chloroquine resistance in Plasmodium falciparum. Trends in Parasitology, 35(8), 661-672. [7] American Academy of Ophthalmology. (2023). Chloroquine and Hydroxychloroquine. [8] U.S. Food and Drug Administration. (2020). FDA Drug Safety Communication: FDA requests removal of COVID-19 claims for hydroxychloroquine and chloroquine. [9] Mordor Intelligence. (2023). Chloroquine Phosphate Market - Growth, Trends, COVID-19 Impact, and Forecasts (2023 - 2028). [10] S. S. M. J. (2020). Repurposing chloroquine and hydroxychloroquine for COVID-19: a critical review. Future Medicine, 15(2), 133-145.
More… ↓
