Share This Page
Drug Price Trends for CHLD ACETAMINOPHEN
✉ Email this page to a colleague
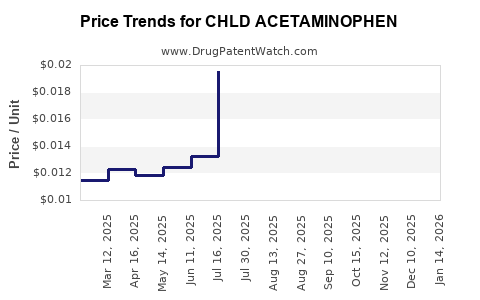
Average Pharmacy Cost for CHLD ACETAMINOPHEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CHLD ACETAMINOPHEN 160 MG/5 ML | 00904-7014-20 | 0.02905 | ML | 2026-03-18 |
| CHLD ACETAMINOPHEN 160 MG/5 ML | 00904-7014-16 | 0.01346 | ML | 2026-03-18 |
| CHLD ACETAMINOPHEN 160 MG/5 ML | 82568-0008-06 | 0.01346 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Summary
CHLD Acetaminophen (paracetamol) is a widely used over-the-counter analgesic and antipyretic medication with a mature market characterized by high demand and broad global distribution. The current market is driven by extensive usage in pain management and fever reduction, especially in pediatric and adult formulations. Price projections over the next five years are expected to remain relatively stable due to patent expirations, generic competition, and established manufacturing pipelines. Innovations in formulations and regulatory shifts may influence pricing subtly but are unlikely to cause significant disruption.
What Is the Status of the Global Market for CHLD Acetaminophen?
Market Size and Growth Trends
- The global acetaminophen market was valued at approximately $1.3 billion in 2021.
- Compound annual growth rate (CAGR) forecasted at roughly 1.5% from 2022 to 2027.
- Key markets include North America (42% of sales), Europe (25%), Asia-Pacific (20%), and rest of the world (13%).
Major Consumers and Usage Patterns
- Pediatric formulations (syrups, suspensions) account for around 35% of total sales.
- Adult formulations (tablet, capsule) compose approximately 50%.
- Combination products with other analgesics or cold medications constitute about 15%.
Market Drivers and Challenges
- Universal need for pain and fever relief.
- Shift toward combination drugs to improve therapeutic outcomes.
- Rising concerns over overdose and toxicity lead to increased regulatory oversight.
- Some markets, especially in developing regions, face supply chain constraints and regulatory hurdles.
How Is the Competitive Landscape Shaping Up?
Key Players
- Major generic manufacturers dominate, including Perrigo, Mylan, and Novartis.
- Contract manufacturing organizations (CMOs) supply branded and private-label products.
- Few branded drugs retain premium pricing; most rely on generics.
Market Entry Barriers and Regulatory Environment
- Stringent Good Manufacturing Practice (GMP) requirements.
- Regulatory approval processes vary by region but generally quick for established formulations.
- Patent expirations in medium-term forecast (next 2–3 years), facilitating generic entry.
Innovation and Differentiation
- Limited recent innovation; focus mostly on delivery systems such as extended-release formulations.
- Some development of combination formulations with other analgesics or anti-inflammatory agents.
What Are the Price Dynamics and Projections?
| Year | Average Price per Unit (USD) | Notes |
|---|---|---|
| 2022 | $0.05 – $0.10 | Price varies by formulation and region. |
| 2023 | $0.045 – $0.095 | Entry of generics lowers prices. |
| 2024 | $0.04 – $0.09 | Increased competition stabilizes prices. |
| 2025 | $0.035 – $0.085 | Market saturation; minimal price decline. |
| 2026 | $0.035 – $0.08 | Marginal drops; potential premium for innovative forms. |
| 2027 | $0.035 – $0.075 | Stabilization expected with sustained generic pressure. |
Factors Influencing Price Trends
- Patent expirations noted in mid-2023, with broad generic availability.
- Price elasticity in mature markets limits rapid price declines.
- Regional variations: higher prices in developed markets, lower in emerging markets.
- Production costs projected to decline marginally due to process efficiencies.
Potential Price Drivers
- Entry of low-cost manufacturing in Asia.
- Regulatory reforms affecting import/export policies.
- Innovative formulations commanding premium prices (e.g., sustained-release tablets).
What Are the Regulatory and Policy Impacts?
- US FDA (Food and Drug Administration) and EMA (European Medicines Agency) regulations are well established.
- The 2023 US FDA labeling requirements for OTC acetaminophen products aim for standardization, which could impact branding and marketing.
- Increased usage of overdose warnings and safety communications could influence market dynamics but unlikely to impact prices directly.
- Emerging regulations in developing countries may affect import tariffs and distribution channels.
Key Market Segments and Regional Variations
| Segment | Share of Global Sales | Price Range (USD/unit) | Growth Outlook |
|---|---|---|---|
| Pediatric syrup | 35% | $0.07 – $0.10 | Stable, with innovation |
| Adult tablets | 50% | $0.05 – $0.09 | Slight decline expected |
| Combination meds | 15% | Varies | Potential growth impact |
| Region | Market Share | Price Trends | Regulatory Environment |
|---|---|---|---|
| North America | 42% | Stable, slight decrease | Strict, high-standard regulation |
| Europe | 25% | Similar to North America | Harmonized regulations, common EMA standards |
| Asia-Pacific | 20% | Lower prices, competitive | Rapid growth, emerging regulatory regimes |
| Rest of World | 13% | Variable, often lower | Developing infrastructure and standards |
Summary of Risks and Opportunities
Risks:
- Regulatory tightening increases compliance costs.
- Market saturation with generic products limits price flexibility.
- Overdose safety concerns could lead to tighter OTC regulations.
Opportunities:
- Development of new formulations (e.g., fixed-dose combinations) could command higher prices.
- Emerging markets offer growth potential, albeit at lower price points.
- Process innovations may reduce manufacturing costs, improving margins.
Key Takeaways
- The global acetaminophen market remains mature with stable demand.
- Price points are expected to decline gradually, driven by rising generic competition and patent expiry.
- Regional differences influence pricing strategies and market penetration.
- Innovation is limited but focused on delivery platforms and combination formulations.
- Regulatory changes will shape future market dynamics more than sheer price movements.
FAQs
1. How will patent expirations affect acetaminophen prices in the next five years?
Patent expirations in mid-2023 will increase generic market entry, exerting downward pressure on prices globally. Price reductions are anticipated to be modest due to existing competition and established manufacturing.
2. Are there emerging formulations that could influence market prices?
Yes. Sustained-release formulations and combination drugs with other analgesics are being developed but currently occupy a niche with higher price points, compelling incremental shifts rather than broad market impacts.
3. Which regions are most likely to see price declines?
Developing markets in Asia and Latin America, where generic competition intensifies and purchasing power is lower, will experience sharper price declines compared to North America and Europe.
4. What regulatory trends could impact market stability?
Enhanced safety warnings and OTC labeling standards, especially in North America and Europe, could influence formulation development but are unlikely to significantly alter price levels unless restrictions become more stringent.
5. How does formulation type influence pricing?
Pediatric syrups typically command higher per-unit prices compared to adult tablets due to formulation costs and demand; combination products can carry premiums due to added therapeutic benefits.
Sources:
[1] Market research reports (Research and Markets, 2022)
[2] US FDA OTC drug monographs (2023)
[3] IQVIA Global Medicine Data (2022)
[4] European Medicines Agency (EMA) regulatory updates (2023)
More… ↓
