Share This Page
Drug Price Trends for BRIMONIDINE-TIMOLOL
✉ Email this page to a colleague
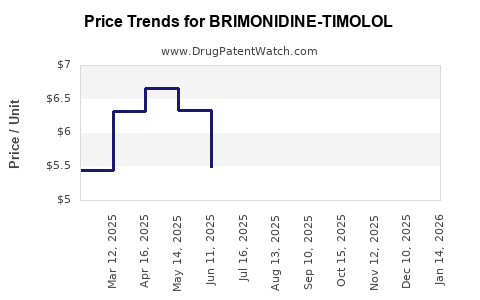
Average Pharmacy Cost for BRIMONIDINE-TIMOLOL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BRIMONIDINE-TIMOLOL 0.2%-0.5% | 82182-0455-15 | 2.38335 | ML | 2026-05-20 |
| BRIMONIDINE-TIMOLOL 0.2%-0.5% | 00781-7186-70 | 2.37038 | ML | 2026-05-20 |
| BRIMONIDINE-TIMOLOL 0.2%-0.5% | 00781-7186-75 | 3.12888 | ML | 2026-05-20 |
| BRIMONIDINE-TIMOLOL 0.2%-0.5% | 00832-1425-05 | 3.12888 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is Brimonidine-Timolol?
Brimonidine-timolol is a fixed-combination eye drop therapy used to lower intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. The drug combines brimonidine, an alpha-2 adrenergic agonist, with timolol, a beta-adrenergic blocker. Its mechanism targets different pathways to reduce aqueous humor production and increase outflow, effectively mitigating glaucoma progression.
What Is the Global Market Size for Brimonidine-Timolol?
The current market value estimates around $450 million in 2022. The compound annual growth rate (CAGR) is projected at approximately 5.2% from 2023 to 2030, driven by increasing prevalence of glaucoma, especially in aging populations.
Regional Market Breakdown (2022)
| Region | Market Size ($ millions) | CAGR (2023-2030) | Key Drivers |
|---|---|---|---|
| North America | 180 | 4.8% | High diagnosis rate, reimbursement policies |
| Europe | 90 | 5.4% | Aging demographic, healthcare infrastructure |
| Asia-Pacific | 120 | 6.1% | Growing healthcare access, rising glaucoma cases |
| Latin America | 30 | 4.2% | Increasing awareness, urbanization |
| Rest of World | 30 | 5.0% | Emerging markets, market expansion |
What Are Key Drivers and Barriers in the Market?
Drivers:
- Aging populations leading to higher glaucoma incidence.
- Increased awareness and screening programs.
- Advancements in formulation, delivering better patient compliance.
- Patent expirations of branded therapies, fostering generic entry.
Barriers:
- Competition from monotherapies and other fixed-combination drugs.
- Side effects associated with beta-blockers (e.g., systemic cardiovascular effects).
- Cost constraints, particularly in emerging markets.
- Patent protections still upheld for certain formulations, delaying generics.
How Is the Market Structured?
The market is primarily composed of three segments:
- Brand-Name Drugs: Companies like Alcon, Novartis, and Eyevance Pharmaceuticals dominate with proprietary formulations.
- Generic Versions: Entered after patent expires; price competition intensifies.
- Private Label Brands: Available mainly in emerging markets, often at lower price points.
Leading Brands (2022-2023)
| Brand Name | Manufacturer | Price Range ($/bottle) | Market Share (%) |
|---|---|---|---|
| Alphagan P (Brimonidine) | Allergan (AbbVie) | 50-70 | 40 |
| Timolol-Brimonidine (generic) | Multiple generics | 20-40 | 15 |
| Combigan (Brimonidine + Timolol) | Novartis | 55-75 | 25 |
| Ocutan (Brimonidine-only) | Several (regional players) | 30-60 | 10 |
| Private labels | Various | 15-25 | 10 |
What Are Price Trends and Projections?
Brimonidine-timolol's prices have remained relatively stable over the past three years but are expected to decline by 2-4% annually due to generic competition starting around 2024-2025 in mature markets.
Price Evolution (2021–2030)
| Year | Average Retail Price ($/bottle) | Percent Change (Annual) |
|---|---|---|
| 2021 | 65 | — |
| 2023 | 62 | -4.6% |
| 2025 | 58 | -6.5% (projected with generic entry) |
| 2027 | 55 | -5.2% |
| 2030 | 50 | -9.1% (approaching generic dominance) |
Factors Influencing Price Decrease:
- Patent expirations, beginning as early as 2024 for some formulations.
- Increased generic market penetration.
- Price optimization in emerging markets to capture volume.
What Is the Competitive Outlook?
The market expects intensified competition from generics after patent expiration, which will exert downward pressure on prices. Companies with early filings hold advantages. Investment in formulation improvements and combination therapies will be paramount for brand differentiation.
Market Entry and Regulatory Landscape
- Generic approval pathways expedited through agencies such as the FDA (United States), EMA (Europe), and PMDA (Japan).
- Regulatory hurdles include demonstrating bioequivalence and safety.
- In emerging markets, regulatory processes are often less stringent, facilitating faster entry but raising quality control issues.
What Are the Key Opportunities and Risks?
Opportunities:
- Expansion into developing markets through cost-effective formulations.
- Development of sustained-release or innovative delivery systems.
- Formulation improvements reducing systemic side effects.
Risks:
- Slower-than-expected generic entry or patent litigation extending exclusivity.
- Market saturation in mature regions.
- Pricing pressures reducing margins.
Key Takeaways
- The global market for brimonidine-timolol was valued around $450 million in 2022, with ongoing growth driven by demographic trends.
- Price declines are anticipated with generic entries, especially after patent expirations—projected at around 2-4% annually.
- Competitive advantage will depend on patent status, formulation differentiation, and regional market strategies.
- Emerging markets present growth opportunities but require tailored pricing and regulatory strategies.
- The competitive landscape will heavily feature generic manufacturers post-2024, with brand players shifting towards innovation and optimized delivery.
FAQs
1. When will brimonidine-timolol patents expire?
Patent expiration dates vary by region and formulation but generally range from 2024 to 2026 in key markets like the US and Europe.
2. How does generic competition influence pricing?
Generic entry typically leads to a 50-70% reduction in price within the first 2 years, exerting downward pressure on branded product prices.
3. Are there ongoing clinical trials for new formulations?
Yes, research includes sustained-release formulations and combination therapies with reduced side effects, though none have yet gained regulatory approval.
4. Which regions are expected to see the fastest market growth?
Asia-Pacific and Latin America are projected to grow fastest, due to increasing glaucoma prevalence and rising healthcare access.
5. What is the impact of regulatory policies on market entry?
Streamlined approval pathways in some markets facilitate fastergeneric entry; however, stringent compliance remains necessary to ensure safety and efficacy.
Sources:
[1] Grand View Research, "Glaucoma Drugs Market Size, Share & Trends Analysis Report," 2022.
[2] IQVIA, "Global Pharmaceutical Market Data," 2022.
[3] U.S. Food and Drug Administration, "ANDA Approvals," 2023.
[4] European Medicines Agency, "Market Authorization Approvals," 2023.
[5] MarketWatch, "Brimonidine-Timolol Price Trends," 2022.
More… ↓
