Share This Page
Drug Price Trends for BEYAZ
✉ Email this page to a colleague
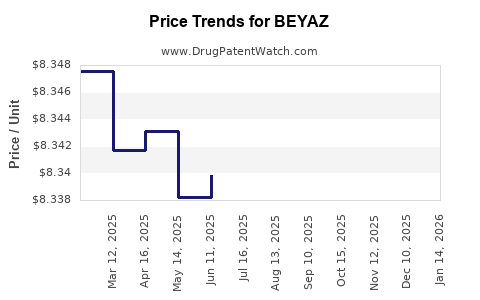
Average Pharmacy Cost for BEYAZ
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BEYAZ 28 TABLET | 50419-0407-01 | 8.28854 | EACH | 2026-03-18 |
| BEYAZ 28 TABLET | 50419-0407-03 | 8.28854 | EACH | 2026-03-18 |
| BEYAZ 28 TABLET | 50419-0407-01 | 8.31212 | EACH | 2026-02-18 |
| BEYAZ 28 TABLET | 50419-0407-03 | 8.31212 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
BEYAZ (Drospirenone/Ethinyl Estradiol/Levomefolate Calcium) Patent Landscape and Market Projections
BEYAZ (drospirenone/ethinyl estradiol/levomefolate calcium) is a combination oral contraceptive. Its patent landscape involves protection for the drug product, its components, and specific manufacturing processes. The market for BEYAZ is influenced by patent expiry, generic competition, and the availability of alternative contraceptive methods.
What are the key patents protecting BEYAZ?
The primary patents protecting BEYAZ and its components are held by Bayer. These patents cover the active pharmaceutical ingredients (APIs), the specific combination formulation, and methods of use.
- Composition of Matter Patents: These patents claim the novel chemical entities themselves. For drospirenone, the original patent was US Patent 4,577,051, filed in 1983 and expiring in 2009. For ethinyl estradiol, it is a well-established compound with expired patents. Levomefolate calcium (the active form of folic acid) is also a known compound with prior art.
- Formulation Patents: Patents specifically claim the BEYAZ tablet formulation, which combines drospirenone, ethinyl estradiol, and levomefolate calcium in a precise ratio. For example, US Patent 7,727,565, covering the combination product and its use, was granted in 2010. The primary expiry for this patent is July 16, 2027.
- Method of Use Patents: These patents protect specific therapeutic uses, such as contraception and potentially managing symptoms associated with conditions like premenstrual dysphoric disorder (PMDD), for which BEYAZ is indicated.
- Manufacturing Process Patents: While less commonly the primary drivers of market exclusivity, patents on specific scalable and efficient manufacturing processes for the APIs or the final drug product can provide a competitive advantage.
The patent expiry dates are critical for understanding market dynamics. The primary formulation patent for BEYAZ is set to expire in July 2027, opening the door for generic manufacturers.
What is the current market status of BEYAZ?
BEYAZ is an established product in the oral contraceptive market. It competes with other combination oral contraceptives, progestin-only pills, and various non-hormonal birth control methods.
- Market Share: Bayer has historically held a significant market share for BEYAZ, driven by its efficacy, safety profile, and the added benefit of levomefolate calcium for women at risk of neural tube defects. Specific market share data fluctuates, but BEYAZ has been a top-selling oral contraceptive.
- Indications: BEYAZ is approved for use as an oral contraceptive to prevent pregnancy and for the treatment of premenstrual dysphoric disorder (PMDD) in women who choose to use an oral contraceptive for this purpose.
- Competition: Key competitors include other drospirenone-containing contraceptives (e.g., YAZ, Yazmin), other combined oral contraceptives (e.g., those containing ethinyl estradiol with different progestins like norgestimate or norethindrone), and generics of these products. The market also sees competition from progestin-only pills, vaginal rings, and transdermal patches.
- Sales Performance: Bayer's sales for its drospirenone-containing products, including BEYAZ and YAZ, have been substantial. For instance, in 2022, Bayer’s Women’s Health division reported significant revenue from its contraceptive portfolio. However, reporting often consolidates sales for multiple drospirenone products, making specific BEYAZ figures proprietary.
The market remains robust due to the consistent demand for reliable contraception. However, the impending patent expiry of key BEYAZ patents suggests a shift toward increased generic penetration.
When is BEYAZ expected to face generic competition?
Generic competition for BEYAZ is anticipated following the expiry of its key formulation patents.
- Key Patent Expiry: US Patent 7,727,565, covering the BEYAZ formulation, is the primary patent that will expire on July 16, 2027.
- Potential for Earlier Entry: While the patent expiry date is definitive, the timeline for generic entry can be influenced by:
- Paragraph IV Certifications: Generic companies may file Abbreviated New Drug Applications (ANDAs) challenging existing patents. If successful, they can achieve an earlier market entry, often with a 180-day exclusivity period.
- Litigation: Patent litigation between the innovator company (Bayer) and potential generic manufacturers can delay or accelerate generic entry.
- ANDA Approval Timelines: The U.S. Food and Drug Administration (FDA) approval process for ANDAs can take several months to over a year.
Based on the July 2027 patent expiry, generic versions of BEYAZ are expected to become available in the U.S. market around mid-2027, assuming no successful Paragraph IV challenges lead to earlier entry or significant delays in FDA approval. Similar patent expiries and generic entry timelines are expected in other major markets.
What are the projected market prices for BEYAZ and its generics?
Price projections are contingent upon generic market entry and the competitive landscape.
- Current Pricing: Branded BEYAZ typically has a higher out-of-pocket cost for patients without comprehensive insurance coverage. Prices can range from $80 to $150+ per month, depending on the pharmacy, insurance plan, and formulary.
- Generic Impact: Upon generic entry, prices for oral contraceptives generally decline significantly.
- Initial Generic Pricing: The first generic entrant often sets a benchmark price, typically 30-50% lower than the branded product.
- Subsequent Price Erosion: As more generic manufacturers enter the market, price competition intensifies, leading to further price reductions of 60-80% or more compared to the branded product within 1-2 years of generic availability.
- Factors Influencing Price:
- Number of Generic Competitors: A crowded generic market leads to lower prices.
- Formulation Complexity: While BEYAZ is a combination pill, the manufacturing process is well-established, allowing for competitive generic production.
- Payer Negotiations: Insurance companies and pharmacy benefit managers negotiate prices, influencing the final cost to consumers.
- Levomefolate Calcium Component: The cost of the levomefolate calcium component may influence the overall cost of generic manufacturing compared to oral contraceptives without this ingredient.
Projected Price Scenarios (Monthly Retail Cost, Out-of-Pocket without Insurance):
| Product Status | Projected Price Range | Notes |
|---|---|---|
| Branded BEYAZ | $80 - $150+ | Current market price, varies by insurer and pharmacy. |
| Generic BEYAZ (First 6-12 months) | $40 - $70 | Assuming 30-50% reduction from branded. |
| Generic BEYAZ (1-2 years post-entry) | $20 - $40 | Assuming 60-80%+ reduction due to increased competition. |
These are retail price estimates and do not reflect wholesale acquisition costs or net prices after rebates and discounts negotiated with payers. The wholesale price is typically significantly lower.
What are the key market dynamics affecting BEYAZ post-patent expiry?
The post-patent expiry market for BEYAZ will be characterized by intensified competition and a shift in market dynamics.
- Increased Generic Penetration: The primary driver will be the introduction of multiple generic versions of BEYAZ. This is common for oral contraceptives once primary patents expire.
- Price Wars: Generic manufacturers will likely engage in aggressive pricing to capture market share, leading to significant price erosion.
- Formulary Placement: Payers will prioritize generic options due to their lower cost, potentially limiting favorable formulary placement for branded BEYAZ.
- Physician Prescribing Habits: While some physicians may continue to prescribe branded BEYAZ for established patients, the cost advantage of generics will encourage a shift towards generic prescriptions, especially for new patients.
- Patient Out-of-Pocket Costs: Patients will benefit from significantly lower co-pays for generic BEYAZ.
- Innovation by Bayer: Bayer may seek to mitigate the impact of generic competition by:
- Developing new formulations or delivery methods with distinct patent protection.
- Focusing marketing and sales efforts on other products in its portfolio.
- Leveraging its established relationships with healthcare providers and payers.
- Continued Demand for Drospirenone: Drospirenone is a widely used progestin in oral contraceptives due to its anti-androgenic and anti-mineralocorticoid properties. Demand for drospirenone-based contraceptives, including generics, is expected to remain strong.
The market for BEYAZ will transition from a branded monopoly (or oligopoly with other drospirenone products) to a highly competitive generic space. Companies with efficient manufacturing and strong distribution networks will be best positioned.
What are the regulatory considerations for generic BEYAZ?
The regulatory pathway for generic BEYAZ in the United States involves the Abbreviated New Drug Application (ANDA) process administered by the U.S. Food and Drug Administration (FDA).
- ANDA Submission: Generic manufacturers must submit an ANDA demonstrating that their product is bioequivalent to the reference listed drug (RLD), which is BEYAZ in this case.
- Bioequivalence: Bioequivalence studies compare the rate and extent to which the generic drug is absorbed into the bloodstream relative to the RLD. This ensures that the generic drug will have the same therapeutic effect.
- Patent Certification: As part of the ANDA, generic companies must certify their position regarding any patents listed in the FDA's Orange Book for the RLD. This typically involves certifying that the patents are expired, invalid, or will not be infringed by the generic product.
- Paragraph IV Certification: If a generic company certifies that a listed patent is invalid, unenforceable, or will not be infringed, this is a Paragraph IV certification. This can trigger a 30-month stay of FDA approval if the patent holder sues for infringement, unless certain conditions are met.
- Labeling: Generic products must have the same active ingredients, dosage form, strength, and route of administration as the RLD. The labeling must also be the same as the RLD’s labeling, except for minor changes not affecting safety or efficacy.
- Quality Standards: Generic manufacturers must adhere to Current Good Manufacturing Practices (cGMP) to ensure the quality, safety, and efficacy of their products.
The regulatory hurdles are generally well-defined for oral contraceptives. The primary challenge for generic companies often lies in navigating the patent landscape and potential litigation.
What is the potential market size for generic BEYAZ?
Estimating the precise market size for generic BEYAZ requires access to proprietary sales data for the branded product and detailed market research on oral contraceptive usage patterns. However, a qualitative assessment can be made.
- BEYAZ as a Leading Product: BEYAZ has been a significant product in the oral contraceptive market. Its established brand recognition and physician acceptance contribute to its market presence.
- Market Value of Oral Contraceptives: The global market for oral contraceptives is substantial, valued in the billions of dollars annually. The U.S. market represents a significant portion of this.
- Cannibalization Effect: Generic entry will lead to a significant price reduction, meaning that while the volume of units sold may increase, the overall dollar value of the BEYAZ market (branded + generic) might initially decrease due to price compression.
- Share of Drospirenone Market: BEYAZ is one of several drospirenone-containing products. Its generic version will compete not only with other BEYAZ generics but also with generics of other drospirenone products and alternative contraceptive options.
Assuming BEYAZ (and its direct generic competitors) holds a meaningful segment of the combined oral contraceptive market, the generic market for BEYAZ, once fully established, could represent several hundred million dollars in annual sales within the U.S. alone, given typical price erosion and volume increases seen with other generic oral contraceptives. This figure is an estimate and subject to market dynamics.
Key Takeaways
- The primary patent protecting BEYAZ's formulation expires in July 2027, paving the way for generic competition.
- Generic entry is expected around mid-2027, with potential for earlier entry based on Paragraph IV challenges and litigation.
- Branded BEYAZ currently retails for approximately $80-$150+ per month; generic versions are projected to range from $40-$70 initially, dropping to $20-$40 within two years.
- The market will shift from a branded product to a highly competitive generic landscape, driven by price wars and payer prioritization of generics.
- Regulatory approval for generic BEYAZ will follow the FDA's ANDA process, requiring demonstration of bioequivalence to the reference listed drug.
Frequently Asked Questions
1. Will BEYAZ be available as a generic before its primary patent expires in July 2027?
Generic entry before the primary patent expiry is possible if a generic manufacturer successfully challenges the patent through a Paragraph IV certification and wins patent litigation or if the patent holder does not file an infringement suit within the specified timeframe. However, the most probable scenario for widespread generic availability is post-expiry.
2. How will the availability of levomefolate calcium in BEYAZ affect the price of its generics?
The inclusion of levomefolate calcium adds a component that requires specific manufacturing and sourcing. While this may slightly increase the cost of goods sold for generic manufacturers compared to oral contraceptives without this ingredient, the highly competitive nature of the generic oral contraceptive market typically drives prices down significantly, absorbing much of this incremental cost.
3. What is the likelihood of Bayer launching its own generic version of BEYAZ (a "first-to-file" generic)?
It is common for innovator companies to develop and launch their own generic versions of their branded products to capture a share of the generic market and mitigate the impact of third-party generic entry. Bayer may pursue this strategy, which would likely involve a slightly different branded product name for the generic version.
4. Are there any remaining patents that could extend BEYAZ's market exclusivity beyond July 2027?
While the primary formulation patent expires in July 2027, there may be other patents covering specific manufacturing processes, polymorphic forms, or methods of use that could theoretically extend exclusivity. However, these are generally less robust in preventing the entry of a bioequivalent generic product compared to a core formulation patent. Generic companies will rigorously review all listed patents.
5. What impact will the generic availability of BEYAZ have on other drospirenone-containing contraceptives?
The generic availability of BEYAZ will contribute to a broader trend of increasing generic competition within the drospirenone-containing oral contraceptive market. This will intensify competition among all drospirenone products, both branded and generic, potentially leading to further price erosion across the entire class of drospirenone-based contraceptives.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Orange Book. Retrieved from https://www.accessdata.fda.gov/scripts/cder/ob/ [2] Bayer AG. (2023). Annual Report 2022. Retrieved from Bayer investor relations website. [3] U.S. Patent and Trademark Office. (n.d.). Patent Full-Text and Image Database. (Accessed for specific patent numbers and grant/expiry dates).
More… ↓
