Share This Page
Drug Price Trends for BENZOYL PEROXIDE
✉ Email this page to a colleague
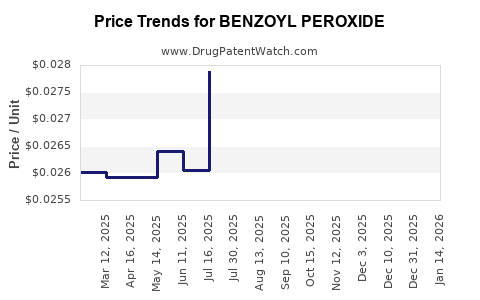
Average Pharmacy Cost for BENZOYL PEROXIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BENZOYL PEROXIDE 10% GEL | 45802-0308-96 | 0.22952 | GM | 2026-04-22 |
| BENZOYL PEROXIDE 10% WASH | 35573-0454-08 | 0.04665 | GM | 2026-04-22 |
| BENZOYL PEROXIDE 10% WASH | 35573-0454-91 | 0.04901 | GM | 2026-04-22 |
| BENZOYL PEROXIDE 10% WASH | 00536-1351-42 | 0.04665 | GM | 2026-04-22 |
| BENZOYL PEROXIDE 10% GEL | 45802-0308-01 | 0.18794 | GM | 2026-04-22 |
| BENZOYL PEROXIDE 2.5% GEL | 45802-0101-96 | 0.21380 | GM | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for BENZOYL PEROXIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BENZOYL PEROXIDE 5%/CLINDAMYCIN PO4 1% GEL JR | Golden State Medical Supply, Inc. | 51672-1381-04 | 50GM | 23.52 | 0.47040 | GM | 2023-06-15 - 2028-06-14 | FSS |
| BENZOYL PEROXIDE 5%/CLINDAMYCIN PO4 1% GEL JR | Golden State Medical Supply, Inc. | 51672-1381-04 | 50GM | 24.08 | 0.48160 | GM | 2023-06-23 - 2028-06-14 | FSS |
| BENZOYL PEROXIDE 5%/CLINDAMYCIN PO4 1% GEL PU | Golden State Medical Supply, Inc. | 51672-1381-05 | 35GM | 33.67 | 0.96200 | GM | 2023-06-15 - 2028-06-14 | FSS |
| BENZOYL PEROXIDE 5%/CLINDAMYCIN PO4 1.2% GEL, | Golden State Medical Supply, Inc. | 51672-1366-06 | 45GM | 17.91 | 0.39800 | GM | 2023-12-15 - 2028-06-14 | FSS |
| BENZOYL PEROXIDE 5%/CLINDAMYCIN PO4 1% GEL PU | Golden State Medical Supply, Inc. | 51672-1381-05 | 35GM | 34.47 | 0.98486 | GM | 2023-06-23 - 2028-06-14 | FSS |
| BENZOYL PEROXIDE 2.5%/CLINDAMYCIN PO4 1.2% PU | Golden State Medical Supply, Inc. | 51672-1367-03 | 50GM | 97.15 | 1.94300 | GM | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Benzoyl Peroxide Market Analysis and Price Projections
Benzoyl peroxide (BPO) is a key active pharmaceutical ingredient (API) primarily used in the dermatological treatment of acne vulgaris. Its market is driven by the prevalence of acne, consumer demand for over-the-counter (OTC) and prescription treatments, and ongoing R&D in dermatological formulations. Price projections are influenced by raw material costs, manufacturing efficiencies, and market competition.
What is the Global Market Size and Growth Outlook for Benzoyl Peroxide?
The global benzoyl peroxide market was valued at approximately USD 950 million in 2023. It is projected to expand at a compound annual growth rate (CAGR) of 4.2% from 2024 to 2030, reaching an estimated USD 1.28 billion by the end of the forecast period. This growth is attributed to an increasing prevalence of acne globally, particularly among adolescents and young adults, coupled with a rising demand for effective and accessible acne treatments. The accessibility of BPO in OTC products contributes significantly to market penetration.
Key Market Drivers:
- Prevalence of Acne Vulgaris: Acne is one of the most common skin conditions, affecting an estimated 85% of adolescents and a significant portion of the adult population worldwide. This sustained high prevalence directly fuels demand for BPO-based treatments [1].
- Growth in Dermatological Treatments: The global dermatology market, encompassing prescription drugs, OTC products, and aesthetic procedures, is experiencing robust growth. BPO is a foundational ingredient in many topical acne therapies within this market [2].
- Consumer Awareness and Self-Care Trends: Increased public awareness regarding skin health and the efficacy of BPO has driven consumer demand for self-administered treatments. The availability of BPO in various OTC formulations supports this trend.
- Advancements in Formulation Technology: Research and development in pharmaceutical formulations are leading to improved BPO delivery systems, enhanced efficacy, and reduced side effects, potentially expanding its application and market appeal.
- Emerging Markets: Growing populations and increasing disposable incomes in developing regions contribute to a rising demand for healthcare products, including dermatological treatments like BPO.
Market Restraints:
- Competition from Alternative Treatments: The market faces competition from other acne treatment modalities, including topical retinoids, antibiotics, oral medications, and newer combination therapies, which may offer different efficacy profiles or fewer side effects for certain patient populations.
- Skin Irritation and Side Effects: While generally well-tolerated, BPO can cause side effects such as dryness, redness, peeling, and stinging, which can limit patient compliance and deter some individuals from using it.
- Regulatory Scrutiny and Manufacturing Costs: Stringent regulatory requirements for API manufacturing and quality control can increase production costs. Fluctuations in the cost of raw materials, such as benzoyl chloride and hydrogen peroxide, also impact profitability.
What are the Key Product Segments and Their Market Share?
The benzoyl peroxide market can be segmented by product form and application.
By Product Form:
- Powder: This is the raw API form, used by pharmaceutical manufacturers to formulate various topical preparations. The powder segment is crucial for the supply chain, with its market share dictated by the volume of finished products manufactured.
- Gels: BPO gels are a popular topical formulation due to their ease of application and absorption. This segment accounts for a significant portion of the market.
- Creams and Lotions: These formulations are also widely used, offering a less occlusive option than gels for some users.
- Cleansers and Washes: BPO is incorporated into facial cleansers and body washes for daily acne management. This segment is growing due to convenience.
Estimated Market Share by Product Form (2023):
| Product Form | Estimated Market Share |
|---|---|
| Gels | 35% |
| Creams and Lotions | 30% |
| Cleansers and Washes | 20% |
| Powder | 15% |
By Application:
- Acne Treatment: This is the dominant application, encompassing both OTC and prescription products.
- Other Dermatological Conditions: BPO also finds limited application in treating other conditions like fungal infections and certain skin ulcers.
Estimated Market Share by Application (2023):
| Application | Estimated Market Share |
|---|---|
| Acne Treatment | 95% |
| Other Applications | 5% |
Who are the Key Manufacturers and What is Their Competitive Landscape?
The benzoyl peroxide market is moderately consolidated, with several global and regional players. Key manufacturers focus on API production, formulation, and marketing of BPO-based dermatological products. Competition is driven by product quality, pricing, regulatory compliance, and innovation in formulation.
Leading Manufacturers:
- Peroxychem, LLC (a wholly-owned subsidiary of Evonik Industries AG): A major global producer of peroxygen chemicals, including BPO.
- United Active Ingredients Inc.: A supplier of APIs, including BPO, to the pharmaceutical industry.
- Solvay SA: A chemical company with a significant presence in the peroxygens market.
- BASF SE: A diversified chemical company that produces BPO as part of its portfolio.
- Honokiol Pharmaceuticals: A significant player in the Asian market for APIs.
- Salicylates and Chemicals Pvt. Ltd.: An Indian manufacturer of pharmaceutical ingredients.
- KAO Corporation: A consumer goods company that also produces BPO for cosmetic and pharmaceutical applications.
- Galderma S.A.: A pharmaceutical company specializing in dermatology, offering BPO-containing prescription and OTC products.
- Johnson & Johnson Consumer Inc.: A major consumer healthcare company with a wide range of BPO-based acne products.
- L'Oréal S.A.: A global beauty company that incorporates BPO into its skincare lines.
The competitive landscape includes API manufacturers and finished product formulators. Pricing strategies vary based on production scale, raw material costs, and geographic market penetration. Mergers, acquisitions, and strategic partnerships are observed as companies aim to expand their market reach and product portfolios.
What are the Key Raw Materials and Their Price Trends?
The primary raw materials for benzoyl peroxide synthesis are benzoyl chloride and hydrogen peroxide. The cost and availability of these precursors significantly influence BPO manufacturing costs.
- Benzoyl Chloride: Derived from benzoic acid, its price is linked to the petrochemical market and the cost of toluene. Fluctuations in crude oil prices can indirectly impact benzoyl chloride costs.
- Hydrogen Peroxide: A widely produced industrial chemical. Its price is influenced by energy costs for production and regional supply-demand dynamics.
Price Trends and Projections (2024-2028):
Raw material prices are subject to volatility. Over the next few years, BPO manufacturers can expect moderate price increases for key raw materials driven by:
- Energy Costs: Continued energy price volatility, particularly natural gas and electricity, will affect the cost of producing hydrogen peroxide and other chemical intermediates.
- Supply Chain Disruptions: Geopolitical factors and logistics challenges can lead to temporary spikes in raw material prices.
- Demand Growth: Increasing demand for industrial chemicals globally can put upward pressure on prices.
However, these increases are expected to be partially offset by:
- Manufacturing Efficiencies: Ongoing investments in process optimization and automation by BPO manufacturers can help absorb some raw material cost increases.
- Scale of Production: Larger-scale API manufacturers often benefit from economies of scale, allowing for more competitive pricing.
General Projection: An average annual increase of 2-4% in raw material costs is anticipated for benzoyl chloride and hydrogen peroxide. This translates to a potential upward pressure on BPO API prices.
What are the Projected Pricing Strategies and Levels for Benzoyl Peroxide?
Benzoyl peroxide pricing is influenced by several factors, including API grade, concentration, formulation type, brand, and distribution channel.
API Pricing:
- Bulk API (Powder): Prices for bulk benzoyl peroxide API are typically quoted per kilogram and vary based on purity, volume purchased, and supplier.
- Estimated Price Range (2024): USD 15 - USD 35 per kilogram. This range reflects differences in manufacturing scale, quality certifications (e.g., GMP compliance), and supplier.
- Price Trend for API: A gradual increase of 3-5% per year is projected for bulk BPO API, primarily driven by rising raw material and manufacturing costs, and increasing demand for finished products.
Finished Product Pricing (Consumer Level):
- Over-the-Counter (OTC) Products:
- Cleansers/Washes (e.g., 5% BPO): USD 10 - USD 25 per unit (e.g., 4-6 oz bottle).
- Gels/Creams (e.g., 2.5% - 10% BPO): USD 15 - USD 35 per unit (e.g., 1-2 oz tube/jar).
- Price Trend for OTC: Moderate increases of 2-4% per year are expected, driven by formulation costs, marketing expenses, and retail markups.
- Prescription Products:
- Pricing is highly variable and influenced by the specific product, concentration, combination with other APIs, and insurance coverage. Prescription products containing BPO, especially in combination therapies, can range from USD 50 to USD 150+ per prescription.
- Price Trend for Prescription: Similar to other prescription pharmaceuticals, price increases are subject to market dynamics, patent exclusivity (if any on specific formulations), and payer negotiations, typically seeing annual increases in the 4-8% range.
Factors Influencing Pricing:
- Concentration: Higher concentrations of BPO (e.g., 10%) generally command higher prices than lower concentrations (e.g., 2.5%).
- Formulation: Advanced formulations (e.g., sustained-release, microencapsulated) or combination products with other active ingredients will be priced at a premium.
- Brand Recognition and Marketing: Established brands with significant marketing investment often sustain higher price points.
- Distribution Channels: Direct-to-consumer (DTC) online sales may offer different pricing than traditional brick-and-mortar retail or pharmacy channels.
- Geographic Market: Pricing can vary significantly by region due to local market conditions, regulatory environments, and competitive intensity.
What are the Regulatory Considerations and Their Market Impact?
Benzoyl peroxide is regulated as both an Active Pharmaceutical Ingredient (API) and a cosmetic ingredient, depending on its intended use and formulation. Regulatory oversight by bodies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) impacts manufacturing, labeling, and marketing.
Key Regulatory Aspects:
- API Manufacturing Standards: BPO manufacturers producing API for pharmaceutical use must adhere to Good Manufacturing Practices (GMP). Compliance involves rigorous quality control, validation of manufacturing processes, and detailed documentation, increasing operational costs.
- OTC Drug Monograph System (USA): In the United States, BPO is an established OTC drug ingredient. Its use in various concentrations (e.g., up to 10% for acne treatment) is permitted under FDA's OTC Drug Monograph system, allowing for wider availability without individual New Drug Applications (NDAs) for basic formulations. Changes to these monographs, though rare, could impact market access.
- Cosmetic Ingredient Regulations: When used in cosmetic products at lower concentrations for mild exfoliation or as a preservative, BPO is subject to cosmetic regulations, which are generally less stringent than pharmaceutical regulations.
- Safety and Efficacy Data: Manufacturers must maintain and provide data demonstrating the safety and efficacy of their BPO products. Any adverse event reports or new safety concerns can trigger regulatory reviews and potential market restrictions.
- Labeling Requirements: Clear and accurate labeling regarding concentration, usage instructions, warnings, and potential side effects is mandated for both pharmaceutical and cosmetic products.
Market Impact of Regulations:
- Barriers to Entry: GMP compliance and the need for regulatory dossiers create significant barriers to entry for new API manufacturers, favoring established players with existing infrastructure and expertise.
- Quality Assurance: Regulatory requirements ensure a baseline level of product quality and safety, which can build consumer trust.
- Cost of Compliance: Adhering to GMP and other regulatory standards adds substantial costs to manufacturing and product development, which are often passed on to consumers through pricing.
- Market Access: Products that meet all regulatory requirements gain access to established sales channels, including pharmacies and regulated retail outlets. Non-compliance can lead to product recalls or market exclusion.
- Innovation Constraints: While regulations ensure safety, they can sometimes slow down the approval process for novel BPO formulations or combinations, impacting the speed of innovation.
What are the Future Trends and Opportunities in the Benzoyl Peroxide Market?
The benzoyl peroxide market is poised for continued growth, with several trends shaping its future:
Key Future Trends:
- Combination Therapies: The development and increased adoption of BPO in combination with other acne-fighting ingredients (e.g., retinoids, antibiotics, niacinamide) are a significant trend. These synergistic formulations offer broader efficacy and improved patient outcomes.
- Advanced Delivery Systems: Research into novel delivery systems such as microencapsulation, liposomes, and sustained-release technologies aims to enhance BPO's efficacy while minimizing irritation and improving tolerability. This will drive demand for premium formulations.
- Focus on Sensitive Skin Formulations: Addressing the skin irritation associated with BPO is a key area of innovation. Manufacturers are developing gentler formulations with lower concentrations, buffering agents, and moisturizing ingredients to cater to a wider range of skin types, including sensitive skin.
- Increased Demand in Emerging Markets: As healthcare access and consumer spending power rise in developing economies, the demand for effective and affordable acne treatments, including BPO, is expected to grow substantially.
- Sustainability in Manufacturing: Growing consumer and regulatory pressure for sustainable manufacturing practices may drive innovation in greener synthesis routes for BPO and more eco-friendly packaging.
Opportunities:
- Development of Novel Formulations: Companies that can successfully develop and commercialize BPO formulations with improved efficacy and reduced side effects will capture significant market share. This includes exploring new combinations and advanced delivery mechanisms.
- Expansion in Emerging Markets: Targeting emerging economies in Asia, Latin America, and Africa presents a substantial growth opportunity for both API manufacturers and finished product brands.
- Pediatric and Adult Acne Solutions: While acne is common in adolescents, adult acne is also a growing concern. Tailoring BPO treatments for these distinct demographics can unlock new market segments.
- Partnerships and Collaborations: Strategic alliances between API suppliers and finished product formulators can streamline product development and market entry. Collaborations with dermatologists and research institutions can validate new formulations and drive clinical adoption.
- Product Diversification: Expanding beyond traditional acne treatments to explore BPO's potential in other dermatological areas or as a preservative in specific cosmetic applications could offer diversification.
Key Takeaways
The benzoyl peroxide market is projected to reach USD 1.28 billion by 2030, driven by the persistent high prevalence of acne and growing demand for accessible dermatological treatments. Key product segments include gels, creams, lotions, and cleansers, with acne treatment dominating applications. The market is moderately competitive, featuring global API producers and major consumer healthcare companies. Raw material costs for benzoyl chloride and hydrogen peroxide are expected to see modest increases, influencing API pricing. Finished product pricing will reflect formulation, brand, and distribution. Regulatory compliance with GMP standards is critical for API manufacturers, while OTC monograph status in the U.S. facilitates broad market access for certain BPO products. Future opportunities lie in the development of advanced combination therapies and delivery systems, expansion into emerging markets, and the creation of gentler formulations for sensitive skin.
Frequently Asked Questions
1. What is the projected CAGR for the benzoyl peroxide market from 2024 to 2030?
The projected CAGR for the benzoyl peroxide market from 2024 to 2030 is 4.2%.
2. What are the primary raw materials used in the synthesis of benzoyl peroxide?
The primary raw materials used in the synthesis of benzoyl peroxide are benzoyl chloride and hydrogen peroxide.
3. In which product form is benzoyl peroxide most prevalent in the market?
Benzoyl peroxide is most prevalent in gel formulations, accounting for an estimated 35% of the market share by product form in 2023.
4. How do regulatory considerations impact the cost of benzoyl peroxide API manufacturing?
Regulatory considerations, such as adherence to Good Manufacturing Practices (GMP), require rigorous quality control, validation, and documentation, which significantly increase the operational costs for API manufacturers.
5. What is a key future trend expected to shape the benzoyl peroxide market?
A key future trend is the development and increased adoption of combination therapies, where benzoyl peroxide is used alongside other active ingredients for synergistic effects in acne treatment.
Citations
[1] American Academy of Dermatology Association. (n.d.). Acne. Retrieved from https://www.aad.org/media/stats-skin
[2] Grand View Research. (2023). Dermatology Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com/industry-analysis/dermatology-market
More… ↓
