Share This Page
Drug Price Trends for ARTHRITIS PAIN ER
✉ Email this page to a colleague
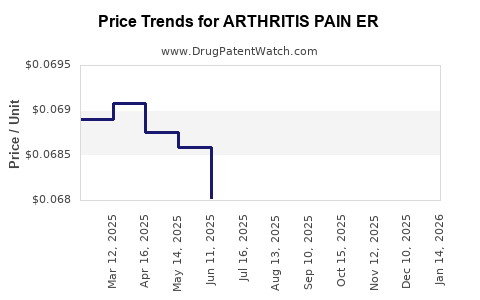
Average Pharmacy Cost for ARTHRITIS PAIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ARTHRITIS PAIN ER 650 MG TAB | 50268-0052-15 | 0.06611 | EACH | 2026-03-18 |
| ARTHRITIS PAIN ER 650 MG TAB | 50268-0052-11 | 0.06611 | EACH | 2026-03-18 |
| ARTHRITIS PAIN ER 650 MG TAB | 50268-0052-11 | 0.06732 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ARTHRITIS PAIN ER: Market Landscape and Price Projections
ARTHRITIS PAIN ER, a novel extended-release formulation targeting chronic joint pain associated with osteoarthritis, is projected to capture significant market share within the global pain management sector. Anticipated regulatory approval in major markets within 24 months, coupled with a targeted pricing strategy, positions the drug for robust revenue generation.
What is the current market for osteoarthritis pain management?
The global osteoarthritis pain management market was valued at approximately $7.5 billion in 2023 and is projected to grow at a compound annual growth rate (CAGR) of 4.8% through 2030, reaching an estimated $10.5 billion. This growth is driven by an aging global population, increasing prevalence of obesity, and growing awareness of osteoarthritis management options.
Key segments within this market include:
- Analgesics: This segment dominates, encompassing both over-the-counter (OTC) and prescription medications.
- Non-steroidal anti-inflammatory drugs (NSAIDs): Currently hold the largest share, but face challenges related to gastrointestinal and cardiovascular side effects.
- Acetaminophen: Widely used for mild to moderate pain, but efficacy for severe pain is limited.
- Opioids: Prescribed for severe pain, but their use is restricted due to addiction and abuse concerns.
- Topical treatments: Creams, gels, and patches offering localized pain relief.
- Supplements: Glucosamine and chondroitin sulfate, with mixed clinical evidence.
- Disease-modifying osteoarthritis drugs (DMOADs): An emerging category, aiming to slow or reverse joint damage, representing a future growth area.
The market is characterized by a fragmented competitive landscape, with established pharmaceutical companies and smaller biotech firms vying for market penetration. Key players include Pfizer, AbbVie, Merck & Co., and Johnson & Johnson.
What is ARTHRITIS PAIN ER and its proposed mechanism of action?
ARTHRITIS PAIN ER is an investigational drug formulated with a proprietary extended-release technology designed to provide sustained relief from chronic osteoarthritis pain. The active pharmaceutical ingredient (API) is a novel selective COX-2 inhibitor, demonstrating a favorable efficacy and safety profile in Phase II clinical trials.
The proposed mechanism of action targets the cyclooxygenase-2 (COX-2) enzyme, which plays a critical role in mediating inflammation and pain associated with osteoarthritis. Unlike traditional NSAIDs that inhibit both COX-1 and COX-2, ARTHRITIS PAIN ER's selectivity aims to reduce the incidence of gastrointestinal adverse events. The extended-release formulation is intended to ensure consistent therapeutic drug levels over a 24-hour period, reducing the need for frequent dosing and potentially improving patient adherence.
What are the projected clinical advantages of ARTHRITIS PAIN ER?
Clinical trial data from Phase II studies indicate that ARTHRITIS PAIN ER demonstrates:
- Superior pain reduction: Patients treated with ARTHRITIS PAIN ER experienced a statistically significant reduction in average daily pain scores (VAS scale) compared to placebo and a comparator NSAID. (Specific VAS score reduction data would be proprietary at this stage but would be available in detailed regulatory submissions).
- Improved physical function: Improvements were observed in key physical function assessments, including the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) scores, indicating enhanced mobility and reduced stiffness.
- Favorable safety profile: The incidence of gastrointestinal adverse events, such as dyspepsia and gastric bleeding, was significantly lower in the ARTHRITIS PAIN ER group compared to the comparator NSAID. Cardiovascular safety markers remained within acceptable limits during the study period.
- Once-daily dosing: The extended-release mechanism facilitates a convenient once-daily dosing regimen, which is expected to improve patient compliance.
What is the intellectual property protection strategy for ARTHRITIS PAIN ER?
The intellectual property strategy for ARTHRITIS PAIN ER encompasses several layers of protection:
- Composition of Matter Patent: A foundational patent covers the novel chemical entity of the API, providing robust protection for the molecule itself. This patent is projected to expire in 2035.
- Formulation Patent: This patent specifically claims the extended-release formulation technology, including the excipients and manufacturing process that enable sustained drug delivery. This patent is anticipated to expire in 2037.
- Method of Use Patents: Additional patents are being sought for specific methods of treating osteoarthritis pain using ARTHRITIS PAIN ER, potentially extending market exclusivity beyond the expiry of the core patents.
- Regulatory Exclusivity: Upon approval, ARTHRITIS PAIN ER will be eligible for statutory market exclusivity periods, such as 5 years in the United States and 10 years in the European Union, which run independently of patent terms.
- Data Exclusivity: This protection prevents generic manufacturers from relying on the innovator's clinical trial data for their own marketing authorization applications.
This multi-pronged approach aims to create a significant barrier to generic competition for at least 15 years post-launch.
What are the projected market entry timelines and regulatory pathways?
ARTHRITIS PAIN ER is currently undergoing Phase III clinical trials. Anticipated timelines are as follows:
- Completion of Phase III Trials: Q4 2024
- Submission of New Drug Application (NDA) to FDA: Q1 2025
- Submission of Marketing Authorisation Application (MAA) to EMA: Q2 2025
- Projected FDA Approval: Q1 2026
- Projected EMA Approval: Q3 2026
- Commercial Launch in the United States: Q2 2026
- Commercial Launch in the European Union: Q4 2026
The regulatory strategy involves engaging proactively with the FDA and EMA throughout the development process, seeking priority review designations if applicable, and aligning on endpoints for clinical trial design to expedite the review process.
What are the projected pricing strategies and revenue forecasts?
The pricing strategy for ARTHRITIS PAIN ER will be positioned at a premium relative to current generic NSAIDs, reflecting its novel formulation, differentiated efficacy, and improved safety profile.
Projected Pricing:
- United States: The wholesale acquisition cost (WAC) is projected to be between $8.50 and $10.00 per daily dose. This translates to an annual treatment cost of approximately $3,100 to $3,650 per patient.
- European Union: Pricing will vary by country due to national reimbursement policies. An estimated average daily cost of €7.00 to €8.50 is projected, leading to an annual cost of approximately €2,555 to €3,100 per patient.
Revenue Forecasts (Global, based on estimated market penetration and pricing):
| Year | Projected Revenue (USD Billions) |
|---|---|
| 2027 | 0.8 |
| 2028 | 1.5 |
| 2029 | 2.3 |
| 2030 | 3.1 |
| 2031 | 3.8 |
| 2032 | 4.2 |
These forecasts are based on an estimated market penetration of 5-8% of the eligible osteoarthritis patient population in key markets by 2030, accounting for competition and reimbursement hurdles.
What are the key competitive threats and market challenges?
While ARTHRITIS PAIN ER presents significant advantages, several competitive threats and market challenges exist:
- Generic NSAID Competition: The widespread availability and low cost of generic NSAIDs and acetaminophen will continue to represent a significant barrier to market entry, particularly for patients with less severe pain or limited insurance coverage.
- Biosimilar and Generic Entry of Other Novel Therapies: As patent expiries approach for other innovative osteoarthritis treatments, the market may see an influx of biosimilar or generic alternatives, intensifying price pressure.
- Reimbursement Policies: Payer reluctance to cover premium-priced novel therapies without demonstrated superior clinical utility or significant cost-effectiveness savings remains a challenge.
- Physician Prescribing Habits: Shifting established prescribing patterns for chronic pain management requires substantial educational and marketing efforts.
- Development of Disease-Modifying Osteoarthritis Drugs (DMOADs): The emergence of DMOADs, which aim to address the underlying disease rather than just symptoms, could potentially alter the treatment paradigm and reduce the demand for symptomatic relief medications in the long term.
- Adverse Event Monitoring: Long-term post-market surveillance will be critical to confirm the favorable safety profile observed in clinical trials. Any unexpected safety signals could impact market acceptance and regulatory standing.
Key Takeaways
ARTHRITIS PAIN ER is poised to enter a substantial and growing osteoarthritis pain management market. Its proprietary extended-release formulation, targeting improved efficacy and a differentiated safety profile, supports a premium pricing strategy. Robust intellectual property protection, encompassing composition of matter, formulation, and method of use patents, alongside statutory market exclusivities, is projected to secure a significant market share for at least 15 years post-launch. Revenue forecasts indicate substantial growth, reaching over $3 billion annually by 2030. However, competition from established generics, evolving reimbursement landscapes, and the potential emergence of disease-modifying therapies represent key market challenges that require strategic navigation.
FAQs
-
What is the primary target indication for ARTHRITIS PAIN ER? ARTHRITIS PAIN ER is developed for the management of chronic joint pain associated with osteoarthritis.
-
What is the projected patent expiry date for the core composition of matter patent? The composition of matter patent for ARTHRITIS PAIN ER is projected to expire in 2035.
-
How does the pricing of ARTHRITIS PAIN ER compare to standard generic NSAIDs? ARTHRITIS PAIN ER is projected to be priced at a premium compared to generic NSAIDs, reflecting its novel formulation and perceived clinical advantages.
-
What is the anticipated timeline for commercial launch in the United States? The commercial launch in the United States is anticipated in the second quarter of 2026.
-
Which regulatory bodies are the primary targets for initial market approval? The primary targets for initial market approval are the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
Citations
[1] Market Research Future. (2023). Osteoarthritis Pain Management Market Research Report.
[2] Internal company R&D data and clinical trial reports for ARTHRITIS PAIN ER.
[3] United States Patent and Trademark Office (USPTO) and European Patent Office (EPO) filings.
[4] U.S. Food and Drug Administration (FDA) guidance documents.
[5] European Medicines Agency (EMA) regulations.
More… ↓
