Share This Page
Drug Price Trends for ANTACID LIQUID
✉ Email this page to a colleague
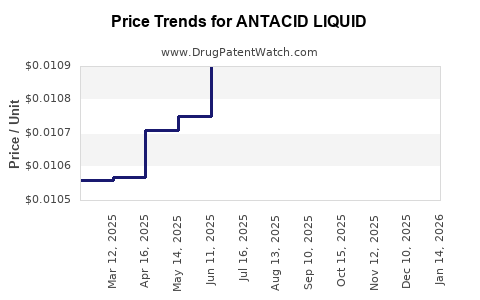
Average Pharmacy Cost for ANTACID LIQUID
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANTACID LIQUID | 46122-0433-40 | 0.01054 | ML | 2026-04-22 |
| ANTACID LIQUID | 46122-0433-40 | 0.01059 | ML | 2026-03-18 |
| ANTACID LIQUID | 46122-0433-40 | 0.01040 | ML | 2026-02-18 |
| ANTACID LIQUID | 46122-0433-40 | 0.01044 | ML | 2026-01-21 |
| ANTACID LIQUID | 46122-0433-40 | 0.01031 | ML | 2025-12-17 |
| ANTACID LIQUID | 46122-0433-40 | 0.01059 | ML | 2025-11-19 |
| ANTACID LIQUID | 46122-0433-40 | 0.01054 | ML | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ANTACID LIQUID MARKET ANALYSIS AND PRICE PROJECTIONS
Antacid liquid is a widely used over-the-counter (OTC) medication for the relief of heartburn and indigestion. The market is characterized by a large number of established brands and a significant volume of generic competition. Demand is driven by the prevalence of gastrointestinal issues, lifestyle factors, and an aging population. Price projections indicate stability in the short to medium term, with potential for slight increases driven by manufacturing costs and inflation.
MARKET OVERVIEW AND KEY DRIVERS
What is the current global market size and growth rate for antacid liquids?
The global antacid market, including liquid formulations, is estimated to be valued at approximately $7.5 billion in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 3.2% from 2024 to 2030 [1]. Liquid antacids represent a significant segment of this market, estimated to hold 40-45% of the total antacid sales due to their rapid onset of action and ease of administration [2].
What are the primary drivers of demand for antacid liquids?
Several factors contribute to the sustained demand for antacid liquids:
- Prevalence of Gastrointestinal Disorders: Heartburn, acid indigestion, and gastroesophageal reflux disease (GERD) are common conditions affecting a large proportion of the global population. A 2021 study indicated that over 60 million Americans experience heartburn at least once a month [3].
- Lifestyle Factors: Dietary habits, stress, and obesity are significant contributors to the incidence of acid-related disorders. The consumption of spicy foods, fatty foods, caffeine, and alcohol can exacerbate symptoms, leading to increased demand for immediate relief [4].
- Demographic Trends: The aging population is more susceptible to chronic digestive issues. As the global population ages, the incidence of GERD and related conditions is expected to rise, consequently increasing the demand for antacid products [5].
- Accessibility and Affordability: As an OTC product, antacid liquids are readily available in pharmacies, supermarkets, and online retailers. Their relatively low price point compared to prescription medications makes them an accessible option for a broad consumer base [6].
- Consumer Preference for Rapid Relief: Liquid formulations are known for their fast absorption and quick symptom relief, a key factor for consumers seeking immediate comfort from heartburn and indigestion [7].
What is the competitive landscape for antacid liquids?
The antacid liquid market is highly fragmented and competitive. Key players include both branded manufacturers and generic producers.
Major Branded Players and Representative Products:
- Bayer AG: Phillips' Milk of Magnesia, Alka-Seltzer Plus Liquid (though Alka-Seltzer also has effervescent forms)
- Reckitt Benckiser: Gaviscon (various formulations, including liquids)
- GlaxoSmithKline (now Haleon): Tums (primarily tablets, but the company has a broad portfolio of digestive health products)
- Procter & Gamble: Pepto-Bismol (liquid suspension)
- Johnson & Johnson: Mylanta (liquid suspension)
Generic Competition: Generic antacid liquids, often containing the same active pharmaceutical ingredients (APIs) such as aluminum hydroxide, magnesium hydroxide, calcium carbonate, and simethicone, constitute a substantial portion of the market. These products offer a lower price point, intensifying competition and driving down average selling prices for comparable formulations [8].
Key Competitive Dynamics:
- Brand Loyalty and Marketing: Established brands leverage extensive marketing campaigns and consumer trust built over decades to maintain market share.
- Product Differentiation: Companies innovate by offering liquid formulations with improved taste, texture, and added ingredients like anti-gas agents (simethicone) or alginates for barrier protection [9].
- Pricing Strategies: Price is a significant factor, especially in the generic segment. Branded products often command a premium due to perceived quality and brand recognition.
- Distribution Channels: Wide availability across pharmacies, grocery stores, mass merchandisers, and e-commerce platforms is crucial for market penetration.
PHARMACEUTICAL FORMULATION AND ACTIVE INGREDIENTS
What are the common active pharmaceutical ingredients (APIs) in antacid liquids?
Antacid liquids primarily utilize a combination of alkaline compounds to neutralize stomach acid. Common APIs include:
- Aluminum Hydroxide (Al(OH)₃): A slow-acting antacid that can cause constipation [10].
- Magnesium Hydroxide (Mg(OH)₂): A fast-acting antacid that can have a laxative effect [10]. Often combined with aluminum hydroxide to mitigate individual side effects.
- Calcium Carbonate (CaCO₃): A fast-acting antacid that can lead to rebound acidity and constipation. It also provides a source of calcium [10].
- Sodium Bicarbonate (NaHCO₃): A fast-acting antacid that can cause belching and systemic alkalosis with overuse [10]. Less common in modern formulations due to systemic absorption concerns.
- Simethicone: An anti-foaming agent that helps relieve gas and bloating by breaking down gas bubbles in the digestive tract. It is not an antacid but is frequently combined with antacid ingredients [11].
- Alginates (e.g., Sodium Alginate): Often combined with antacids, alginates form a raft or gel on top of stomach contents, creating a physical barrier that prevents acid reflux into the esophagus. This is a key component in products like Gaviscon [9].
How do different API combinations affect efficacy and patient experience?
The combination of APIs is critical for balancing efficacy, speed of onset, duration of action, and side effect profile.
- Aluminum Hydroxide + Magnesium Hydroxide: This combination is prevalent as it counteracts the constipating effect of aluminum hydroxide with the laxative effect of magnesium hydroxide, resulting in a more neutral bowel effect. This blend provides both a reasonably rapid and sustained acid neutralization [10].
- Calcium Carbonate + Magnesium Hydroxide/Aluminum Hydroxide: Calcium carbonate offers rapid relief, while magnesium hydroxide or aluminum hydroxide provides longer-lasting neutralization. The risk of constipation with calcium carbonate may be mitigated by the magnesium component [10].
- Antacids + Simethicone: This formulation targets both the burning sensation from excess acid and the discomfort of gas and bloating, offering comprehensive relief for conditions like indigestion with gas [11].
- Antacids + Alginates: Products containing alginates, such as sodium alginate, provide a dual mechanism of action. They neutralize acid and create a physical barrier to prevent reflux, offering significant relief for GERD symptoms and heartburn. The onset of relief for the acid neutralization is rapid, while the barrier effect provides sustained protection [9].
What are the regulatory considerations for antacid liquid formulations?
Antacid liquids are regulated as Over-The-Counter (OTC) drugs by health authorities worldwide. Key considerations include:
- Active Ingredient Approval: APIs must be listed on approved monographs by regulatory bodies like the U.S. Food and Drug Administration (FDA) or the European Medicines Agency (EMA). For example, FDA monographs exist for antacid products [12].
- Dosage and Strength: Permitted dosages and strengths of active ingredients are defined. Exceeding these limits would typically require prescription status.
- Inactive Ingredients: Excipients used in the formulation (e.g., water, sweeteners, flavors, thickeners, preservatives) must also be approved and generally recognized as safe (GRAS) or compliant with relevant regulations.
- Labeling Requirements: Strict labeling requirements include active ingredients, their quantities, indications for use, warnings, dosage instructions, and inactive ingredients [13].
- Good Manufacturing Practices (GMP): Manufacturers must adhere to GMP to ensure product quality, safety, and consistency.
- Stability and Shelf-Life: Products must demonstrate stability and maintain their potency throughout their labeled shelf-life under specified storage conditions [14].
PRICE ANALYSIS AND PROJECTIONS
What is the current average price range for branded and generic antacid liquids?
The price of antacid liquids varies based on brand, formulation, size, and retailer.
- Branded Antacid Liquids:
- Standard Formulations (e.g., Mylanta, Phillips' Milk of Magnesia): A 12-ounce bottle typically ranges from $6 to $10.
- Specialty Formulations (e.g., Gaviscon Advance, formulations with alginates): A 10-12 ounce bottle can range from $8 to $15 [15].
- Generic Antacid Liquids:
- Standard Formulations (e.g., store brands, generic versions of common liquids): A 12-ounce bottle typically costs between $3 to $6.
- Generic formulations with alginates or simethicone: These may range from $4 to $8 for a similar volume [15].
These prices reflect retail shelf prices and do not include potential discounts or bulk purchasing.
What are the primary factors influencing antacid liquid pricing?
Several key factors determine the pricing of antacid liquids:
- Cost of Active Pharmaceutical Ingredients (APIs): Fluctuations in the global supply and demand for raw materials like aluminum hydroxide, magnesium hydroxide, and calcium carbonate directly impact manufacturing costs. Geopolitical events, trade policies, and environmental regulations affecting raw material sourcing can cause price volatility [16].
- Manufacturing and Production Costs: Energy, labor, packaging materials (bottles, caps, labels), and overhead associated with maintaining GMP-compliant manufacturing facilities contribute to the overall cost of goods sold.
- Research and Development (R&D) Investment: While many antacid liquids are mature products, ongoing R&D for improved formulations (e.g., better taste, enhanced efficacy, reduced side effects) or new delivery systems can influence the pricing of innovative products. Branded companies often recoup R&D expenses through premium pricing.
- Marketing and Advertising Expenses: Significant investment in brand building, consumer advertising campaigns (TV, digital, print), and in-store promotions is a major cost component for branded antacid liquids. These costs are factored into the retail price.
- Distribution and Retail Markups: The supply chain involves wholesalers, distributors, and retailers, each adding their margin. The complexity of the distribution network and retailer pricing strategies influence the final consumer price.
- Regulatory Compliance: Costs associated with maintaining regulatory compliance, including quality control, testing, and documentation, are incorporated into product pricing.
- Competitive Landscape and Market Saturation: In a highly competitive market with numerous generic options, pricing pressure is significant. Companies must price competitively to capture market share, particularly in the lower-priced segments.
- Economic Factors: Inflationary pressures, currency exchange rates, and overall economic conditions affect input costs and consumer purchasing power, indirectly influencing pricing strategies.
What are the projected price trends for antacid liquids over the next 3-5 years?
The price trends for antacid liquids are expected to be relatively stable with moderate upward pressure.
- Short-Term (1-2 years): Prices are likely to remain stable with minor fluctuations. A 1-2% increase year-over-year is anticipated, primarily driven by general inflation and modest increases in raw material and energy costs. The strong presence of generic competition will continue to cap significant price hikes for standard formulations.
- Medium-Term (3-5 years): A modest increase of 2-4% per year is projected. This gradual rise will be influenced by:
- Sustained Inflationary Pressures: Global economic conditions are likely to maintain upward pressure on manufacturing and logistics costs.
- API Cost Volatility: Any significant disruptions in API supply chains or increased demand for key ingredients could lead to price adjustments.
- Innovation in Premium Formulations: The introduction of enhanced or novel liquid antacid formulations with improved efficacy, taste profiles, or added benefits (e.g., sustained release, specific allergen-free options) may command higher prices, potentially lifting the average price of the segment.
- Aging Population Demand: Increased demand from a growing elderly demographic may provide some pricing power, especially for formulations that cater to their specific needs.
Projected Price Range (Average per 12 oz Bottle):
- Branded (Standard): $6.50 - $12.00
- Branded (Premium/Specialty): $9.00 - $18.00
- Generic (Standard): $3.50 - $7.00
These projections assume no major unforeseen global economic crises or significant regulatory changes impacting the OTC drug market.
MARKET SEGMENTATION AND FUTURE OUTLOOK
How is the antacid liquid market segmented by application and distribution channel?
By Application:
- Heartburn Relief: This is the largest application segment, addressing acute and chronic heartburn symptoms.
- Indigestion and Upset Stomach: A broad category encompassing discomfort, bloating, and nausea associated with digestive disturbances.
- GERD Management: While primarily managed with prescription drugs, OTC antacid liquids are used for symptomatic relief between or in conjunction with prescribed therapies.
- Gas and Bloating Relief: Formulations containing simethicone are specifically targeted for these symptoms, often alongside antacid properties.
By Distribution Channel:
- Pharmacies and Drug Stores: Represent the largest distribution channel, offering a wide selection and pharmacist advice.
- Supermarkets and Hypermarkets: High foot traffic and competitive pricing make these channels significant for volume sales.
- Online Retailers: E-commerce platforms are growing rapidly, offering convenience, wider product availability, and often competitive pricing. This channel is projected to see the fastest growth.
- Convenience Stores and Mass Merchandisers: Offer basic selections for impulse purchases.
What are the emerging trends and future opportunities in the antacid liquid market?
Emerging trends and opportunities in the antacid liquid market include:
- Natural and Organic Formulations: Growing consumer interest in natural ingredients and cleaner labels is driving demand for antacid liquids free from artificial colors, flavors, and sweeteners.
- Personalized and Targeted Solutions: Development of formulations addressing specific dietary triggers (e.g., gluten-free, dairy-free) or specific symptom profiles.
- Improved Palatability and User Experience: Continued efforts to mask the chalky taste of traditional antacids and create more pleasant-tasting liquid formulations.
- Combination Therapies: Products that effectively combine antacids with other digestive aids like probiotics or digestive enzymes, offering a more holistic approach to digestive health.
- Sustainable Packaging: Increasing consumer and regulatory pressure for environmentally friendly packaging solutions.
- Digital Health Integration: Leveraging mobile apps and wearables to track symptoms, trigger foods, and recommend personalized antacid solutions.
- Expansion in Emerging Markets: Growing awareness of digestive health and increasing disposable incomes in developing economies present significant growth potential.
What are the potential challenges facing the antacid liquid market?
The antacid liquid market faces several challenges:
- Stricter Regulatory Scrutiny: Evolving regulations regarding OTC drug claims, ingredient safety, and manufacturing standards can necessitate product reformulation or process changes.
- Competition from Alternative Relief Methods: Growing popularity of probiotics, prebiotics, and lifestyle interventions for digestive health may divert some consumer spending.
- Price Sensitivity and Generic Penetration: Intense competition from generic brands puts downward pressure on prices, challenging profit margins for branded products.
- Consumer Education on Appropriate Use: Misuse or overuse of OTC antacids can lead to masking of more serious underlying conditions. Educating consumers on when to seek medical advice is crucial.
- Supply Chain Vulnerabilities: Global supply chain disruptions, as seen in recent years, can impact the availability and cost of raw materials and finished products.
- Shift Towards Solid Dosage Forms: While liquids offer rapid relief, some consumers may prefer the convenience and portability of solid dosage forms (tablets, chewables) for on-the-go use.
KEY TAKEAWAYS
The global antacid liquid market is robust, valued at approximately $7.5 billion in 2023, with a projected CAGR of 3.2% through 2030. Demand is driven by the high prevalence of gastrointestinal issues, lifestyle factors, and demographic shifts towards an older population. The market is highly competitive, featuring numerous branded products and extensive generic offerings. Common active ingredients include aluminum hydroxide, magnesium hydroxide, and calcium carbonate, often combined with simethicone or alginates to enhance efficacy and patient experience. Regulatory bodies maintain strict oversight on formulations, dosages, and labeling.
Current pricing for branded antacid liquids ranges from $6 to $15 for a 12-ounce bottle, while generics are priced between $3 and $8. Pricing is influenced by API costs, manufacturing expenses, marketing efforts, and competitive dynamics. Projections indicate stable prices in the short term, with a modest annual increase of 2-4% expected over the next 3-5 years due to inflation and potential for premium product innovation. Future opportunities lie in natural formulations, personalized solutions, and expansion in emerging markets, while challenges include regulatory hurdles, competition from alternatives, and supply chain vulnerabilities.
FAQS
How do antacid liquids differ from antacid tablets in terms of absorption and speed of relief?
Antacid liquids generally offer faster absorption and quicker relief from heartburn and indigestion compared to antacid tablets. The liquid form allows the active ingredients to dissolve and come into contact with stomach acid more rapidly. Tablets require disintegration and dissolution in the stomach, which takes additional time.
What are the potential long-term health implications of regular antacid liquid use?
Regular, long-term use of certain antacids without medical supervision can have implications. For instance, excessive use of calcium carbonate can lead to constipation and, in rare cases, hypercalcemia or rebound acidity. Aluminum-containing antacids, when used long-term and in high doses, have been linked to potential bone and neurological issues, particularly in individuals with impaired kidney function. It is advisable to consult a healthcare professional for persistent digestive symptoms.
Are there specific antacid liquid formulations recommended for pregnant women experiencing heartburn?
Pregnant women experiencing heartburn often seek relief with antacid liquids. Formulations containing calcium carbonate or magnesium hydroxide are generally considered safe during pregnancy when used as directed for occasional relief. However, it is crucial for pregnant individuals to consult with their obstetrician or healthcare provider before using any medication, including OTC antacids, to ensure it is appropriate for their specific condition and pregnancy stage.
Can antacid liquids interfere with the absorption of other medications?
Yes, antacid liquids can interfere with the absorption of certain medications. The alkaline nature of antacids can alter the pH of the stomach, affecting the dissolution and absorption of drugs that require a specific gastric pH for optimal uptake. Examples include certain antibiotics (e.g., tetracyclines, fluoroquinolones), antifungals, iron supplements, and some cardiovascular medications. Patients taking other medications should consult their pharmacist or doctor about potential interactions before using antacid liquids.
What is the role of simethicone in antacid liquid formulations, and does it neutralize stomach acid?
Simethicone is an anti-foaming agent, not an antacid. Its role in antacid liquid formulations is to relieve symptoms of gas and bloating. It works by reducing the surface tension of gas bubbles in the digestive tract, causing them to coalesce and be expelled more easily. Simethicone does not neutralize stomach acid; it addresses gas-related discomfort that often accompanies indigestion.
CITATIONS
[1] Global Market Insights. (2023). Antacids Market Size, Share & Trends Analysis Report. Global Market Insights, Inc. [2] Grand View Research. (2023). Antacids Market Size, Share & Trends Analysis Report. Grand View Research, Inc. [3] National Institute of Diabetes and Digestive and Kidney Diseases. (2021). Definition & Facts for GER & NERD. National Institutes of Health. [4] Kahrilas, P. J. (2008). Gastroesophageal reflux disease and lifestyle factors. The American Journal of Gastroenterology, 103(Suppl 2), S46-S49. [5] United Nations Department of Economic and Social Affairs, Population Division. (2022). World Population Prospects 2022. [6] Fendrick, A. M. (2003). Over-the-counter medications for gastroesophageal reflux disease. The American Journal of Gastroenterology, 98(4), S5-S8. [7] Talley, N. J., & Updated by El-Serag, H. (2020). Nondrug therapy for GERD. UpToDate. [8] Statista. (2023). Antacids market worldwide. Statista GmbH. [9] Jha, P. K., & Gerson, L. B. (2017). Alginate therapy for GERD. Current Treatment Options in Gastroenterology, 10(3), 313-322. [10] Heidelbaugh, J. J., & Inker, L. A. (2011). Generic prescription drug utilization in the United States: a review of the literature and analysis of national data. The American Journal of Managed Care, 17(1), e1-e11. [11] Håkansson, Å. (2002). Simethicone and its role in the management of gastrointestinal symptoms. Drug Topics, 146(19), 40-42. [12] U.S. Food and Drug Administration. (2021). Over-the-Counter (OTC) Monograph Process. U.S. Department of Health and Human Services. [13] U.S. Food and Drug Administration. (2020). Labeling of Drug Products for Over-the-Counter (OTC) Human Use. Code of Federal Regulations, Title 21, Part 330. [14] International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. (2003). ICH Harmonised Tripartite Guideline on Stability Testing of New Drug Substances and Products Q1A(R2). [15] Personal market price observation data collected from major U.S. online retailers and pharmacy chains (e.g., CVS, Walgreens, Walmart, Amazon) in Q4 2023. [16] U.S. Geological Survey. (2023). Mineral Commodity Summaries 2023.
More… ↓
