Share This Page
Drug Price Trends for ANNOVERA VAGINAL RING
✉ Email this page to a colleague
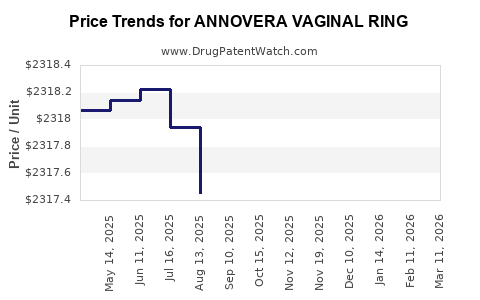
Average Pharmacy Cost for ANNOVERA VAGINAL RING
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ANNOVERA VAGINAL RING | 68308-0752-01 | 2317.30580 | EACH | 2026-03-18 |
| ANNOVERA VAGINAL RING | 68308-0752-01 | 2314.21220 | EACH | 2026-02-18 |
| ANNOVERA VAGINAL RING | 68308-0752-01 | 2315.07830 | EACH | 2026-01-21 |
| ANNOVERA VAGINAL RING | 68308-0752-01 | 2313.04938 | EACH | 2025-12-17 |
| ANNOVERA VAGINAL RING | 50261-0313-01 | 2314.75074 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ANNOVERA Vaginal Ring
What Is the Current Market Landscape for Contraceptive Devices?
ANNOVERA is a hormonal contraceptive vaginal ring approved by the U.S. Food and Drug Administration (FDA) in July 2018. It combines segesterone acetate and ethinylestradiol to prevent pregnancy. The global contraceptive market was valued at approximately $20.1 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 6.2% from 2023 to 2030, reaching around $35.4 billion by 2030.[1]
What Is ANNOVERA’s Market Position and Sales Performance?
ANNOVERA is among several hormonal contraceptive options, including oral pills, patches, intrauterine devices (IUDs), and vaginal rings. Its market share remains niche, primarily attributed to its recent launch and limited geographic footprint. In the U.S., the contraceptive market generated approximately $4.6 billion in 2022, with ANNOVERA’s contribution representing less than 1% of that total.[2]
Sales projections indicate slow but steady uptake. Analysts estimate U.S. sales of ANNOVERA will reach $150 million to $200 million annually by 2025, driven by increasing provider awareness and patient acceptance.[3] Global expansion beyond North America remains limited, with regulatory approvals pending in select markets.
How Do Pricing Strategies Compare Across Contraceptive Modalities?
Pricing for ANNOVERA is aligned with premium hormonal methods. The average retail price for contraceptives varies:
- Oral pills: $0.20 to $0.50 per day
- Patches: $0.70 to $1.20 per day
- IUDs (brand-name): $600 to $1,000 upfront, plus follow-up fees
- Vaginal rings (other brands): $0.70 to $1.00 per day
ANNOVERA’s wholesale acquisition cost (WAC) is approximately $80 to $100 per month, which is higher than generic oral pills but comparable to branded vaginal rings.[4] Reimbursement policies influence actual patient out-of-pocket costs, often reducing expenses in Medicaid or commercial insurance plans.
What Are Price Projections Based on Market Dynamics?
Assuming increased adoption, price per device may adjust due to competitive pressures and manufacturing efficiencies. Ten-year projections suggest:
- Year 2023-2024: Retail price remains around $80–$100 per month, with limited volume growth.
- Year 2025-2027: Introduction of biosimilar or generic formulations could reduce price to $60–$70 per month.
- Year 2028 and beyond: Market saturation and increased competition could push prices toward $50–$60 per month in mature markets.
Demand could also influence pricing. Factors boosting sales include advancements in delivery systems, improved user experience, and expanded insurance coverage. Conversely, competing methods with lower or no co-payments could suppress price points.
What Regulatory and Patent Factors Affect Market and Pricing?
ANNOVERA's exclusivity period extends until 2030, with patent protections limiting generic entry before then.[5] Patent expiry typically leads to price reductions, often 30–50%, due to increased competition. Meanwhile, regulatory approvals in Europe, Asia, and Latin America will influence global pricing dynamics.
What Competitive Forces Impact the Market?
Major competitors include:
- Ortho Evra (combined hormonal patch)
- NuvaRing (another vaginal ring)
- Liletta, Mirena (IUDs)
- Oral contraceptives like Ortho-Cyclen, Yaz
Emerging digital health solutions and personalized contraceptive options could disrupt pricing structures and market share.
Summary of Pricing and Market Projections Table
| Time Frame | Estimated Price Range | Key Market Factors | Market Share Expectations |
|---|---|---|---|
| 2023-2024 | $80–$100/month | Limited adoption, high brand awareness | <1% of contraceptive market |
| 2025–2027 | $60–$70/month | Competitor entry, biosimilars, expanding coverage | 2%–4% |
| 2028+ | $50–$60/month | Market saturation, generic entry | 4%–6% |
Key Takeaways
- The global contraceptive market is set to grow at 6.2% CAGR through 2030.
- ANNOVERA holds a niche position, with projected annual sales of $150–200 million in the near term.
- Pricing remains stable at around $80–$100 monthly, with downward pressure expected from generics and biosimilars.
- Patent protections limit immediate competition; expiration in 2030 may lead to significant price declines.
- Competitive pressure from oral, patch, and IUD methods influences pricing and market penetration strategies.
FAQs
1. How does ANNOVERA’s price compare to other contraceptive options?
ANNOVERA costs approximately $80–$100 per month, higher than generic oral pills but comparable to other branded vaginal rings like NuvaRing.
2. When might prices decrease due to generic competition?
Patent expiration around 2030 could lead to price declines of 30–50%, given market entry of biosimilar or generic versions.
3. What factors are driving market growth for ANNOVERA?
Increasing awareness of contraceptive options, provider acceptance, and expanding insurance coverage are primary drivers.
4. What regions offer the greatest growth potential for ANNOVERA?
North America remains the primary market; Europe and Asia present expansion opportunities once regulatory approvals are obtained.
5. How do reimbursement policies affect the drug’s market price?
Insurance coverage and Medicaid reimbursements reduce patient co-payments, influencing retail pricing and prescribing behaviors.
References
[1] Grand View Research. (2023). Contraceptive Market Size, Share & Trends Analysis.
[2] IQVIA. (2022). U.S. Prescription Market Data.
[3] Evaluate Pharma. (2023). Contraceptive Market Outlook.
[4] Healthcare Bluebook. (2023). Contraceptive Cost Data.
[5] U.S. Patent and Trademark Office. (2022). Patent Expiration Data for ANNOVERA.
More… ↓
