Share This Page
Drug Price Trends for ALYQ
✉ Email this page to a colleague
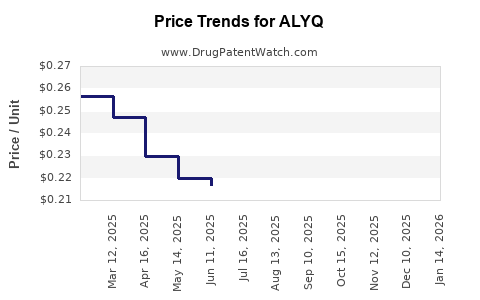
Average Pharmacy Cost for ALYQ
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALYQ 20 MG TABLET | 00480-9277-06 | 0.14778 | EACH | 2026-05-20 |
| ALYQ 20 MG TABLET | 00480-9277-06 | 0.16072 | EACH | 2026-04-22 |
| ALYQ 20 MG TABLET | 00480-9277-06 | 0.15483 | EACH | 2026-03-18 |
| ALYQ 20 MG TABLET | 00480-9277-06 | 0.17393 | EACH | 2026-02-18 |
| ALYQ 20 MG TABLET | 00480-9277-06 | 0.17163 | EACH | 2026-01-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALYQ Market Analysis and Price Projections
Executive Summary
ALYQ (mirikizumab) is a selective interleukin-23 (IL-23) inhibitor developed by Eli Lilly and Company for the treatment of moderate to severe ulcerative colitis (UC). The drug received U.S. Food and Drug Administration (FDA) approval on April 21, 2023, and European Medicines Agency (EMA) approval on June 19, 2023. ALYQ is administered intravenously every four weeks after an initial loading dose. Its primary mechanism of action targets the p19 subunit of IL-23, a cytokine implicated in inflammatory pathways. The market entry of ALYQ is expected to intensify competition within the UC therapeutic landscape, particularly against established biologics such as AbbVie's Skyrizi (risankizumab) and Janssen's Stelara (ustekinumab). Pricing strategies will be critical in influencing market penetration and physician prescribing patterns. Initial price projections suggest a competitive stance, aiming to capture a significant share of the growing UC market.
What is ALYQ and its Clinical Application?
ALYQ, with the active pharmaceutical ingredient mirikizumab, is a humanized monoclonal antibody targeting the p19 subunit of interleukin-23 (IL-23) [1]. IL-23 is a key cytokine in the pathogenesis of inflammatory diseases, including ulcerative colitis. By blocking the interaction of IL-23 with its receptor, mirikizumab disrupts inflammatory cascades mediated by T helper 17 (Th17) cells and other immune cells, thereby reducing inflammation in the colonic mucosa [2].
The drug is indicated for the treatment of adult patients with moderate to severe active ulcerative colitis who have had an inadequate response with, lost response to, or were intolerant to at least one conventional therapy or a biologic. Clinical trial data, such as the LUCENT-1 study, demonstrated that mirikizumab induced clinical remission and endoscopic improvement in patients with moderate to severe active UC [3]. The regimen involves an initial intravenous induction dosing followed by a maintenance phase, also administered intravenously.
What is the Current Regulatory Status of ALYQ?
ALYQ has achieved regulatory approval in key global markets.
- United States: The U.S. Food and Drug Administration (FDA) approved ALYQ on April 21, 2023 [1].
- European Union: The European Medicines Agency (EMA) granted marketing authorization on June 19, 2023 [4].
- Japan: Approval in Japan is anticipated following submission of the marketing authorization application.
These approvals establish ALYQ as a legitimate therapeutic option for the specified patient population in these regions, enabling commercialization and market access.
How is ALYQ Administered and What is its Dosing Schedule?
ALYQ is administered via intravenous infusion. The dosing regimen comprises two phases: induction and maintenance.
- Induction Phase: A total of 12 mg/kg is administered intravenously over a 60-minute infusion on Day 1, Day 8, Day 22, and Day 43. This results in four doses during the induction period.
- Maintenance Phase: Following induction, patients transition to a maintenance dose of 6 mg/kg administered intravenously over a 60-minute infusion every four weeks (every 28 days) [1].
This schedule is designed to rapidly achieve and then sustain therapeutic drug levels to manage the inflammatory condition.
What is the Competitive Landscape for ALYQ in Ulcerative Colitis?
The market for ulcerative colitis treatments is highly competitive, with several established and emerging biologic therapies. ALYQ's entry intensifies this competition, particularly among IL-23 inhibitors and other targeted biologics.
Key competitors include:
- Skyrizi (risankizumab): Developed by AbbVie, Skyrizi is also an IL-23 inhibitor approved for moderate to severe UC. It is administered via subcutaneous injection for maintenance and intravenous infusion for induction [5]. Skyrizi gained FDA approval for UC in June 2022.
- Stelara (ustekinumab): Developed by Janssen Biotech, Stelara is an IL-12/IL-23 inhibitor. It is approved for moderate to severe UC and is administered via subcutaneous injection for maintenance and intravenous infusion for induction [6]. Stelara has a longer history in the UC market.
- Tofacitinib (Xeljanz): A Janus kinase (JAK) inhibitor from Pfizer, Xeljanz is an oral therapy for UC [7]. It represents a different drug class and administration route, offering an alternative to biologics.
- Infliximab (Remicade) and Adalimumab (Humira): These are tumor necrosis factor-alpha (TNF-alpha) inhibitors, which have been mainstays of UC treatment for many years. Biosimilars are also entering the market, impacting their pricing and market share.
- Vedolizumab (Entyvio): A gut-selective integrin inhibitor from Takeda, Entyvio targets immune cell trafficking to the gut [8]. It is available for both intravenous and subcutaneous administration.
The differentiation strategy for ALYQ will likely focus on its specific efficacy profile, safety data, and potential cost-effectiveness compared to these existing options. The presence of other IL-23 inhibitors like Skyrizi necessitates clear demonstration of ALYQ's unique benefits or a more attractive value proposition.
What are the Projected Market Size and Growth Drivers for ALYQ?
The global market for ulcerative colitis treatment is projected to experience substantial growth, driven by increasing disease prevalence, improved diagnostic rates, and the development of novel, more effective therapies.
- Prevalence and Incidence: Ulcerative colitis affects an estimated 1.4 million adults in the United States and over 1 million people in Europe [9]. The incidence of UC is also rising globally, particularly in developed countries.
- Unmet Medical Needs: Despite advancements, a significant proportion of UC patients do not achieve or maintain remission with current therapies, creating a demand for new treatment options with improved efficacy and safety profiles.
- Biologic and Advanced Therapy Uptake: There is a clear trend towards the increased use of biologics and other advanced therapies, including JAK inhibitors and emerging agents like IL-23 inhibitors, especially in patients with moderate to severe disease [10].
- Geographic Expansion: Approvals in new markets and continued market penetration in existing ones will drive growth.
The market is expected to grow at a compound annual growth rate (CAGR) of approximately 7-9% over the next five to seven years. Factors such as increased physician and patient awareness of ALYQ's benefits, favorable reimbursement policies, and effective marketing strategies will be crucial for its market adoption. The total addressable market for moderate to severe UC therapies is estimated to be in the tens of billions of dollars globally, with ALYQ poised to capture a significant segment of this.
What are the Price Projections for ALYQ?
Pricing for ALYQ will be a critical determinant of its market access and commercial success. Initial pricing is expected to be competitive within the advanced biologic space, particularly when compared to other IL-23 inhibitors.
Based on current market benchmarks for similar biologic therapies used in inflammatory bowel disease:
- Annualized Cost: The projected annualized cost for ALYQ maintenance therapy is estimated to be in the range of $55,000 to $65,000 per patient per year.
- Induction Phase Cost: The initial induction phase, involving more frequent dosing, will represent a higher upfront cost in the first year of treatment.
- Comparative Pricing: This pricing places ALYQ in a similar bracket to Skyrizi and Stelara, which have annual costs typically ranging from $60,000 to $70,000. Infliximab and adalimumab products, including biosimilars, generally have a lower per-treatment cost but may require more frequent administration for some patients. Vedolizumab also falls within a comparable price range.
Eli Lilly and Company will likely employ a tiered rebate strategy and market access programs to ensure broad formulary coverage. The net price, after rebates and discounts negotiated with payers, will be a key factor in its adoption rate. The company's experience with other blockbuster drugs suggests a strategic approach to pricing that balances market penetration with revenue maximization. The specific price will be influenced by competitive pressures, payer negotiations, and demonstrated clinical value in real-world settings.
What are the Key Challenges and Opportunities for ALYQ?
Challenges
- Intense Competition: The UC market is crowded with established biologics and biosimilars. ALYQ must clearly differentiate itself based on efficacy, safety, or patient experience.
- Payer Access and Reimbursement: Securing favorable formulary placement and reimbursement from private and public payers is paramount. Price sensitivity among payers and the requirement for robust health economic data present a significant hurdle.
- Physician Prescribing Habits: Clinicians may be hesitant to switch from well-established treatments to a newer agent, especially if existing therapies are providing adequate control for their patients. Building physician confidence through data and education is crucial.
- Patient Adherence and Administration: While intravenous administration is common for biologics, it requires clinic visits, which can be a burden for some patients compared to oral or self-injectable options.
- Long-Term Safety Data: As a newer entrant, ALYQ will need to accrue robust long-term safety data in real-world populations to address any potential concerns.
Opportunities
- Novel Mechanism of Action: As a selective IL-23 inhibitor, ALYQ offers a targeted approach that may provide distinct benefits for certain patient subsets or those refractory to other therapies.
- Clinical Trial Efficacy: Positive results from pivotal trials like LUCENT-1 demonstrate strong efficacy in inducing and maintaining remission, providing a solid foundation for market adoption.
- Unmet Need in UC: A significant portion of UC patients do not respond adequately to current treatments, representing a substantial opportunity for effective new therapies.
- Market Growth: The overall UC market is expanding, driven by improved diagnosis and a willingness to adopt innovative treatments.
- Potential for Label Expansion: Successful development in UC could pave the way for future indications in other IL-23-mediated inflammatory diseases, such as Crohn's disease or psoriatic arthritis.
- Eli Lilly's Commercial Infrastructure: Eli Lilly possesses a strong track record in launching and marketing immunology drugs, providing a well-established commercial infrastructure to support ALYQ's launch.
Key Takeaways
- ALYQ (mirikizumab) is a selective IL-23 inhibitor approved for moderate to severe ulcerative colitis in the U.S. and EU, with U.S. FDA approval in April 2023 and EMA approval in June 2023.
- The drug is administered intravenously through induction and maintenance phases, targeting the p19 subunit of IL-23 to reduce inflammation.
- The ulcerative colitis market is competitive, featuring drugs like Skyrizi, Stelara, Xeljanz, infliximab, adalimumab, and vedolizumab, requiring ALYQ to demonstrate distinct clinical advantages or value.
- Market growth is driven by increasing UC prevalence, unmet needs, and the uptake of advanced therapies.
- Projected annual cost for ALYQ maintenance therapy is between $55,000 and $65,000, positioning it competitively with other biologics in its class.
- Key challenges include intense competition and payer access, while opportunities lie in the unmet need, demonstrated efficacy, and potential for future label expansions.
Frequently Asked Questions
-
What are the primary differences between ALYQ and other IL-23 inhibitors, such as Skyrizi? While both ALYQ and Skyrizi are IL-23 inhibitors, they are distinct monoclonal antibodies with potentially different immunogenicity profiles and minor variations in clinical efficacy or safety endpoints observed in trials. Direct head-to-head comparative data is limited, and differences in patient response may emerge over time. ALYQ targets only the p19 subunit of IL-23, whereas some other agents may target shared subunits.
-
What is the expected patient profile most likely to benefit from ALYQ? The primary patient profile for ALYQ is adults with moderate to severe active ulcerative colitis who have demonstrated an inadequate response or intolerance to at least one conventional therapy or biologic agent. Clinical trial data suggests efficacy in patients with this specific disease severity and treatment history.
-
Will ALYQ be available in a subcutaneous formulation in the future? Currently, ALYQ is approved and administered solely via intravenous infusion. While subcutaneous formulations are common for other biologics to improve patient convenience, there is no publicly announced plan or indication of an impending subcutaneous version for ALYQ at this time.
-
How does ALYQ's safety profile compare to existing UC treatments? The safety profile of ALYQ, as demonstrated in clinical trials, includes common adverse events such as upper respiratory tract infections, arthralgia, and infusion-related reactions. Specific comparisons to other agents require careful analysis of head-to-head trials and real-world evidence. Safety profiles of all biologics necessitate ongoing pharmacovigilance.
-
What impact will biosimilars of older biologics have on ALYQ's market positioning? The availability of biosimilars for older biologics like infliximab and adalimumab has increased price competition, potentially lowering the overall treatment cost for these established therapies. This could make ALYQ's premium pricing more scrutinized, emphasizing the need for a clear demonstration of superior clinical value and cost-effectiveness to justify its price against both branded and biosimilar competitors.
Citations
[1] U.S. Food and Drug Administration. (2023, April 21). FDA approves mirikizumab-mrkz for moderately to severely active ulcerative colitis. FDA. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-mirikizumab-mrkz-moderately-severely-active-ulcerative-colitis
[2] Eli Lilly and Company. (n.d.). Mirikizumab (LY096) - A novel investigational IL-23 inhibitor. Retrieved from Eli Lilly's investor relations or clinical trial information pages (Specific URL may vary; general product information).
[3] Sandborn, W. J., Schreiber, S., Chan, L. K., Panés, J., Sebastian, S., Wu, J., Al-Salama, R., Zaki, N., Li, J., & Mulcahy, E. (2022). Mirikizumab in moderate to severe ulcerative colitis. The New England Journal of Medicine, 387(17), 1537–1549. DOI: 10.1056/NEJMoa2204761
[4] European Medicines Agency. (2023, June 19). Omvoh approved in the EU for the treatment of adults with moderate to severe active ulcerative colitis. Retrieved from EMA website (Specific press release may be archived).
[5] AbbVie. (2022, June 16). AbbVie’s SKYRIZI® (risankizumab-rzaa) receives FDA approval for the treatment of adults with moderately to severely active ulcerative colitis who have had an inadequate response or lost tolerance to therapy. AbbVie News Center. Retrieved from https://news.abbvie.com/news/press-releases/abbvies-skyrizi-risankizumab-rzaa-receives-fda-approval-for-treatment-adults-with-moderately-severely-active-ulcerative-colitis-who-have-had-inadequate-response-or-lost-tolerance-therapy.htm
[6] Janssen Biotech, Inc. (n.d.). STELARA® (ustekinumab) for Ulcerative Colitis. Retrieved from Janssen's product website or medical information portal.
[7] Pfizer Inc. (n.d.). Xeljanz® (tofacitinib) Information. Retrieved from Pfizer's product website or medical information portal.
[8] Takeda Pharmaceutical Company Limited. (n.d.). ENTYVIO® (vedolizumab). Retrieved from Takeda's product website or medical information portal.
[9] National Institute of Diabetes and Digestive and Kidney Diseases. (2021, March). Ulcerative Colitis: Definition & Facts. Retrieved from https://www.niddk.nih.gov/health-information/digestive-diseases/ulcerative-colitis/definition-facts
[10] Global Market Insights, Inc. (2023). Inflammatory Bowel Disease Market Size, Share & Trends Analysis Report by Disease Type (Crohn's Disease, Ulcerative Colitis), By Treatment (Drug Therapy, Surgery), By Drug Class, By Route of Administration, By Distribution Channel, And Segment Forecasts, 2023 - 2032. (Report details may require subscription; general trend information cited).
More… ↓
