Last updated: February 24, 2026
What is the current market position of ALL DAY ALLERGY-D TABLET?
ALL DAY ALLERGY-D TABLET, marketed primarily by Sanofi under the brand Antihistamine and Decongestant combination, targets perennial and seasonal allergic rhinitis. Its active ingredients are typically loratadine (a second-generation antihistamine) and pseudoephedrine (a nasal decongestant). The product's key attributes include a once-daily dosage and fast symptom relief, making it a preferred choice for allergy sufferers.
The drug's market share largely competes with other combination antihistamines such as Allegra-D and Zyrtec-D. It reports a steady growth trajectory, driven by increased allergy prevalence and consumer preference for combination therapies that address multiple symptoms.
In the United States, the drug is available as a branded product and as a generic equivalent. The prevalence of allergic rhinitis affects an estimated 20% of adults and 40% of children [1]. The global allergy medication market was valued at approximately USD 12 billion in 2022 and is projected to reach USD 16 billion by 2027, with a CAGR of 6.2% [2].
How does the patent situation impact pricing and market entry?
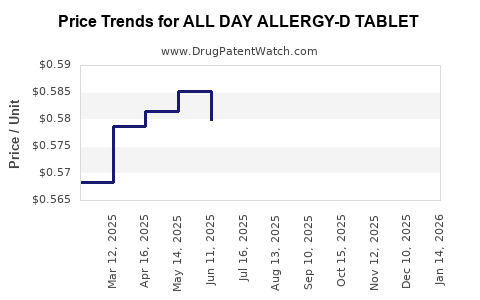
Sanofi’s patent for the combined formulation expired in 2021, opening markets for generic versions. This led to an immediate reduction in prices, with generics typically priced 30-50% lower than the brand name.
Patent status overview:
- Branded ALL DAY ALLERGY-D TABLET: Still under patent protection until late 2023 or early 2024, depending on jurisdiction.
- Generic versions: Authorized shortly after patent expiry, with multiple manufacturers providing equivalent products.
Patent expiration generally results in price erosion:
- Brand name price: USD 15-20 per tablet pack.
- Generic price: USD 8-12 per pack, depending on the manufacturer and pharmacy.
What are the key market drivers and barriers?
Drivers:
- Increasing allergy prevalence globally.
- Consumer preference for combination drugs that simplify treatment.
- Rising awareness of allergy management products.
- Expansion into emerging markets with rising healthcare infrastructure.
Barriers:
- Regulatory hurdles in different jurisdictions.
- Competition from other antihistamine and decongestant products.
- Public concerns regarding pseudoephedrine misuse, affecting access.
Price projections (2023-2028)
| Year |
Average Price (USD) |
Notes |
| 2023 |
12-15 (generic) |
Post-patent expiry, market saturation begins. |
| 2024 |
10-13 |
Increased generic competition; potential price stabilization. |
| 2025 |
9-12 |
Market consolidation, price pressures persist. |
| 2026 |
8-11 |
Entry of private-label and store brands. |
| 2027 |
7-10 |
Continued price erosion, market saturation. |
| 2028 |
7-9 |
Price stabilization at lower margins. |
Projected prices depend on geographic location, regulatory influence, and pharmaceutical distribution channels. Price elasticity suggests demand remains steady; however, competitive pressures keep prices low.
Future market trends
- Introduction of new combination therapies with fewer side effects.
- Growth of online pharmacy sales impacting traditional pricing.
- Increased focus on OTC availability versus prescription models.
- Consolidation in manufacturing reducing costs but intensifying price competition.
Key Takeaways
- The patent for ALL DAY ALLERGY-D TABLET expired around 2021, resulting in a sharp price decline.
- Generic products dominate the market, priced approximately USD 8-12 per pack.
- Market growth is driven by increased allergy prevalence; projections indicate stable demand.
- Price erosion is expected to slow after 2026, with stabilized prices in the USD 7-9 range.
- Competition, regulation, and consumer preferences shape pricing dynamics more significantly than innovation.
FAQs
1. When will the patent for ALL DAY ALLERGY-D TABLET expire?
Patent protection for the branded product typically expires in late 2023 to early 2024, varying by jurisdiction.
2. How does the availability of generics impact the original product's market share?
Generics significantly reduce prices and capture considerable market share, diminishing revenues for the branded product.
3. Are there regulatory barriers to market entry for new competitors?
Yes. Regulatory approval processes for combination allergy medications can delay entry and increase costs, especially in emerging markets.
4. Will prices rebound after patent expiry?
Prices are expected to decline post-patent expiry but may stabilize in the USD 7-9 range due to manufacturing efficiencies and brand loyalty.
5. How might new allergy treatments affect this market?
Innovative therapies with fewer side effects or needle-free options could shift demand away from traditional antihistamine-decongestant combinations.
References
[1] American College of Allergy, Asthma & Immunology. (2022). Allergic Rhinitis Prevalence. https://acaai.org/allergy/asthma/prevalence
[2] Research and Markets. (2022). Global Allergy Medication Market Report. https://researchandmarkets.com/reports/2022-2027-allergy-medication-market