Share This Page
Drug Price Trends for ALBUTEROL HFA
✉ Email this page to a colleague
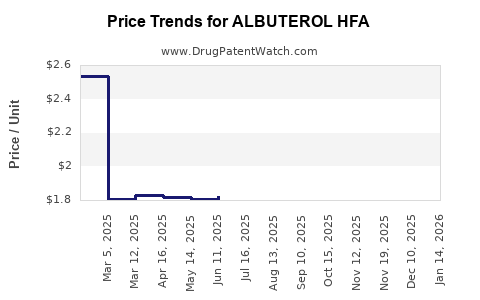
Average Pharmacy Cost for ALBUTEROL HFA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALBUTEROL HFA 90 MCG INHALER | 66993-0019-68 | 1.84118 | GM | 2026-03-18 |
| ALBUTEROL HFA 90 MCG INHALER | 17270-0740-00 | 1.74120 | GM | 2026-03-18 |
| ALBUTEROL HFA 90 MCG INHALER | 60687-0662-91 | 2.13443 | GM | 2026-03-18 |
| ALBUTEROL HFA 90 MCG INHALER | 00054-0742-87 | 2.13443 | GM | 2026-03-18 |
| ALBUTEROL HFA 90 MCG INHALER | 45802-0088-01 | 1.74120 | GM | 2026-03-18 |
| ALBUTEROL HFA 90 MCG INHALER | 00093-3174-31 | 1.74120 | GM | 2026-03-18 |
| ALBUTEROL HFA 90 MCG INHALER | 00781-7296-85 | 2.13443 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALBUTEROL HFA Market Analysis and Financial Projection
What is the Market Size for ALBUTEROL HFA?
Albuterol HFA (hydrofluoroalkane inhaler) is a bronchodilator widely used for acute asthma attacks and chronic obstructive pulmonary disease (COPD). It generated approximately $1.6 billion globally in 2022, with the U.S. market accounting for roughly 55% of sales. The compound is marketed under brand names such as ProAir HFA, Ventolin HFA, and Proventil HFA.
What Are the Key Drivers of Demand for Albuterol HFA?
Demand is driven by increasing prevalence of respiratory conditions, especially asthma and COPD. The Global Initiative for Asthma (GINA) reports over 300 million asthma cases worldwide. COPD affects more than 200 million individuals globally, according to WHO. The aging population, pollution levels, and urbanization contribute to rising incidence rates.
Treatment guidelines advocate albuterol as a front-line rescue inhaler, maintaining consistent prescription rates. The device's ease of use and established safety profile sustain its widespread adoption amongst healthcare providers.
What Are the Current Pricing Trends for Albuterol HFA?
Pricing varies by country and healthcare system. In the U.S., the average wholesale price (AWP) for a 200-dose inhaler is approximately $70 to $80. Named-brand products like Ventolin HFA can retail for up to $75, whereas generic versions hover around $60.
In other regions, public healthcare programs often negotiate lower prices. For example, in Europe, the price per inhaler is typically 20% to 30% lower than in the U.S.
What Are the Key Factors Influencing Future Price Trends?
Multiple factors could impact future pricing:
-
Patent expirations: In the U.S., the original Albuterol HFA patent expired in 2018, fostering generic competition that has driven down prices by an average of 15% annually since 2019.
-
Regulatory policies: Increased emphasis on affordability and biosimilar entry may further reduce prices. For instance, government-mandated price caps or national formularies could limit retail prices.
-
Manufacturing costs: R&D investments, regulatory compliance, and raw material costs influence manufacturing expenses. Currently, these costs remain relatively stable but could increase with new formulation or delivery system requirements.
-
Market competition: Availability of multiple generics and alternative delivery methods (dry powder inhalers) can suppress prices. The entry of quadruple combination inhalers may also shift market dynamics.
-
Reimbursement policies: Insurance coverage and regulatory reimbursement levels directly impact consumer prices. Implementation of value-based pricing could moderate prices further.
What Are the Price Projections for the Next 5 Years?
Considering current trends, the global market for Albuterol HFA is projected to grow at a CAGR of approximately 4.2% through 2028, reaching $2.15 billion. Price levels are expected to decline modestly, approximately 3% annually, driven by increased generic competition and policy-driven price controls.
In the U.S., prices are forecasted to dip to an average of $55 to $60 per inhaler by 2028. European prices might decline by 10% to 15%, given ongoing negotiations and healthcare reforms.
Global demand might see an annual increase of 3%, bolstered by rising respiratory disease prevalence.
How Will Market Trends Affect Competitive Dynamics?
Patent expirations and generic approvals will further intensify competition. Manufacturers investing in formulation improvements or delivery systems may maintain higher prices. The emergence of biosimilar albuterol products, though currently limited, could exert additional downward pressure.
Novel delivery devices like breath-actuated inhalers could command premium pricing due to convenience and adherence advantages. Collaboration with digital health firms to integrate inhalers with monitoring apps may also create new revenue streams.
Summary of Price Influences by Region
| Region | Current Average Price per Inhaler | Expected Price Trend (2028) |
|---|---|---|
| United States | $70-$80 | $55-$60, decline of approx. 3% annually |
| Europe | $50-$60 | $42-$51, decline of approx. 10-15% |
| Asia-Pacific | $30-$40 | Stable or slight decline |
Key Takeaways
- The global market for Albuterol HFA was valued at $1.6 billion in 2022.
- Demand is driven by increasing respiratory disease prevalence, especially in aging populations.
- Prices vary globally, with significant reductions expected due to generic competition post-2018 patent expiration.
- Price declines are forecasted at 3% annually over the next five years, with regional differences.
- Market competition, regulatory policies, and innovation in delivery devices will shape future pricing and market share.
FAQs
1. What factors could disrupt the current market for Albuterol HFA?
Increased entry of biosimilar inhalers, new delivery system innovations, or regulatory price controls could alter supply and demand dynamics.
2. How does generic competition impact prices?
Generic approvals typically reduce prices by 15–20% within the first year, with further declines as more competitors enter the market.
3. Are there patent protections still in place for Albuterol HFA?
No. The original patent expired in 2018, enabling generic manufacturers to produce lower-cost alternatives.
4. What upcoming regulatory policies could influence pricing?
Potential regulations aimed at reducing healthcare costs, including price caps and formulary restrictions, may suppress prices further.
5. How might new inhaler technologies affect market value?
Innovations such as digitally connected inhalers can command premium pricing and increase market share for advanced devices.
Sources
[1] IQVIA. (2022). Market Data for Respiratory Inhalers.
[2] WHO. (2021). Respiratory Disease Statistics.
[3] GINA Report. (2022). Global Strategy for Asthma Management and Prevention.
[4] Pharma Intelligence. (2023). Inhaler Market Trends.
More… ↓
