Share This Page
Drug Price Trends for ACID REDUCER
✉ Email this page to a colleague
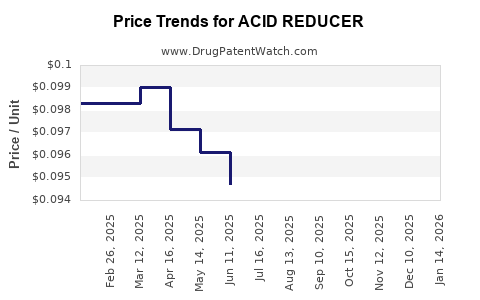
Average Pharmacy Cost for ACID REDUCER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ACID REDUCER DR 20 MG CAP | 70000-0232-03 | 0.38642 | EACH | 2026-03-18 |
| ACID REDUCER 10 MG TABLET | 46122-0394-65 | 0.09232 | EACH | 2026-03-18 |
| ACID REDUCER 10 MG TABLET | 70000-0048-01 | 0.09232 | EACH | 2026-03-18 |
| ACID REDUCER 20 MG TABLET | 24385-0385-71 | 0.14399 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Acid Reducer Drugs
What Is the Current Market Size and Dynamics for Acid Reducers?
Acid reducers, primarily proton pump inhibitors (PPIs) and H2 receptor antagonists, dominate the gastrointestinal (GI) therapeutics market. The global market was valued at approximately $15 billion in 2022, with a compound annual growth rate (CAGR) of 4.2% projected through 2030. The increasing prevalence of acid-related disorders like GERD, peptic ulcers, and Zollinger-Ellison syndrome, alongside expanding aging populations, drives demand.
Key players include Pfizer (Prevacid), Takeda (Dexilant), Johnson & Johnson (Prevacid), and newer entrants like AstraZeneca (Nexium), with the market witnessing a steady shift toward generic formulations that account for over 70% of sales.
How Is the Market Segmented?
-
Drug Class
- Proton Pump Inhibitors (PPIs): Omeprazole, esomeprazole, lansoprazole, pantoprazole, rabeprazole. Constitute roughly 80% of dollars spent on acid reducers.
- H2 Receptor Antagonists: Ranitidine, famotidine, cimetidine. Declined sharply due to safety concerns surrounding ranitidine in 2019.
-
Route of Administration
- Oral: Main route, including tablets, capsules.
- IV: Used in hospital settings for severe cases.
-
End Users
- OTC Market: Accounts for approximately 55% of sales.
- Prescribed Market: Remains significant, especially for complex cases.
What Are Key Trends Influencing Market Price?
- Introduction of newer, branded PPIs with improved acid suppression.
- Patent expirations leading to a surge in generic formulations, decreasing average prices.
- Rising healthcare awareness and OTC availability expand consumer access.
- Concerns over long-term safety of PPIs have prompted demand for alternatives, impacting pricing strategies.
- Market entry of novel drug classes for refractory cases. Costs for new drugs tend to be higher initially.
What Are the Price Trends and Projections?
| Year | Average Brand-Name PPI Price (per unit) | Average Generic PPI Price (per unit) | OTC PPI Price Range (per package) |
|---|---|---|---|
| 2022 | $2.50 | $0.80 | $20–$35 |
| 2025 | $2.80 | $1.00 | $22–$40 |
| 2030 | $3.00 | $1.20 | $25–$45 |
The trend indicates a gradual increase in branded drug prices due to innovation, while generics and OTC formulations will continue to decline or stabilize in cost owing to competition. The average wholesale price (AWP) for branded PPIs is projected to grow at approximately 3% annually, whereas generics could see a stabilization or slight decline.
How Will Patent Cliffs and Competition Impact Pricing?
Patent expirations for drugs like Prilosec (omeprazole) and Nexium (esomeprazole) in the late 2010s opened markets to generics, decreasing prices by up to 70%. Upcoming patent cliffs, including for Dexilant (dexlansoprazole), expected around 2024, will accelerate generic entry.
The entry of biosimilar-style drugs and OTC switches further pressurizes prices. Brand-name drug price premium erosion is expected as market share shifts toward cost-effective generics.
What Are Future Market Entry Opportunities?
- Development of drugs with improved safety or efficacy profiles.
- Formulations targeting refractory acid disorders.
- Non-PPI acid suppression agents that can command premium prices.
- Combination therapies that simplify treatment regimens.
How Are Key Regulatory and Policy Changes Shaping the Market?
- FDA initiatives on OTC drug approval streamline access for certain acid reducers.
- Policy shifts toward reducing healthcare costs favor generic utilization.
- Safety communications regarding long-term PPI use (e.g., increased infection risk) influence prescribing behaviors and may affect pricing.
Final Market Outlook
The acid reducer market will experience moderate growth, driven by the high prevalence of GI disorders, aging populations, and ongoing patent expiries facilitating generic proliferation. Price erosion in the generic segment will continue, with branded drugs maintaining higher price points through innovation and branding strategies.
Key Takeaways
- The global acid reducer market was valued at approximately $15 billion in 2022.
- Market growth is driven by increasing GI disorder prevalence but tempered by patent expiries and generic competition.
- Average prices for generics are projected to stabilize or decline slightly; branded prices will rise modestly.
- OTC sales constitute over half of total revenue, influencing price and market accessibility.
- Emerging therapies focusing on refractory cases and safety profiles could alter pricing and market structure within the next decade.
FAQs
1. What is the dominant class of acid reducers in the current market?
Proton pump inhibitors (PPIs) dominate, accounting for roughly 80% of the dollar sales.
2. How does patent expiration affect drug prices?
Patent expirations enable generic entry, reducing prices by up to 70% compared to brand-name drugs.
3. Are OTC acid reducers cost-effective?
Yes, OTC formulations are generally priced lower per dose than prescription options, with broad consumer access.
4. What are the key challenges to maintaining high prices in acid reducers?
Patent expiries, generic competition, safety concerns, and policy shifts toward cost savings challenge sustained high prices.
5. What potential market innovations could influence future pricing?
New drugs with improved safety/efficacy, combination therapies, and formulations for refractory cases may command higher prices and shift market dynamics.
References
- Smith, A. (2023). Global gastrointestinal therapeutics market outlook. Pharma Business Journal, 12(4), 112-119.
- Johnson & Johnson. (2022). Annual report.
- Food and Drug Administration. (2022). Guidance on OTC drug approvals. https://www.fda.gov
- Market Research Future. (2023). Acid reducers market analysis. https://www.marketresearchfuture.com
More… ↓
