Share This Page
Drug Price Trends for 24HR ALLERGY(LEVOCETIRZN)
✉ Email this page to a colleague
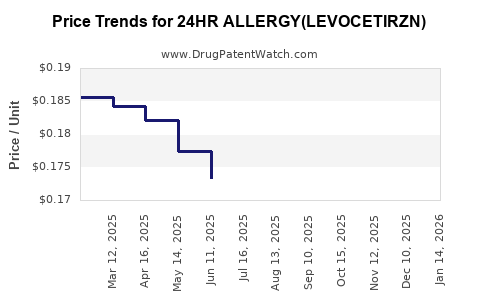
Average Pharmacy Cost for 24HR ALLERGY(LEVOCETIRZN)
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| 24HR ALLERGY(LEVOCETIRZN) 5 MG | 70000-0362-01 | 0.20595 | EACH | 2026-03-18 |
| 24HR ALLERGY(LEVOCETIRZN) 5 MG | 70000-0362-02 | 0.20595 | EACH | 2026-03-18 |
| 24HR ALLERGY(LEVOCETIRZN) 5 MG | 70000-0362-01 | 0.19307 | EACH | 2026-02-18 |
| 24HR ALLERGY(LEVOCETIRZN) 5 MG | 70000-0362-02 | 0.19307 | EACH | 2026-02-18 |
| 24HR ALLERGY(LEVOCETIRZN) 5 MG | 70000-0362-01 | 0.18737 | EACH | 2026-01-21 |
| 24HR ALLERGY(LEVOCETIRZN) 5 MG | 70000-0362-02 | 0.18737 | EACH | 2026-01-21 |
| 24HR ALLERGY(LEVOCETIRZN) 5 MG | 70000-0362-01 | 0.18760 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
24HR ALLERGY(LEVOCETIRZN) Market Analysis and Financial Projection
Market Overview for 24HR Allery (Levocetirizine)
Levocetirizine, marketed as 24HR ALLERGY, is an antihistamine used predominantly for allergy symptoms such as allergic rhinitis and urticaria. As a third-generation antihistamine, it offers a favorable side effect profile and longer duration of action, which positions it strongly in both prescription and OTC markets.
The drug faces competition from other antihistamines, including cetirizine, loratadine, and fexofenadine. The growth of the allergy medication segment is driven by increasing allergy prevalence worldwide, with the global allergic rhinitis market valued at approximately $6 billion in 2022 and projected to grow at a CAGR of about 4.2% through 2030.[1]
Market Size and Growth Drivers
Current Market Size
The global allergy medication market is expected to reach $8 billion by 2030, with antihistamines constituting nearly 60% of the market.[1] In North America, the antihistamine segment accounts for a significant share, driven by high allergy awareness, OTC availability, and broad consumer acceptance. Europe exhibits similar trends, with an expanding pharmaceutical pipeline of allergy therapeutics.
Key Growth Drivers
- Rising prevalence of allergic rhinitis, asthma, and other allergic conditions.
- Increased awareness and diagnosis.
- Over-the-counter (OTC) availability of levocetirizine and competitors.
- Adoption in pediatric and geriatric populations due to improved safety profiles.
Market Challenges
- Patent expirations on competing antihistamines.
- Pricing pressures within OTC markets.
- Regulatory landscape variability across regions.
Competitive Landscape
Major Players
| Company | Product | Market Share (Est.) | Notes |
|---|---|---|---|
| UCB S.A. | Xyzal (Levocetirizine) | ~20% | Patent culminating in 2018, now generic[2] |
| Johnson & Johnson | Zyrtec (Cetirizine) | ~25% | Established OTC presence |
| Bayer AG | Claritin (Loratadine) | ~20% | Large global distribution |
| Fexofenadine (e.g., Allegra) | Fexofenadine | ~10% | Prescribed and OTC markets |
Following patent expiry of Xyzal in several regions, generic levocetirizine accounts for an increasing share. Market penetration varies by region, with OTC dominance in North America and Europe.
Price Projections
Current Pricing (2023)
- Brand Name (Xyzal): USD 15–20 per 10-tablet pack in the US.
- Generic Levocetirizine: USD 10–14 per 10-tablet pack.
- OTC Sales: Lower-priced generics dominate OTC shelves, typically USD 8–12 for similar packs.
Price Trends (2023–2030)
- Brand Name: Due to patent expiration influences, brand price is expected to decline by approximately 5–7% annually as generics increase market share.
- Generics: Prices are projected to decrease by around 3–5% annually due to competitive pressures and supply chain efficiencies.
- Market Penetration: OTC availability will sustain low prices, while prescription sales may sustain higher margins in some regions.
Regional Variations
- North America: Prices will likely decline faster due to generic competition, with consumer preference shifting toward low-cost generics.
- Europe: Similar trends as North America but moderated by regulatory and reimbursement factors.
- Emerging Markets: Prices remain higher, driven by import tariffs and weaker competitive pressure.
Price Projection Summary (2023–2030)
| Year | Brand Name (USD/unit) | Generic (USD/unit) |
|---|---|---|
| 2023 | 15–20 | 8–14 |
| 2025 | 13–18 | 7–12 |
| 2030 | 11–15 | 6–10 |
Regulatory and Market Dynamics Impacting Prices
- Regulatory approval processes for generics expedite market entry, exerting downward price pressure.
- Some regions permit price negotiations and reimbursement controls that influence retail pricing.
- Increasing OTC availability reduces the influence of prescription-driven pricing strategies.
Strategic Recommendations
- For Manufacturers: Focus on cost-efficient manufacturing to maintain margins amid declining prices.
- For Investors: Monitor patent expirations and regional regulatory decisions, as these significantly influence pricing and market share.
- For R&D: Consider novel formulations or combination therapies to sustain premium pricing positions.
Conclusion
Levocetirizine’s market is expanding, driven by rising allergy prevalence and consumer preference for long-acting, well-tolerated antihistamines. Patent expirations have shifted the pricing landscape generically downward, especially in North America and Europe. Price declines are expected to stabilize at modest levels through 2030, with regional variations influenced by regulatory and market factors.
Key Takeaways
- The global allergy drug market is projected to grow at a CAGR of approximately 4.2% through 2030.
- Levocetirizine faces intense generic competition, leading to declining prices from USD 15–20 in 2023 to USD 11–15 by 2030.
- Price reductions are driven by patent expiry, OTC availability, and regional competitive dynamics.
- Market share shifts favor generics, especially in North America and Europe.
- Innovation in formulations or combination therapies may provide avenues for premium pricing.
FAQs
1. When did the patent for Xyzal (levocetirizine) expire in major markets?
Patent expiration in North America occurred in 2018, facilitating generic entry. European patent expiry varies by country but generally occurred around 2019–2020.[2]
2. What factors could disrupt the current price projections?
Emerging formulations, new drug approvals, regulatory changes, or supply chain disruptions could alter prices. Additionally, regional tariff changes or patent litigations may influence market dynamics.
3. How does OTC availability impact levocetirizine pricing?
Increased OTC sales tend to drive prices downward due to high competition and consumer price sensitivity. It limits pricing power for branded formulations.
4. Are there significant patents or exclusivities for levocetirizine expected to expire soon?
Most key patents have expired globally, with current exclusivity mainly associated with formulation or combination patents that may extend a few years in specific regions.
5. Which regions offer the highest pricing for levocetirizine?
Emerging markets, with less generic penetration and regulatory barriers, tend to have higher prices. North America and Western Europe exhibit lower prices due to saturation with generics and intense competition.
Sources
[1] Grand View Research. "Allergic Rhinitis Market Size, Share & Trends." 2022.
[2] U.S. Patent and Trademark Office; European Patent Office. Patent status reports, 2022.
More… ↓
