Share This Page
Drug Price Trends for ZONISAMIDE
✉ Email this page to a colleague
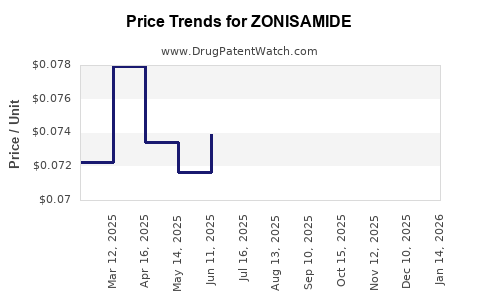
Average Pharmacy Cost for ZONISAMIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ZONISAMIDE 100 MG CAPSULE | 50268-0816-15 | 0.10762 | EACH | 2026-04-22 |
| ZONISAMIDE 100 MG CAPSULE | 50268-0816-11 | 0.10762 | EACH | 2026-04-22 |
| ZONISAMIDE 50 MG CAPSULE | 76282-0227-01 | 0.09823 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ZONISAMIDE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ZONEGRAN 100MG CAP | Amdipharm Limited | 59212-0680-10 | 100 | 1970.02 | 19.70020 | EACH | 2023-01-01 - 2026-09-28 | FSS |
| ZONEGRAN 100MG CAP | Amdipharm Limited | 59212-0680-10 | 100 | 1203.98 | 12.03980 | EACH | 2021-09-29 - 2026-09-28 | Big4 |
| ZONEGRAN 25MG CAP | Amdipharm Limited | 59212-0681-10 | 100 | 1481.25 | 14.81250 | EACH | 2022-08-01 - 2026-09-28 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Zonisamide Market Analysis and Price Projections
Executive Summary
The global market for zonisamide, an antiepileptic drug (AED), is projected to experience moderate growth driven by increasing epilepsy prevalence and its efficacy in specific patient populations. Patent expiries and the emergence of generics have led to significant price erosion for originator products, with future pricing heavily influenced by generic competition and healthcare reimbursement policies. Key market segments include partial-onset seizures and adjunctive treatment for Lennox-Gastaut syndrome.
What is Zonisamide?
Zonisamide is a sulfonamide anticonvulsant. Its mechanism of action involves blocking voltage-sensitive sodium and calcium channels, which inhibits neuronal firing. It also exhibits weak carbonic anhydrase inhibition. Zonisamide is approved for the treatment of partial-onset seizures in adults and as adjunctive therapy for patients with seizures associated with Lennox-Gastaut syndrome (LGS).
What is the Current Market Landscape for Zonisamide?
The market for zonisamide is characterized by a mature product lifecycle, with significant generic penetration. The originator product, Zonegran, developed by Dainippon Sumitomo Pharma, has faced patent expiries in major markets, opening the door for generic manufacturers.
Key Market Drivers:
- Increasing Epilepsy Prevalence: Global estimates suggest epilepsy affects approximately 50 million people worldwide, with a disproportionately higher incidence in low- and middle-income countries. This growing patient pool supports sustained demand for effective AEDs.
- Efficacy in Specific Indications: Zonisamide's established efficacy in treating partial-onset seizures and as adjunctive therapy for LGS positions it as a valuable option for certain patient segments.
- Generic Availability: The widespread availability of generic zonisamide has made the treatment more accessible and cost-effective, driving volume growth in many markets.
Key Market Restraints:
- Competition from Newer AEDs: The development of novel AEDs with potentially improved efficacy, tolerability profiles, and novel mechanisms of action poses a competitive threat.
- Side Effect Profile: Zonisamide can cause side effects such as somnolence, dizziness, anorexia, and cognitive impairment, which can limit its use in some patients and may necessitate dose adjustments or discontinuation.
- Pricing Pressures: Intense generic competition has led to significant downward pressure on pricing, impacting revenue for both branded and generic manufacturers.
How Has Zonisamide Pricing Evolved?
Pricing for zonisamide has undergone substantial transformation following patent expiries. The initial introduction of the branded product commanded premium pricing, reflecting R&D investment and market exclusivity. However, the advent of generic competition has drastically reduced prices.
Originator Pricing (Zonegran): Prior to patent expiry, Zonegran pricing varied by region but was typically in the range of USD $5-10 per milligram for oral formulations, translating to several hundred dollars per month for a typical patient regimen.
Generic Zonisamide Pricing: Following generic entry, prices have plummeted.
- United States: A 30-count bottle of 100 mg zonisamide capsules (generic) can range from USD $20 to $100, depending on the pharmacy, insurance coverage, and specific manufacturer. This represents a reduction of over 80-90% from originator pricing.
- Europe: Similar price reductions are observed across European markets, with national health services often negotiating significant discounts for generic drugs.
- Emerging Markets: In countries with less robust patent protection or developing healthcare systems, generic zonisamide pricing can be even lower, often driven by tenders and bulk purchasing.
Factors Influencing Generic Pricing:
- Number of Generic Manufacturers: A higher number of competing generic companies typically leads to more aggressive pricing.
- Manufacturing Costs: The efficiency and scale of generic manufacturing operations directly impact cost of goods and subsequent pricing.
- Reimbursement Policies: Government reimbursement rates and formulary placements by insurance providers significantly influence market access and achievable pricing.
- Excipient Costs: While the active pharmaceutical ingredient (API) is the primary cost driver, the cost of excipients and formulation can also play a minor role.
What are the Key Geographic Markets for Zonisamide?
The market for zonisamide is global, with significant demand concentrated in developed markets due to higher epilepsy diagnosis rates and healthcare expenditure.
North America (United States, Canada): This region represents a substantial market due to a large patient population and well-established healthcare infrastructure. Generic availability has led to a highly competitive pricing environment.
Europe (Germany, France, UK, Spain, Italy): European markets are characterized by diverse reimbursement systems and national healthcare policies that influence drug pricing and access. Zonisamide is widely available generically.
Asia-Pacific (Japan, China, India, South Korea): Japan, as the origin country for zonisamide, has a mature market. China and India represent significant growth potential due to large populations and increasing healthcare access, although pricing can be more sensitive. Generic manufacturing in India is a key factor in global supply.
Rest of the World (Latin America, Middle East, Africa): These regions often exhibit a higher reliance on generics due to cost considerations. Market penetration is influenced by local regulatory frameworks and physician prescribing habits.
What are the Intellectual Property (IP) Considerations?
The primary IP barrier for zonisamide, its compound patent, has expired in major markets. However, secondary patents related to specific formulations, methods of use, or manufacturing processes may still exist, though their impact on the broad generic market is diminishing.
- Compound Patent Expiry: The core patent protecting the zonisamide molecule has expired in most key jurisdictions, including the US and Europe.
- Formulation Patents: While less impactful now, initial patent filings may have covered specific crystalline forms or delivery systems. The expiration of these allows for broader generic development.
- Method of Use Patents: Patents claiming the use of zonisamide for specific indications (e.g., adjunctive therapy for LGS) can provide some extended protection, but these are often challenged or become less relevant once generic versions are approved for broader indications.
- Regulatory Exclusivity: Data exclusivity periods granted by regulatory agencies (e.g., FDA in the US, EMA in Europe) protect the originator’s clinical trial data for a specified period, preventing generic approval based solely on bioequivalence during that time. This exclusivity has largely lapsed for zonisamide.
What are the Future Price Projections for Zonisamide?
Zonisamide pricing is expected to remain under pressure from generic competition. Any significant upward price movements are unlikely without the introduction of novel delivery systems or new indications that warrant distinct patent protection.
Projected Price Trends:
- Short-Term (1-3 years): Prices will likely continue to decline or stabilize at current low generic levels. Competition among multiple generic manufacturers will keep prices competitive.
- Medium-Term (3-5 years): Modest price erosion may continue, particularly in emerging markets as generic penetration increases. In mature markets, pricing will largely be dictated by tender processes and payer negotiations.
- Long-Term (5+ years): The market will remain dominated by generics. Prices will be driven by manufacturing costs and the overall cost-containment strategies of healthcare systems. The potential introduction of new AEDs could, however, lead to a slight shift in market share, indirectly impacting zonisamide volumes.
Factors Influencing Future Pricing:
- Generic Manufacturer Consolidation: A reduction in the number of generic players could lead to slightly less aggressive pricing, but significant increases are improbable.
- API Cost Fluctuations: While typically stable for established generics, unforeseen supply chain disruptions or raw material cost increases for zonisamide API could marginally impact pricing.
- Healthcare Policy Changes: Government policies on drug pricing, reimbursement, and the promotion of generic utilization will be a dominant factor.
- Competition from New Therapies: The development and market adoption of next-generation epilepsy treatments could reduce the overall demand for older drugs like zonisamide, potentially leading to volume-driven price adjustments.
What are the Key Takeaways?
The zonisamide market is characterized by mature, genericized products. Pricing has significantly decreased due to patent expiries and intense generic competition. Growth will be moderate, driven by epilepsy prevalence and established efficacy. Future pricing will remain low, dictated by generic competition and healthcare cost-containment measures.
Frequently Asked Questions (FAQs)
-
Are there any new patent applications related to zonisamide that could impact its market exclusivity? While the primary compound patent has expired, new patent applications might emerge concerning novel formulations, delivery devices, or combination therapies. However, such applications would likely face significant hurdles given the drug's established generic status and extensive prior art.
-
What is the projected growth rate for the global zonisamide market in the next five years? The global zonisamide market is projected to grow at a compound annual growth rate (CAGR) of approximately 3-5% over the next five years. This growth is primarily volume-driven, offset by continued price erosion.
-
How do regulatory approvals for new epilepsy treatments affect the demand and pricing of zonisamide? The approval of novel epilepsy treatments can cannibalize market share from established drugs like zonisamide, particularly if the new therapies offer superior efficacy or tolerability profiles. This competitive pressure can lead to reduced demand and potentially further price reductions for zonisamide.
-
What are the primary manufacturing challenges for generic zonisamide producers? Generic zonisamide producers face challenges in optimizing manufacturing processes for cost-efficiency, ensuring consistent API sourcing from reliable suppliers, and navigating complex regulatory approval pathways in different international markets. Maintaining competitive pricing requires meticulous control over production costs.
-
What impact do healthcare system budget constraints have on zonisamide procurement and pricing? Healthcare system budget constraints often lead to increased pressure on drug pricing. In many countries, this translates into favoring generic medications, including zonisamide, through competitive bidding processes, formulary restrictions, and mandated price reductions to manage overall pharmaceutical expenditure.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Approvals and Databases. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases
[2] European Medicines Agency. (n.d.). Medicines. Retrieved from https://www.ema.europa.eu/en/medicines
[3] World Health Organization. (n.d.). Epilepsy. Retrieved from https://www.who.int/news-room/fact-sheets/detail/epilepsy
[4] PharmaCompass. (n.d.). Zonisamide Product Information and Pricing Data. (Subscription-based access).
[5] IQVIA. (n.d.). Global Pharmaceutical Market Data and Analytics. (Subscription-based access).
More… ↓
