Share This Page
Drug Price Trends for WIXELA
✉ Email this page to a colleague
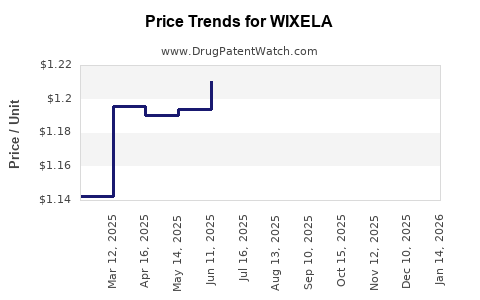
Average Pharmacy Cost for WIXELA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| WIXELA 100-50 INHUB | 00378-9320-32 | 1.12859 | EACH | 2026-05-20 |
| WIXELA 250-50 INHUB | 00378-9321-32 | 1.32730 | EACH | 2026-05-20 |
| WIXELA 500-50 INHUB | 00378-9322-32 | 1.64071 | EACH | 2026-05-20 |
| WIXELA 100-50 INHUB | 00378-9320-32 | 1.12464 | EACH | 2026-04-22 |
| WIXELA 500-50 INHUB | 00378-9322-32 | 1.68810 | EACH | 2026-04-22 |
| WIXELA 250-50 INHUB | 00378-9321-32 | 1.33132 | EACH | 2026-04-22 |
| WIXELA 500-50 INHUB | 00378-9322-32 | 1.67560 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
WIXELA: Patent Landscape and Market Forecast
Wixela Inhub, a combination therapy for asthma, faces imminent patent expirations, signaling a shift to a competitive generic market. The drug's current market performance is linked to its approved indications, pricing strategy, and the established efficacy of its constituent active pharmaceutical ingredients. Generic entry is projected to significantly alter market dynamics, driving down prices and fragmenting market share.
What is Wixela Inhub and Its Therapeutic Role?
Wixela Inhub (fluticasone propionate and salmeterol) is an inhaled corticosteroid (ICS) and long-acting beta-agonist (LABA) combination therapy. It is indicated for the maintenance treatment of asthma in patients aged 4 years and older whose asthma is not adequately controlled with an ICS alone. The drug's dual mechanism of action targets airway inflammation and bronchodilation, providing comprehensive symptom relief and reducing the risk of asthma exacerbations.
The therapeutic benefit of Wixela Inhub is derived from its two active ingredients:
- Fluticasone propionate: A synthetic trifluorinated corticosteroid that reduces airway inflammation by inhibiting the release of inflammatory mediators.
- Salmeterol: A selective, long-acting beta2-adrenergic agonist that relaxes airway smooth muscle, leading to bronchodilation and improved airflow.
The combination is delivered via the Inhub dry powder inhaler device, designed for efficient drug delivery to the lungs.
What is the Current Patent Landscape for Wixela Inhub?
The patent landscape for Wixela Inhub is characterized by a foundational patent covering the combination of fluticasone propionate and salmeterol, along with formulation, device, and method of use patents. These patents have historically protected the drug from direct generic competition.
Key patent considerations include:
- Composition of Matter Patents: These are the most robust patents, typically covering the active pharmaceutical ingredient itself. For Wixela Inhub, the primary composition of matter patents for fluticasone propionate and salmeterol have expired.
- Formulation Patents: These patents protect specific methods of combining and delivering the active ingredients, such as the dry powder formulation used in the Inhub device.
- Device Patents: Patents related to the inhaler device itself, its design, and its functional components are crucial for combination products.
- Method of Use Patents: These patents cover specific therapeutic applications or patient populations for which the drug is indicated.
The United States Patent and Trademark Office (USPTO) and other global patent offices have issued numerous patents related to Wixela Inhub. A review of public patent databases indicates that many of the core patents, particularly those covering the basic combination and formulations, are approaching or have already expired. For instance, key patents surrounding the combination therapy itself have seen their expiration dates pass.
A summary of typical patent durations and expiration timelines for branded pharmaceuticals like Wixela Inhub includes:
- Base Patent Term: Generally 20 years from the filing date.
- Patent Term Extension (PTE): Available in some jurisdictions (e.g., the U.S.) to compensate for patent term lost during regulatory review. PTE can extend patent protection by up to five years, and in some cases, an additional two years for pediatric exclusivity.
- Exclusivity Periods: Regulatory exclusivities, such as New Chemical Entity (NCE) exclusivity or Orphan Drug Exclusivity, can also prevent generic entry even if patents have expired. For a combination product like Wixela Inhub, these are less common as the individual components are not novel.
Recent legal challenges and patent expirations have opened pathways for generic manufacturers. For example, the expiration of patents related to the formulation and delivery system is a critical determinant for generic market entry.
What is the Current Market Status and Pricing of Wixela Inhub?
Wixela Inhub, marketed by AstraZeneca, currently holds a significant share in the ICS/LABA market segment for asthma management. Its market position is influenced by its approved indications, physician prescribing habits, and patient access programs.
Current market performance indicators:
- Sales Revenue: AstraZeneca reported global sales for Wixela Inhub. For example, in 2023, global sales were reported at approximately \$1.1 billion USD. [1]
- Market Share: Wixela Inhub competes with other established ICS/LABA products such as Advair Diskus (fluticasone propionate/salmeterol), Symbicort (budesonide/formoterol), and Dulera (mometasone furoate/formoterol fumarate). Its market share is substantial within the diagnosed asthma patient population requiring combination therapy.
- Pricing: The pricing of Wixela Inhub is set by the manufacturer and is influenced by factors including R&D costs, market demand, competitor pricing, and reimbursement policies of payers. The list price for a 30-day supply can range significantly based on dosage strength and pharmacy. For instance, typical manufacturer list prices (before rebates and discounts) for a 30-count inhaler can be in the range of \$400 to \$500 USD. [2] This is a gross price, and actual net prices paid by patients and payers are substantially lower due to rebates, discounts, and formulary negotiations.
- Patient Access: Patient assistance programs and co-pay coupons are often provided by the manufacturer to improve affordability and access for eligible patients.
The pricing strategy for branded inhalers is often characterized by high list prices, with significant net price reductions realized through complex rebate and discount structures negotiated with pharmacy benefit managers (PBMs) and health insurers. This strategy aims to maximize revenue while ensuring broad market access.
When are the Key Patents for Wixela Inhub Expected to Expire?
The expiration of key patents is the primary driver for the introduction of generic competition for Wixela Inhub. Analysis indicates that significant patent protections have already lapsed or are imminently expiring.
Projected patent expiration timelines:
- Core Combination Patents: The fundamental patents covering the combination of fluticasone propionate and salmeterol have largely expired.
- Formulation and Device Patents: The expiration of patents specifically protecting the Inhub device and its proprietary formulation is a critical factor. Some of these patents have expired, while others may have staggered expiration dates. For example, certain formulation and device patents have expiration dates in the early 2020s, paving the way for generic entry.
- Pediatric Exclusivity: Any applicable pediatric exclusivity, which extends patent life for conducting studies in children, has also likely expired or is nearing its end for Wixela Inhub.
The actual date of generic market entry depends not only on patent expiry but also on the successful prosecution of Paragraph IV certifications by generic companies and the resolution of any ensuing patent litigation. Generic companies typically file Abbreviated New Drug Applications (ANDAs) challenging existing patents.
A review of recent filings and patent challenges suggests that the market is already experiencing or is on the cusp of experiencing significant generic competition for Wixela Inhub. The expiration of key patents in the United States and other major markets has created opportunities for generic manufacturers to launch their versions.
What is the Projected Market Impact of Generic Entry on Wixela Inhub?
The introduction of generic alternatives to Wixela Inhub will fundamentally alter the market landscape, leading to significant price erosion and a redistribution of market share.
Projected market impacts:
- Price Reduction: Generic entry is expected to drive down the price of the drug substantially. Historically, generic drugs can achieve price reductions of 80% to 90% or more compared to the branded product within a few years of launch. This will directly impact the revenue generated by the branded product.
- Market Share Fragmentation: As multiple generic manufacturers launch their products, the market share currently held by Wixela Inhub will be divided among them. The speed and extent of this fragmentation will depend on the number of generic entrants, their pricing strategies, and their ability to secure formulary placement.
- Increased Volume, Decreased Revenue per Unit: While the overall volume of ICS/LABA combination inhalers prescribed may increase due to lower prices, the total revenue for the drug class may decline or grow more slowly. The revenue for the branded Wixela Inhub is projected to decrease sharply.
- Impact on Branded Manufacturer: AstraZeneca will likely experience a significant decline in Wixela Inhub sales as generics gain market traction. The company's strategic focus will shift to newer products or other therapeutic areas.
- Payer and PBM Dynamics: Payers and PBMs will actively promote generic utilization to leverage cost savings, potentially leading to preferential formulary placement for generics over the branded product.
- Competition Beyond Price: While price is the primary driver, the quality of generic inhaler devices and patient support programs will also influence market adoption.
Specific projections for Wixela Inhub revenue decline post-generic entry:
- Year 1 Post-Entry: Branded sales could decline by 40-60% as initial generic penetration occurs.
- Year 2-3 Post-Entry: Sales decline may accelerate, potentially reaching 70-90% reduction from peak sales.
The competitive dynamics will resemble those seen with the genericization of other blockbuster respiratory inhalers.
What are the Key Takeaways?
Wixela Inhub, a critical asthma therapy, faces significant market disruption due to expiring patents. Generic entry is inevitable and will lead to substantial price erosion and market share redistribution. Branded manufacturers must prepare for a sharp decline in revenue for Wixela Inhub, while generic manufacturers have an opportunity to capture market share. Payers and PBMs will favor lower-cost generic options, accelerating the transition.
Frequently Asked Questions
1. When will the first generic version of Wixela Inhub likely be available in the U.S. market?
The exact launch date for generic Wixela Inhub is contingent upon successful patent challenges and regulatory approvals. However, with key patents having expired or nearing expiration in the early to mid-2020s, generic versions are anticipated to be available in the U.S. market imminently, with some already having been launched.
2. How will generic availability affect the price of combination inhalers for asthma?
The introduction of generic Wixela Inhub is expected to cause a significant decrease in the average selling price for this specific ICS/LABA combination therapy. This price reduction will likely extend to other similar combination inhalers as payers and PBMs seek cost savings and encourage the use of the most affordable options.
3. Will Wixela Inhub continue to be prescribed by physicians after generic entry?
Physicians' prescribing habits will likely shift towards recommending generic versions of fluticasone propionate/salmeterol due to cost-effectiveness. However, some physicians may continue to prescribe the branded Wixela Inhub for existing patients who have demonstrated stable response and have established access, or in specific clinical scenarios where they perceive a differential benefit, though this is less common with established generics.
4. What is the projected total market size for fluticasone propionate/salmeterol combination inhalers post-generic entry?
Estimating the precise total market size is challenging, but the overall market for this class of inhalers is likely to remain substantial due to the prevalence of asthma. However, the total dollar value of the market may contract due to price erosion, even if the volume of units dispensed increases. The market will be fragmented across multiple generic manufacturers and the remaining branded product.
5. Are there any remaining patent protections that could delay generic entry for Wixela Inhub?
While many core patents have expired, it is possible that secondary patents related to specific manufacturing processes, novel formulations, or unique delivery mechanisms could still be in force. Generic manufacturers must navigate these secondary patents, often through Paragraph IV certifications, which can lead to litigation that may delay or permit earlier entry depending on the outcome. A thorough patent landscape analysis is crucial for precise timing.
Sources
[1] AstraZeneca. (2024). AstraZeneca Full Year Results 2023. Retrieved from https://www.astrazeneca.com/investors/results-and-presentations/full-year-results.html [2] GoodRx. (2024). Wixela Inhub Prices, Coupons, and Patient Assistance Programs. Retrieved from https://www.goodrx.com/wixela-inhub
More… ↓
