Share This Page
Drug Price Trends for TUSSIN
✉ Email this page to a colleague
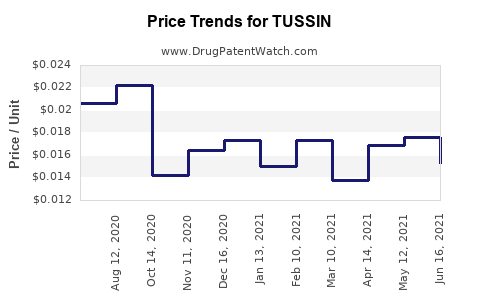
Average Pharmacy Cost for TUSSIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TUSSIN DM 400-20 MG/20 ML LIQ | 70000-0628-01 | 0.02209 | ML | 2026-03-18 |
| TUSSIN DM 200-20 MG/20 ML LIQ | 70000-0670-01 | 0.02009 | ML | 2026-03-18 |
| TUSSIN CF MAX SEVERE M-S COLD | 70000-0623-01 | 0.02378 | ML | 2026-03-18 |
| TUSSIN DM 20-200 MG/10 ML LIQ | 82568-0015-04 | 0.01109 | ML | 2026-03-18 |
| TUSSIN MUCUS-CONG 200 MG/10 ML | 82568-0012-08 | 0.01280 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
TUSSIN Market Analysis and Financial Projection
What is TUSSIN?
TUSSIN is a over-the-counter (OTC) cough and cold medicine primarily used to suppress cough, relieve chest congestion, and alleviate minor sore throat symptoms. It is marketed under the name TUSSIN in various formulations, often containing active ingredients like dextromethorphan (DXM), guaifenesin, or alcohol-based solutions.
Who manufactures TUSSIN and what is its market position?
The TUSSIN brand is owned by Prestige Consumer Healthcare Inc., which commands a significant portion of the OTC cough and cold sector in North America. It competes mainly with brands such as Robitussin (Johnson & Johnson), Mucinex (Bayer), and store brands.
What are the current market dynamics for OTC cough and cold medicines like TUSSIN?
The global cough and cold remedy market was valued at approximately USD 9.5 billion in 2022, with North America holding nearly 40% of the share. The market growth rate is projected at 4-6% annually through 2027, driven by increased consumer demand for OTC medicines, aging populations, and new product developments.
In the United States, OTC cough and cold remedies generate an estimated USD 2 billion annually. TUSSIN holds a share of around 10-15% in the North American OTC cough and cold market, according to Nielsen data. The segment faces challenges from regulatory concerns over ingredients like dextromethorphan and usage restrictions, especially among adolescents.
How is TUSSIN positioned within the market?
TUSSIN maintains its position through brand recognition, product variety (liquid, syrup, and specialized formulations), and favorable shelf placement. Its core consumer segment targets adults seeking fast relief from cough and congestion.
What factors influence TUSSIN's pricing strategy?
Pricing for OTC cough and cold medications is influenced by:
- Brand positioning: TUSSIN is positioned as a mid-priced OTC brand.
- Competition: Competitors like Robitussin and Mucinex are priced within USD 5-10 for standard bottles (4-8 oz).
- Distribution channels: Drugstores, mass merchandisers, and e-commerce outlets affect margin structures.
- Regulatory environment: Restrictions on certain ingredients and formulation changes may impact costs.
- Consumer perception: Brand loyalty and perceived efficacy influence willingness to pay premium prices.
Average retail prices for TUSSIN products range from USD 4 to USD 8 per bottle, similar to competitors.
What are the projections for TUSSIN sales and pricing over the next five years?
Sales Projections
- Growth Rate: The OTC cough and cold segment is expected to grow 4-6% annually.
- Market Share Stability: TUSSIN's market share is projected to remain stable at 10-15%, barring new product launches or regulatory challenges.
- Revenue Estimates: Assuming current market conditions, annual OTC TUSSIN sales could reach USD 200-300 million in North America by 2027.
Price Projections
- Average Price: Maintained around USD 4-8 per bottle in the short term.
- Inflation Adjustment: Prices may rise by 2-3% annually to account for inflation and packaging costs.
- Premiumization Trends: Introduction of value-added formulations could command higher retail prices, pushing average prices to USD 8-10 per pack.
Competitive Factors Influencing Prices
- Regulatory crackdowns on ingredients like DXM could lower product efficacy, pressuring prices or forcing reformulation.
- Consumer preference shift toward natural or organic remedies might lead to premium pricing for alternative formulations.
- Store-brand competitors could exert pricing pressure.
How does regulation impact TUSSIN's market and pricing?
The U.S. Food and Drug Administration (FDA) regulates active ingredients in OTC cough medicines. Dextromethorphan, a key ingredient in TUSSIN, has faced scrutiny due to misuse potential among teenagers. Recent proposals for stricter sales restrictions—such as age verification—could limit sales volume and allow for increased prices to offset revenue declines.
In 2018, the Combat Meth Act limited OTC sales of Dextromethorphan-containing products to certain retail outlets and imposed age restrictions, which affected sales. Future regulations could further restrict sales or lead to formulation bans, impacting TUSSIN's market and pricing strategies.
Key Market Drivers and Risks
Drivers:
- Aging population with increased prevalence of cough and cold symptoms.
- Consumer preference for OTC remedies for quick symptom relief.
- Innovations in delivery formats, such as natural extracts or sugar-free options.
Risks:
- Regulatory changes restricting active ingredients.
- Competition from natural or herbal remedies.
- Price sensitivity among consumers, especially during economic downturns.
- Generic proliferation, reducing brand differentiation.
Conclusions
TUSSIN's market position is stable within the North American OTC cough and cold segment. Sales are likely to grow modestly at 4-6% annually, driven by demographic factors and consumer demand. Pricing will remain relatively flat in the short term, with potential upward pressure from inflation and product differentiation strategies. Regulatory developments present significant risks, possibly constraining sales volume or necessitating reformulation, which can impact pricing and market share.
Key Takeaways
- TUSSIN is a mid-priced OTC cough and cold brand held by Prestige Consumer Healthcare.
- Market size in North America is USD 2 billion, with TUSSIN holding 10-15% share.
- Expected sales growth is 4-6% annually through 2027.
- Retail prices range from USD 4 to USD 8; inflation and innovation could push prices higher.
- Regulatory restrictions on active ingredients like dextromethorphan pose risks to sales and pricing.
FAQs
1. Will regulatory restrictions on dextromethorphan affect TUSSIN sales?
Yes. Restrictions and potential bans could limit availability, reduce sales volume, and compel reformulation, impacting revenue and pricing strategies.
2. How does TUSSIN compete against store brands?
TUSSIN leverages brand recognition, formulation variety, and shelf placement. Store brands often compete on price, but TUSSIN’s perceived efficacy and trusted branding help maintain market share.
3. Are natural or herbal alternatives impacting TUSSIN?
Emerging consumer interest in natural remedies may reduce demand for traditional OTC formulas. This trend could pressure pricing and sales unless TUSSIN expands into natural variants.
4. What future product innovations could influence prices?
Introduction of combination therapies, sugar-free options, or natural extracts could command higher retail prices, especially if combined with marketing emphasizing efficacy or health benefits.
5. How are supply chain disruptions affecting TUSSIN pricing?
Global supply chain challenges may increase costs for packaging, ingredients, and distribution, prompting moderate price increases or impacting product availability.
References
- Grand View Research. OTC Cough & Cold Remedy Market Size & Trends (2023).
- Nielsen. OTC Cough and Cold Market Share Reports (2022).
- U.S. FDA. Regulations and Guidance on Dextromethorphan (2021).
- Prestige Consumer Healthcare Inc. Annual Report (2022).
More… ↓
