Last updated: February 13, 2026
What is Trifluoperazine?
Trifluoperazine is an antipsychotic medication classified as a typical (first-generation) dopamine antagonist. It is primarily used to treat schizophrenia, anxiety, and agitation. Developed in the 1950s, it remains on the market, predominantly in generic form.
Current Market Overview
Market Presence and Usage
- Trifluoperazine is approved in several countries, including the U.S., India, and parts of Europe.
- Its market share is limited compared to newer atypical antipsychotics due to efficacy and safety profiles.
- It is often prescribed in cases where patients are intolerant to atypicals or where cost constraints exist.
Patent Status and Competition
- No active patent protections exist, as patents expired decades ago.
- Market is dominated by generic manufacturers.
- Limited branding: companies like Teva, Mylan, and Sandoz supply trifluoperazine generics.
Regulatory Environment
- Regulatory approval exists in major markets; however, marketing restrictions are minimal due to its age.
- Generic drugs face pricing pressure from biosimilar and alternative therapies.
Market Size and Revenue
- The global antipsychotics market was valued at approximately USD 14.4 billion in 2020.
- Trifluoperazine's sector is a small fraction, estimated under USD 50 million annually.
- North America accounts for roughly 50% of the antipsychotics market but uses trifluoperazine minimally.
Drivers and Barriers
Drivers:
- Low-cost alternative to atypical antipsychotics.
- Use in developing countries with limited healthcare budgets.
Barriers:
- Side effect profile (extrapyramidal symptoms, sedation).
- Availability of newer medications with better safety profiles.
- Limited market promotion due to its age and patent status.
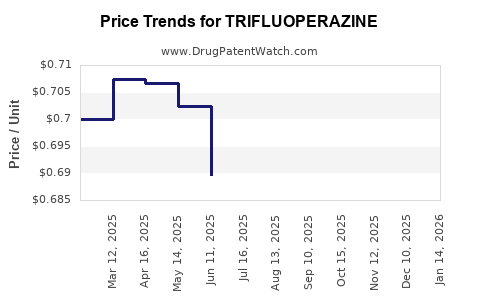
Price Trends and Projections
Current Pricing Data
- Generic trifluoperazine tablets (10 mg) retail for USD 0.05 to USD 0.10 per tablet.
- Monthly treatment costs range from USD 1.50 to USD 4.00, depending on dosage and bargaining power.
Historical Price Trends
- Slight decline over past decade, driven by increased generic competition.
- Price erosion persists, with no significant upward trend reported.
Short-term Market Projections (2023-2028)
| Year |
Estimated Market Size (USD Millions) |
Key Factors |
| 2023 |
40 |
Stable generic supply, restrained growth |
| 2024-2025 |
45–50 |
Rise in demand in low-income regions |
| 2026-2028 |
50–55 |
Market saturation, pricing stabilizes |
Long-term Outlook
No major price increases expected; limited clinical innovation for trifluoperazine likely keeps prices stagnant.
Competitive Landscape
- Generics dominate, with market fragmentation among multiple small manufacturers.
- Little to no competition from branded therapies due to expired patents.
- Shipping costs, formulation quality, and regulatory compliance influence profitability.
Regulatory and Market Risks
- Potential market shrinkage if newer therapies prove safer.
- Regulatory limitations on off-label use.
- Price wars among generics could further depress prices.
Key Market Factors for Investors and R&D
- Entry into emerging markets could expand volume.
- Formulation improvements or new delivery methods may boost select segments.
- Market decline expected without clinical repositioning or innovation.
Key Takeaways
- Trifluoperazine remains a low-cost, off-patent antipsychotic used mainly in low-resource settings.
- The global market size is small, under USD 50 million annually.
- Price per tablet has declined modestly, with little expectation of significant increase.
- It faces competition from newer therapies and safety concerns.
- Growth prospects are limited; the focus remains on cost-effective prescribing rather than innovation.
FAQs
1. Can trifluoperazine be off-patent for new indications?
It is primarily off-patent for existing indications; new uses would require regulatory approval and clinical evidence.
2. Are there any patent protections or exclusivities scheduled?
No; the compound and formulations have been off-patent for decades.
3. What are the main barriers to increasing trifluoperazine sales?
Safety profile concerns, competition from newer drugs, and limited marketing efforts.
4. Is there potential for reformulation or delivery optimization?
Yes; depot formulations or combination therapies could shift usage patterns but are not currently prevalent.
5. How does pricing compare globally?
Prices are generally consistent in low-resource countries; in high-income markets, substitution and healthcare policies limit pricing variance.
References
- MarketWatch. "Antipsychotics Market Size, Share & Trends Analysis." 2021.
- IQVIA. "Global Psychotropic Drugs Market Report," 2022.
- FDA. "Approved Drugs Database," 2023.
- WHO. "List of Essential Medicines," 2019.
- Local pharmacopoeias and pricing databases.