Share This Page
Drug Price Trends for TRAVOPROST
✉ Email this page to a colleague
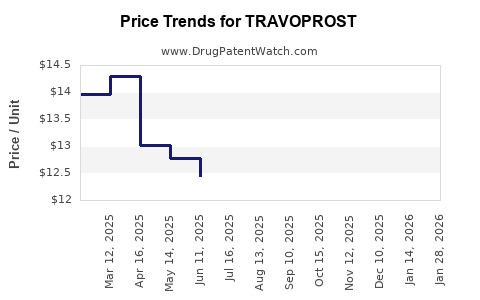
Average Pharmacy Cost for TRAVOPROST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TRAVOPROST 0.004% EYE DROP | 25021-0002-05 | 10.27347 | ML | 2026-04-22 |
| TRAVOPROST 0.004% EYE DROP | 42571-0130-21 | 10.27347 | ML | 2026-04-22 |
| TRAVOPROST 0.004% EYE DROP | 25021-0002-02 | 9.36788 | ML | 2026-04-22 |
| TRAVOPROST 0.004% EYE DROP | 42571-0130-27 | 9.36788 | ML | 2026-04-22 |
| TRAVOPROST 0.004% EYE DROP | 00378-9651-32 | 9.36788 | ML | 2026-04-22 |
| TRAVOPROST 0.004% EYE DROP | 70069-0587-01 | 10.27347 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TRAVOPROST
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TRAVOPROST 0.004% SOLN,OPH | Sandoz, Inc. | 00781-6185-75 | 5ML | 37.13 | 7.42600 | ML | 2023-08-15 - 2028-08-14 | FSS |
| TRAVATAN Z 0.004% SOLN | Sandoz, Inc. | 00078-0946-25 | 5ML | 292.39 | 58.47800 | ML | 2023-09-29 - 2028-08-14 | FSS |
| TRAVOPROST 0.004% SOLN,OPH | Sandoz, Inc. | 00781-6185-75 | 5ML | 28.37 | 5.67400 | ML | 2024-01-01 - 2028-08-14 | FSS |
| TRAVATAN Z 0.004% SOLN | Sandoz, Inc. | 00078-0946-25 | 5ML | 294.34 | 58.86800 | ML | 2024-01-01 - 2028-08-14 | FSS |
| TRAVATAN Z 0.004% SOLN | Sandoz, Inc. | 00078-0946-40 | 2.5ML | 146.26 | 58.50400 | ML | 2023-09-29 - 2028-08-14 | FSS |
| TRAVATAN Z 0.004% SOLN | Sandoz, Inc. | 00078-0946-40 | 2.5ML | 146.79 | 58.71600 | ML | 2024-01-01 - 2028-08-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Travoprost: Market Analysis and Price Projections
Travoprost is a prostaglandin analog used to treat glaucoma and ocular hypertension. It lowers intraocular pressure by increasing the outflow of aqueous humor. This analysis projects market trends and pricing for travoprost based on patent expiration, generic competition, and therapeutic landscape evolution.
What is the Current Market Landscape for Travoprost?
The global market for travoprost is characterized by a mature supply chain dominated by generic manufacturers following patent expirations. The primary indication for travoprost is the management of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension, conditions that affect millions worldwide and represent a significant segment of the ophthalmic pharmaceutical market.
The U.S. market for travoprost was initially driven by branded products like Travatan Z (Alcon). However, the expiration of key patents has led to intense generic competition. The average wholesale price (AWP) for branded travoprost formulations has historically been in the range of \$250-\$350 for a 2.5 mL bottle. Post-patent expiration, generic travoprost ophthalmic solutions are available at significantly lower price points. For instance, a 2.5 mL bottle of generic travoprost 0.004% can be sourced for approximately \$25-\$40 from various pharmaceutical wholesalers, representing a 85%-90% price reduction from branded equivalents.
The European market mirrors the U.S. trend, with early patent expiries in major markets such as Germany, France, and the UK paving the way for multiple generic entrants. Pricing in these regions, before the onset of significant generic competition, saw branded travoprost products priced between €200-€300 per 2.5 mL bottle. Post-genericization, prices for generic travoprost have settled in the range of €20-€30 per bottle.
Key market dynamics influencing current travoprost sales include:
- Generic Penetration: Over 90% of travoprost prescriptions in developed markets are now filled by generic formulations.
- Manufacturer Landscape: The market is populated by numerous generic pharmaceutical companies, including but not limited to Teva Pharmaceuticals, Viatris (formerly Mylan), and Sun Pharma, among others. These companies compete on price and market share.
- Therapeutic Class: Travoprost belongs to the prostaglandin analog class, which also includes latanoprost, bimatoprost, and tafluprost. This class is a first-line treatment option for many glaucoma patients due to its efficacy and once-daily dosing.
- Competition from Other Drug Classes: While prostaglandin analogs remain dominant, fixed-dose combinations (e.g., travoprost and timolol, travoprost and brimonidine) and other drug classes (beta-blockers, alpha-agonists, carbonic anhydrase inhibitors) continue to offer alternative treatment options. The availability and pricing of these alternatives indirectly influence travoprost market share and pricing leverage.
The overall volume of travoprost prescriptions remains substantial due to its established efficacy and cost-effectiveness as a generic medication. The demand is driven by the chronic nature of glaucoma and the need for lifelong treatment.
What are the Key Patents and Their Expiration Dates for Travoprost?
The patent landscape for travoprost has largely expired in major markets, enabling widespread generic manufacturing. The original composition of matter patent for travoprost was filed by Alcon (now part of Novartis) and granted in the United States as U.S. Patent No. 5,352,700. This patent was set to expire on February 23, 2011.
Several secondary patents and formulation patents also contributed to the protection of branded travoprost products. For example, patents related to specific formulations, such as the benzalkonium chloride-preserved version (Travatan Z), also expired. U.S. Patent No. 7,279,505, relating to a travoprost formulation, expired on November 15, 2022.
Key patent expiration milestones:
- U.S. Composition of Matter Patent (U.S. 5,352,700): Expired February 23, 2011. This was the foundational patent for travoprost.
- U.S. Formulation Patent (U.S. 7,279,505 - Travatan Z): Expired November 15, 2022. This patent protected a specific preservative system.
- European Patent Applications: Corresponding patents in Europe also followed similar expiration timelines, with the primary patent protection ceasing in the early 2010s across major European Union countries.
The expiration of these key patents has been the primary driver for the market's transition to a generic-dominated landscape. This shift has resulted in significantly lower pricing and increased accessibility for patients. The limited number of currently active patents predominantly relate to manufacturing processes or novel delivery systems, which do not present significant barriers to the existing generic market.
How Will Patent Expirations Affect Future Market Share and Pricing?
The market share and pricing of travoprost are irrevocably tied to its patent status. With the primary patents expired, the market has already transitioned to a generic environment. Future market share will be maintained by generics due to their cost-effectiveness and established therapeutic profile.
Market Share Projections:
- Continued Generic Dominance: Generic travoprost is projected to maintain over 95% market share in developed markets for the foreseeable future. This is driven by formulary inclusion, physician prescribing habits, and patient cost considerations.
- Limited Brand Revival: The resurgence of branded travoprost market share is highly improbable without significant innovation, such as a novel delivery system or a combination product with a new mechanism of action.
- Therapeutic Advancements Impact: Future market share for travoprost could be indirectly affected by the development of new drug classes or novel treatment modalities for glaucoma that offer superior efficacy, safety, or convenience, or by a shift towards surgical interventions for glaucoma management. However, given the chronic nature of the disease and the established efficacy of prostaglandin analogs, travoprost is expected to remain a staple treatment.
Price Projections:
The pricing of travoprost is expected to remain stable at current low levels, with potential for marginal declines due to ongoing competition and payer pressure.
- Price Stabilization: The average selling price (ASP) for generic travoprost in the U.S. is currently in the range of \$25-\$40 per 2.5 mL bottle. This level is expected to persist. Price erosion has largely stabilized as generic manufacturers operate on thin margins.
- Evolving Payer Influence: Pharmacy benefit managers (PBMs) and insurance providers will continue to exert downward pressure on pricing, prioritizing the lowest-cost generic options. Rebate strategies by manufacturers may offer slight price fluctuations, but the base wholesale acquisition cost (WAC) will remain depressed.
- Global Price Variations: Prices will continue to vary by region based on local market dynamics, regulatory environments, and national healthcare policies. However, the trend of significant price reductions post-patent expiration will be consistent globally. For example, in emerging markets where branded products may still hold a larger share due to access issues, generic travoprost will drive down prices as it gains traction.
- Potential for Minimal Increases: While unlikely, minor price increases might occur if there are significant disruptions in the supply chain or if the number of active generic manufacturers decreases substantially. However, the low barrier to entry for generic ophthalmic solutions makes this scenario improbable.
The primary competition for travoprost comes from other prostaglandin analogs (latanoprost, bimatoprost, tafluprost) and fixed-dose combinations. The pricing of these alternatives will also play a role. Latanoprost, being one of the earliest generic prostaglandin analogs, generally exhibits similar or slightly lower pricing than generic travoprost.
What is the Competitive Landscape for Travoprost?
The competitive landscape for travoprost is characterized by a high degree of generic competition and a diverse set of therapeutic alternatives for glaucoma and ocular hypertension management.
Direct Generic Competitors:
The market for travoprost is saturated with multiple generic manufacturers. Key players include:
- Teva Pharmaceuticals: A major global generic pharmaceutical company with a significant presence in ophthalmic solutions.
- Viatris (formerly Mylan): Another large generic manufacturer with a broad portfolio, including prostaglandin analogs.
- Sun Pharma: A significant Indian generic drug manufacturer with global operations and a strong ophthalmic division.
- Apotex: A Canadian-based global pharmaceutical company known for its generic drug offerings.
- Endo International: Though facing its own challenges, Endo has historically been a player in the generic ophthalmic space.
- Numerous Smaller Manufacturers: A long tail of smaller regional or specialized generic companies also contribute to the competitive intensity.
These companies compete primarily on price, product availability, and established distribution channels. The low profit margins in the generic ophthalmic space necessitate high-volume sales.
Indirect Competitors (Other Drug Classes and Formulations):
Travoprost competes with other classes of ophthalmic medications and alternative therapeutic approaches:
-
Other Prostaglandin Analogs:
- Latanoprost: The first-in-class prostaglandin analog, widely available generically. Often considered the benchmark for efficacy and price within the class.
- Bimatoprost: Available as a generic and in branded formulations (e.g., Lumigan).
- Tafluprost: Another prostaglandin analog, available generically and in some branded formulations.
- Comparison: All prostaglandin analogs are generally considered first-line treatments with similar efficacy in lowering IOP. Price and patient response can differentiate choices. Generic travoprost and latanoprost are typically among the most cost-effective.
-
Fixed-Dose Combinations (FDCs):
- Travoprost + Timolol: Combines a prostaglandin analog with a beta-blocker. Offers increased IOP lowering with once-daily dosing.
- Travoprost + Brimonidine: Combines a prostaglandin analog with an alpha-agonist.
- Latanoprost + Timolol: A very common and widely prescribed FDC.
- Bimatoprost + Timolol: Another popular FDC.
- Comparison: FDCs simplify treatment regimens but are generally more expensive than monotherapy generics. They are prescribed when monotherapy is insufficient or for improved adherence.
-
Other Ophthalmic Drug Classes:
- Beta-Blockers (e.g., Timolol, Betaxolol): Often used as first-line or adjunctive therapy. Generic timolol is very inexpensive.
- Alpha-Adrenergic Agonists (e.g., Brimonidine): Used as monotherapy or in combination.
- Carbonic Anhydrase Inhibitors (e.g., Dorzolamide, Brinzolamide): Available as monotherapy or in combinations.
- Rho Kinase Inhibitors (e.g., Netarsudil): A newer class, often used when other agents are insufficient. Netarsudil has a higher price point.
-
Surgical Interventions:
- Laser Trabeculoplasty: A minimally invasive procedure.
- Glaucoma Surgery (e.g., Trabeculectomy, Glaucoma Drainage Devices): Reserved for more advanced cases or when medical therapy fails.
The competitive advantage of generic travoprost lies in its well-established efficacy, once-daily dosing, favorable safety profile compared to some older agents, and, crucially, its low cost. Physicians and payers prioritize cost-effectiveness for chronic conditions like glaucoma, making generic travoprost a default choice for many patients.
What are the Price Projections and Key Market Drivers for Travoprost?
Price projections for travoprost indicate continued stability at low levels, with modest potential for further erosion. The market is primarily driven by the dynamics of generic drug markets and the sustained demand for glaucoma treatments.
Price Projections:
- U.S. Market: The average selling price (ASP) for a 2.5 mL bottle of generic travoprost is projected to remain between \$25 and \$40. The aggressive pricing strategies of numerous generic manufacturers have compressed margins, leading to a plateau. Manufacturers may engage in competitive bidding for contracts with large healthcare systems and PBMs, resulting in further localized price reductions.
- European Market: Prices in Europe are anticipated to stay within the €20-€30 range per 2.5 mL bottle. Harmonization of pricing across EU member states, coupled with the strong presence of generic suppliers, will ensure continued price stability.
- Global Markets: In emerging markets, as generic travoprost gains wider access and adoption, prices will likely align with or slightly exceed those in developed markets, but significantly below branded historical pricing. This will be influenced by local regulatory approval processes and market entry strategies of generic manufacturers.
- Potential for Marginal Decline: While stabilization is the most probable scenario, a very gradual price decline of 1-2% annually is possible due to ongoing competitive pressures and increased payer leverage, especially if new, highly effective, and competitively priced treatments emerge.
Key Market Drivers:
- Glaucoma Prevalence: The increasing global prevalence of glaucoma, driven by aging populations and risk factors like diabetes and hypertension, ensures a consistent and growing demand for effective IOP-lowering treatments. According to the World Health Organization, glaucoma is a leading cause of irreversible blindness globally, affecting an estimated 64.3 million people worldwide in 2020.
- Generic Accessibility and Affordability: The low cost of generic travoprost makes it accessible to a broad patient population, including those with limited insurance coverage or high deductibles. This affordability is a primary driver of its widespread use.
- Physician Prescribing Habits: Ophthalmologists and optometrists are familiar with travoprost's efficacy and safety profile, making it a reliable choice for initiating or continuing glaucoma therapy. The established treatment algorithms often place prostaglandin analogs, including travoprost, as first-line options.
- Payer and Formulary Preference: Health insurance plans and PBMs strongly favor generic medications due to their cost-effectiveness, frequently placing generic travoprost on preferred drug lists and requiring its use before approving more expensive alternatives.
- Established Efficacy and Safety Profile: Travoprost has a proven track record of effectively reducing IOP and is generally well-tolerated. Its once-daily dosing regimen contributes to good patient adherence.
- Competition Dynamics: The intense competition among multiple generic manufacturers, including large multinational corporations and specialized ophthalmic companies, prevents significant price increases and promotes downward pricing pressure.
- Limited Innovation in New Monotherapy Agents: While innovation continues in drug delivery and combination therapies, the development of entirely new classes of IOP-lowering monotherapy drugs has been slow, allowing existing generic agents like travoprost to maintain their market position.
The continued demand for glaucoma treatment, coupled with the established advantages of generic travoprost, ensures its sustained presence and stable, low-cost market position.
Key Takeaways
- Generic Domination: Travoprost has transitioned to a fully generic market following the expiration of its key patents, with generic formulations holding over 95% market share in major developed economies.
- Price Compression: The average selling price for a 2.5 mL bottle of generic travoprost is expected to remain stable at approximately \$25-\$40 in the U.S. and €20-€30 in Europe, with potential for minor year-over-year declines.
- Sustained Demand: The increasing global prevalence of glaucoma, driven by an aging population, underpins consistent demand for effective and affordable IOP-lowering treatments like travoprost.
- Competitive Intensity: A large number of generic manufacturers compete aggressively on price, limiting profit margins and preventing significant price inflation.
- Payer Influence: Insurance providers and PBMs prioritize cost-effectiveness, solidifying the position of generic travoprost on preferred formularies and reinforcing its market share.
FAQs
What is the primary therapeutic use of travoprost?
Travoprost is primarily used to reduce elevated intraocular pressure (IOP) in patients diagnosed with open-angle glaucoma or ocular hypertension.
How does travoprost work to lower eye pressure?
Travoprost is a prostaglandin analog that enhances the outflow of aqueous humor from the eye through the uveoscleral pathway, thereby reducing intraocular pressure.
Is travoprost available as a branded medication?
While branded formulations of travoprost, such as Travatan Z, previously existed, the market is now predominantly served by generic travoprost due to patent expirations.
What are the most common side effects associated with travoprost use?
Common side effects include eye redness, changes in iris color (becoming more brown), eyelash growth, and itching or stinging of the eyes.
Can travoprost be used in combination with other glaucoma medications?
Yes, travoprost can be used as monotherapy or in combination with other classes of glaucoma medications, and fixed-dose combinations containing travoprost are also available.
Citations
[1] U.S. Patent 5,352,700. (1994). Ophthalmic prostaglandin derivatives. Alcon Laboratories, Inc. [2] U.S. Patent 7,279,505. (2007). Ophthalmic compositions containing prostaglandin analogs. Alcon, Inc. [3] World Health Organization. (2021). Blindness and vision impairment. https://www.who.int/news-room/fact-sheets/detail/blindness-and-vision-impairment
More… ↓
