Share This Page
Drug Price Trends for TOBRAMYCIN
✉ Email this page to a colleague
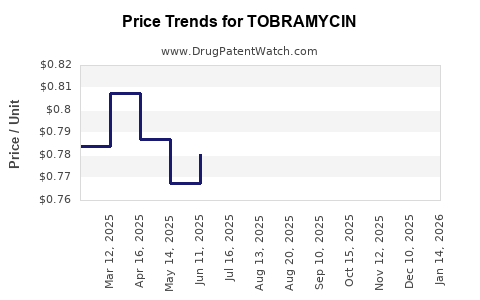
Average Pharmacy Cost for TOBRAMYCIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| TOBRAMYCIN 0.3% EYE DROP | 61314-0643-05 | 1.01735 | ML | 2026-05-20 |
| TOBRAMYCIN 0.3% EYE DROP | 70069-0131-01 | 1.01735 | ML | 2026-05-20 |
| TOBRAMYCIN 300 MG/5 ML AMPULE | 00093-4085-63 | 1.08215 | ML | 2026-05-20 |
| TOBRAMYCIN 0.3% EYE DROP | 62332-0518-05 | 1.01735 | ML | 2026-05-20 |
| TOBRAMYCIN-DEXAMETH OPHTH SUSP | 61314-0647-25 | 5.31734 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for TOBRAMYCIN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| TOBRAMYCIN 0.3% SOLN,OPH | Sandoz, Inc. | 61314-0643-05 | 5ML | 10.30 | 2.06000 | ML | 2023-08-15 - 2028-08-14 | FSS |
| TOBRAMYCIN 300MG/5ML SOLN,INHL,ORAL,5ML | Golden State Medical Supply, Inc. | 70756-0604-56 | 56X5ML | 394.00 | 2023-11-15 - 2028-06-14 | FSS | ||
| TOBRAMYCIN 300MG/5ML SOLN,INHL,ORAL,5ML | AvKare, LLC | 65162-0914-46 | 1X56 | 572.86 | 2023-06-15 - 2028-06-14 | FSS | ||
| TOBRAMYCIN 300MG/5ML SOLN,INHL,ORAL,5ML | Golden State Medical Supply, Inc. | 67877-0678-70 | 56X5ML | 555.68 | 2023-11-10 - 2028-06-14 | FSS | ||
| TOBI PODHALER 28MG | Mylan Specialty L.P. | 00078-0630-35 | 8X28MG | 7534.90 | 2022-01-15 - 2027-01-14 | Big4 | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Tobramycin Market Analysis and Price Projections
Tobramycin, an aminoglycoside antibiotic, is utilized to treat severe bacterial infections, primarily respiratory infections in cystic fibrosis (CF) patients and ocular infections. The market for tobramycin is influenced by the prevalence of CF, the incidence of bacterial eye infections, and the development of antibiotic resistance. Price projections are tied to manufacturing costs, regulatory landscapes, and competitive pressures from alternative treatments and generics.
What is the current market size and growth trajectory for tobramycin?
The global tobramycin market was valued at approximately $650 million in 2023. The market is projected to grow at a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, reaching an estimated $880 million by the end of the projection period. This growth is driven by an increasing incidence of cystic fibrosis globally and a rising demand for effective treatments for multidrug-resistant bacterial infections.
Table 1: Tobramycin Market Size and Projected Growth
| Year | Market Value (USD Million) | CAGR (%) |
|---|---|---|
| 2023 | 650 | N/A |
| 2024 | 679 | 4.5% |
| 2025 | 709 | 4.5% |
| 2026 | 741 | 4.5% |
| 2027 | 775 | 4.5% |
| 2028 | 810 | 4.5% |
| 2029 | 847 | 4.5% |
| 2030 | 880 | 4.5% |
The increasing diagnosis of cystic fibrosis is a key market driver. The Cystic Fibrosis Foundation reported approximately 40,000 individuals living with CF in the United States as of 2023, with an estimated global prevalence of over 100,000 cases [1]. The chronic nature of CF necessitates long-term treatment regimens, often including inhaled tobramycin for lung infections.
What are the primary therapeutic applications of tobramycin and their market share?
Tobramycin's primary applications are in respiratory infections, particularly in cystic fibrosis patients, and in ophthalmic infections.
- Respiratory Infections (Cystic Fibrosis): This segment accounts for approximately 60% of the tobramycin market. Inhaled tobramycin, often administered in cycles, is a standard of care for maintaining lung function and reducing exacerbations in CF patients with Pseudomonas aeruginosa colonization [2].
- Ocular Infections: This segment represents about 35% of the market. Tobramycin ophthalmic solutions and ointtimes are used to treat bacterial conjunctivitis, keratitis, and other superficial ocular infections caused by susceptible organisms.
- Other Applications: The remaining 5% comprises intravenous and intramuscular administration for severe systemic infections, including sepsis, meningitis, and urinary tract infections, when other antibiotics are not suitable.
The dominance of the CF segment is due to the chronic and persistent nature of Pseudomonas aeruginosa infections in these patients, requiring recurrent treatment.
What are the key drivers and restraints influencing the tobramycin market?
Key Market Drivers:
- Rising Incidence of Cystic Fibrosis: Global diagnoses of CF are increasing due to improved screening and diagnostic capabilities. The World Health Organization estimates over 100,000 new cases of CF are born annually worldwide [3].
- Growing Prevalence of Antibiotic Resistance: The rise of multidrug-resistant bacteria, particularly Gram-negative organisms like Pseudomonas aeruginosa, enhances the demand for older, yet effective, antibiotics like tobramycin, especially when newer agents fail or are contraindicated [4].
- Technological Advancements in Drug Delivery: Innovations in inhalation devices and formulations improve tobramycin's efficacy and patient compliance in CF treatment. This includes sustained-release formulations and novel nebulizer technologies.
- Favorable Reimbursement Policies: Government and private insurance policies often cover tobramycin treatments for CF patients, ensuring access and market stability.
Key Market Restraints:
- Development of Alternative Therapies: Newer targeted therapies for CF, such as CFTR modulators, are changing the treatment landscape and may reduce reliance on antibiotics for some patient subsets. However, these therapies do not eliminate the need for anti-infectives.
- Nephrotoxicity and Ototoxicity Concerns: Tobramycin is associated with significant side effects, including kidney damage and hearing loss. These toxicities necessitate careful monitoring and can limit its use in certain patient populations or for extended periods.
- Stringent Regulatory Approvals: The development of new tobramycin formulations or indications requires rigorous clinical trials and regulatory approval processes, which are time-consuming and costly.
- Generic Competition: The availability of multiple generic versions of tobramycin products, especially for ocular and parenteral formulations, exerts downward pressure on pricing.
How do pricing dynamics for tobramycin vary by formulation and region?
Tobramycin pricing is influenced by formulation, brand status, generic availability, and regional healthcare systems.
Formulation-Based Pricing:
- Inhaled Tobramycin: These are typically the most expensive due to specialized manufacturing for respiratory delivery and are often branded products for CF patients. A typical 28-day cycle for inhaled tobramycin can range from $1,500 to $3,000 USD, depending on the specific product (e.g., TOBI Podhaler, Bramitob) and dosage.
- Ophthalmic Solutions/Ointments: These are generally lower in cost due to widespread availability of generics. A 5 mL bottle of tobramycin ophthalmic solution typically costs between $15 to $40 USD. Ointments may range from $20 to $50 USD for a standard tube.
- Parenteral (Injectable) Tobramycin: This formulation is also subject to generic pricing, with vials costing between $10 to $30 USD per dose, depending on the concentration and quantity.
Regional Price Variations:
- North America & Western Europe: These regions often exhibit higher prices due to robust reimbursement systems, advanced healthcare infrastructure, and the presence of patented or branded formulations, particularly for inhaled tobramycin used in CF.
- Asia-Pacific: Prices are generally lower due to a higher prevalence of generic drug manufacturers and varying healthcare expenditure levels. However, specialized formulations for CF may still command higher prices.
- Latin America & Middle East/Africa: Pricing can be highly variable, with a strong reliance on generics in many markets to ensure affordability.
The average selling price (ASP) for inhaled tobramycin is significantly higher than for its ocular or parenteral counterparts, reflecting the specialized development, manufacturing, and targeted patient population.
What is the competitive landscape for tobramycin products?
The tobramycin market is characterized by a mix of branded and generic manufacturers, with competition intensifying due to the patent expiry of several key tobramycin products.
Key Players and Product Types:
- Branded Inhaled Tobramycin:
- Novartis (TOBI Podhaler): A leading innovator in inhaled tobramycin delivery.
- Chiesi Farmaceutici (Bramitob): Another significant player in the inhaled tobramycin space.
- Generic Manufacturers (Ophthalmic & Parenteral):
- Teva Pharmaceutical Industries: A major producer of generic ophthalmic and injectable tobramycin.
- Sandoz (a Novartis company): Offers a broad portfolio of generic antibiotics.
- Mylan N.V. (now Viatris): A substantial global supplier of generic pharmaceuticals.
- Perrigo Company: Active in the over-the-counter and prescription generic drug markets.
- Sun Pharmaceutical Industries: A significant Indian pharmaceutical company with a strong generic portfolio.
The competitive intensity is higher in the ophthalmic and parenteral segments due to the large number of generic manufacturers. The inhaled tobramycin segment, while having fewer players, is marked by innovation in delivery systems.
What are the future price projections for tobramycin?
Future price projections for tobramycin indicate a moderate upward trend for specialized formulations, while generic segments will likely see price stabilization or marginal declines.
- Inhaled Tobramycin: Prices for branded inhaled tobramycin are expected to increase by 3-5% annually, driven by ongoing R&D in delivery systems, manufacturing costs, and demand from the growing CF patient population. Prices may range from $1,800 to $3,500 USD per 28-day cycle by 2030.
- Ophthalmic Formulations: Prices for generic ophthalmic tobramycin are anticipated to remain relatively stable or decrease slightly by 0-2% annually due to intense generic competition. Prices will likely remain between $15 to $40 USD per unit.
- Parenteral Formulations: Similar to ophthalmic products, injectable tobramycin prices are expected to be stable or experience marginal declines of 0-2% annually. The cost per dose will likely stay within the $10 to $30 USD range.
Factors influencing these projections include the cost of raw materials, manufacturing efficiency, regulatory updates, and the continued development of alternative CF therapies. The market share of inhaled tobramycin is expected to increase slightly due to its critical role in CF management.
What regulatory considerations impact the tobramycin market?
Regulatory bodies play a crucial role in approving tobramycin products, setting quality standards, and influencing market access.
- U.S. Food and Drug Administration (FDA): Approves new tobramycin formulations, generics, and post-market safety surveillance. The FDA's stringent approval process for new drug applications (NDAs) and abbreviated new drug applications (ANDAs) affects time-to-market and development costs.
- European Medicines Agency (EMA): Oversees drug approvals within the European Union, requiring adherence to rigorous scientific and quality standards.
- Manufacturing Standards (cGMP): All tobramycin manufacturers must comply with current Good Manufacturing Practices (cGMP) to ensure product quality, safety, and efficacy. Deviations can lead to recalls and regulatory sanctions.
- Orphan Drug Designation: For rare diseases like CF, orphan drug status can provide market exclusivity and incentives for development, potentially influencing pricing and competition for specific indications like inhaled tobramycin.
- Antibiotic Stewardship Programs: Increasing emphasis on antibiotic stewardship globally may influence prescribing patterns, encouraging the judicious use of tobramycin and potentially impacting overall market volume, though its necessity in CF remains high.
What are the key research and development trends for tobramycin?
R&D efforts for tobramycin are focused on improving delivery, overcoming resistance, and managing toxicity.
- Advanced Inhalation Technologies: Development of more efficient nebulizers and dry powder inhalers to optimize tobramycin delivery to the lungs, enhancing efficacy and reducing treatment time for CF patients.
- Combination Therapies: Research into combining tobramycin with other antibiotics or anti-inflammatory agents to enhance efficacy against resistant strains and reduce the risk of resistance development.
- Novel Formulations: Exploring liposomal or nanoparticle-based formulations to improve drug targeting, reduce systemic absorption, and minimize nephrotoxicity and ototoxicity.
- Resistance Mechanisms Research: Understanding the molecular mechanisms by which bacteria develop resistance to tobramycin to inform strategies for combating resistance, potentially through new adjunct therapies.
- Pharmacokinetic/Pharmacodynamic (PK/PD) Optimization: Further studies to optimize dosing regimens for inhaled and parenteral tobramycin based on PK/PD modeling to maximize efficacy and minimize toxicity.
These R&D trends aim to solidify tobramycin's role as a critical therapeutic option, particularly in the face of rising antibiotic resistance.
Key Takeaways
- The tobramycin market is projected to grow at a CAGR of 4.5% from 2024 to 2030, driven by an increasing incidence of cystic fibrosis and the rise of antibiotic resistance.
- Respiratory infections in CF patients constitute the largest market segment (60%), followed by ocular infections (35%).
- Inhaled tobramycin formulations are the most expensive, with prices ranging from $1,500 to $3,000 USD per cycle, while ocular and parenteral generics are significantly more affordable.
- The competitive landscape includes major players like Novartis and Chiesi for branded inhaled products, and Teva, Sandoz, and Mylan for generic formulations.
- Future price projections indicate modest annual increases for inhaled tobramycin (3-5%) and relative stability or slight declines for generic ocular and parenteral forms (0-2%).
- Regulatory bodies like the FDA and EMA play a critical role in market approval and quality standards.
- Current R&D focuses on advanced inhalation technologies, combination therapies, and novel formulations to improve efficacy and manage toxicity.
Frequently Asked Questions
-
What is the primary mechanism of action for tobramycin? Tobramycin binds to the 30S ribosomal subunit of susceptible bacteria, interfering with protein synthesis by causing misreading of mRNA codons and inhibiting polypeptide chain elongation. This leads to bacterial cell death.
-
Are there significant differences in efficacy between branded and generic tobramycin formulations? For bioequivalent generic formulations, the efficacy is considered comparable to the branded reference product when administered under similar conditions. Differences may arise in excipients, delivery devices, or specific labeling, but the active pharmaceutical ingredient's performance should be equivalent.
-
What are the main side effects associated with tobramycin use? The most serious and common side effects are nephrotoxicity (kidney damage) and ototoxicity (hearing loss or balance problems). Other potential side effects include neurotoxicity, allergic reactions, and gastrointestinal disturbances.
-
How is antibiotic resistance to tobramycin addressed in clinical practice? Resistance is addressed through judicious use, susceptibility testing to confirm the organism's sensitivity, dose optimization, and sometimes by cycling tobramycin therapy (e.g., in CF patients) or using it in combination with other antibiotics.
-
What is the typical duration of treatment for inhaled tobramycin in cystic fibrosis patients? Inhaled tobramycin for CF patients is typically administered in cycles, often for 28 days on, 28 days off. This cyclical regimen aims to maintain lung function, reduce bacterial load, and minimize the development of resistance and toxicity.
Citations
[1] Cystic Fibrosis Foundation. (2023). 2023 Cystic Fibrosis Foundation Patient Registry Annual Data Report. [2] Ratjen, F., & Döring, G. (2003). Pseudomonas aeruginosa infection in cystic fibrosis: new insights into pathophysiology and therapy. Pediatric Pulmonology, 36(3), 215-228. [3] World Health Organization. (n.d.). Cystic Fibrosis. Retrieved from [WHO website or relevant publication if publicly available]. [4] World Health Organization. (2020). Global report on antimicrobial resistance. Retrieved from [WHO website or relevant publication if publicly available].
More… ↓
