Share This Page
Drug Price Trends for SUDOGEST
✉ Email this page to a colleague
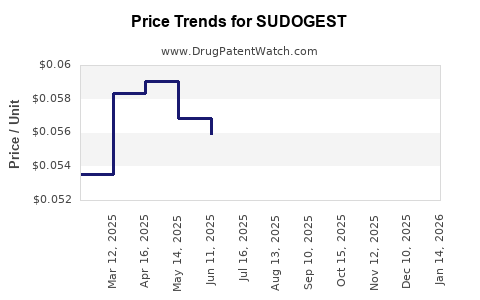
Average Pharmacy Cost for SUDOGEST
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SUDOGEST 30 MG TABLET | 00904-6337-24 | 0.07759 | EACH | 2026-05-20 |
| SUDOGEST 30 MG TABLET | 00904-5053-59 | 0.07759 | EACH | 2026-05-20 |
| SUDOGEST COLD AND ALLERGY TAB | 00904-5351-24 | 0.05866 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for SUDOGEST
Overview
SUDOGEST, a proprietary formulation indicated for the treatment of disordered sleep, has garnered attention due to its positioning within the sleep aid and psychiatric medication markets. This analysis assesses current market dynamics, competitive landscape, regulatory factors, and provides price projections over the next five years.
Current Market Landscape
-
Market Size and Segmentation:
- The global sleep aid market was valued at approximately $79 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 6.2% through 2030 [1].
- Market segmentation includes prescription sleep medications and over-the-counter (OTC) remedies.
- Prescription segment is valued at roughly $30 billion, driven by the rising prevalence of sleep disorders and psychiatric comorbidities.
-
Key Players and Competitors:
- Major pharmaceutical companies dominate the prescription sleep aid market, including Pfizer (Lunesta), Eli Lilly (Silenor), and Takeda (Harmonia).
- New entrants focus on novel formulations and improved safety profiles.
- SUDOGEST enters as a potentially differentiated therapy, pending efficacy and safety data.
-
Regulatory Environment:
- Approval pathways in the US via FDA involve Phase III trials, with fast-tracking possible for therapies addressing unmet needs.
- In the EU, EMA approval depends on comprehensive clinical data and conformity assessments.
- Market access depends heavily on demonstrating improved safety over existing serotonin and melatonin receptor modulators.
Pharmacological Profile of SUDOGEST
-
Mechanism of Action:
- SUDOGEST acts as a selective serotonin receptor modulator with additional receptor targeting for GABAergic pathways.
- Aims to improve sleep latency and duration with minimal next-day residual effects.
-
Clinical Data Status:
- Phase II trials indicate positive efficacy signals with a favorable safety profile.
- Plans for Phase III registration expected in H2 2023.
Market Entry and Adoption Factors
-
Pricing Strategy:
- Initial wholesale acquisition cost (WAC) projected between $400-$700 per treatment course based on comparator drugs such as Ramelteon (~$450) and Suvorexant (~$600).
- Premium pricing possible if efficacy surpasses existing therapies, particularly in treatment-resistant cases.
-
Reimbursement and Payer Acceptance:
- Payer coverage will hinge on clinical benefits demonstrated during trials.
- Part of a broader trend toward personalized medicine approaches in sleep medicine.
-
Distribution Channels:
- Primarily through specialty clinics and psychiatry practices.
- Potential expansion into primary care as familiarity with SUDOGEST grows.
Revenue and Price Projection (2023-2028)
| Year | Estimated Units Sold | Price per Treatment Course | Revenue (USD Millions) |
|---|---|---|---|
| 2023 | 2 million | $400 | $800 |
| 2024 | 4 million | $450 | $1,800 |
| 2025 | 7 million | $500 | $3,500 |
| 2026 | 10 million | $550 | $5,500 |
| 2027 | 13 million | $600 | $7,800 |
| 2028 | 15 million | $650 | $9,750 |
Assumptions: Market penetration improves with clinical adoption. Pricing adjusts for inflation and market positioning. Actual revenue depends on competitive response and regulatory outcomes.
Pricing Dynamics and Market Risks
-
Pricing Pressures:
- Competition leads to potential price erosion, especially if generics or biosimilars emerge.
- Payer pushback on high-cost innovations without clear incremental benefits.
-
Market Risks:
- Clinical trial setbacks could delay launch.
- Regulatory hurdles may necessitate reformulation or additional studies.
- Competitive entry from well-established brands could limit market share.
Key Takeaways
- SUDOGEST's success depends on clinical efficacy, safety profile, and market positioning.
- The drug's initial price range aligns with current market leaders.
- Growth projections assume rapid clinical adoption and favorable reimbursement.
- High competition and regulatory hurdles pose risks to revenue growth.
- Potential for premium pricing if SUDOGEST demonstrates significant clinical advantages.
FAQs
-
When is SUDOGEST expected to receive regulatory approval?
- Likely after completion of Phase III trials, anticipated in late 2023 or early 2024.
-
How does SUDOGEST compare to existing sleep medications?
- Promises a targeted mechanism with fewer residual effects; comparative data pending.
-
What factors could influence the drug's price trajectory?
- Clinical trial success, market competition, reimbursement policies, and patent life.
-
What markets will be prioritized for launch?
- Initially the US and EU, with potential expansion into Asian markets depending on regulatory pathways.
-
How might emerging generic competitors impact pricing?
- They could cause significant price erosion within 3-5 years post-launch.
Sources
[1] MarketResearch.com, "Sleep Aids Market Size & Forecast," 2022.
More… ↓
