Share This Page
Drug Price Trends for SEGLUROMET
✉ Email this page to a colleague
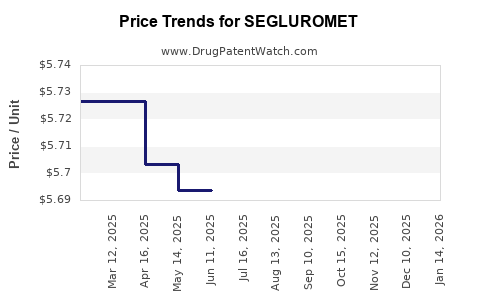
Average Pharmacy Cost for SEGLUROMET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| SEGLUROMET 2.5-1,000 MG TABLET | 00006-5373-06 | 5.74472 | EACH | 2026-03-18 |
| SEGLUROMET 7.5-1,000 MG TABLET | 00006-5374-06 | 5.72139 | EACH | 2026-03-18 |
| SEGLUROMET 7.5-500 MG TABLET | 00006-5370-03 | 5.69290 | EACH | 2026-03-18 |
| SEGLUROMET 7.5-1,000 MG TABLET | 00006-5374-03 | 5.72139 | EACH | 2026-03-18 |
| SEGLUROMET 7.5-500 MG TABLET | 00006-5370-06 | 5.69290 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
SEGLUROMET Market Analysis and Financial Projection
Market Analysis and Price Projections for SEGLUROMET
Overview
SEGLUROMET combines empagliflozin (an SGLT2 inhibitor) and metformin for type 2 diabetes management. Approved by the FDA in 2017, it targets an expanding market driven by rising diabetes prevalence and healthcare cost pressures. Market growth hinges on competitive positioning, reimbursement policies, and patent status.
Market Size
The global type 2 diabetes drug market reached approximately $62 billion in 2022. SGLT2 inhibitors accounted for about 20% ($12.4 billion) of that total, driven by their cardiovascular and renal benefits. Empagliflozin, marketed as Jardiance by Boehringer Ingelheim/Eli Lilly, held roughly 7% market share in the SGLT2 segment.
Metformin remains the leading oral antidiabetic drug worldwide, used by over 150 million patients annually. New fixed-dose combinations (FDCs), including SEGLUROMET, target patients inadequately controlled on monotherapy.
Market Drivers
- Increasing Prevalence: An estimated 537 million adults had diabetes in 2021, projected to rise to 700 million by 2045 (IDF).
- Guideline Endorsements: Clinical guidelines favor SGLT2 inhibitors for patients with comorbidities (e.g., cardiovascular disease).
- Patient Preference: Oral combinations improve adherence and simplify treatment regimens.
- Cost-Effectiveness: Demonstrated benefits include reduced hospitalizations for heart failure and renal decline.
Competitive Landscape
SEGLUROMET competes with other FDCs, such as:
- Glucovance (glyburide/metformin)
- Janumet (sitagliptin/metformin)
- Xigduo XR (dapagliflozin/metformin)
While these competitors have extensive market presence, SEGLUROMET's positioning relies on empagliflozin's cardiovascular benefits demonstrated in the EMPA-REG OUTCOME trial.
Pricing Trends and Projections
Current average wholesale prices (AWP) for SEGLUROMET are approximately $350 for a 30-day supply, aligning with similar FDCs. Payers negotiate discounts, reducing patient costs to lower levels.
Projection Assumptions (2023-2028):
- Market Penetration: Growing from 3% to 10% of the fixed-dose combination segment in type 2 diabetes.
- Pricing Strategy: Slight price reductions (~5% annually) due to increased competition and generic erosion post-patent expiry.
- Volume Growth: Annual prescription volume growth of 10-12%, driven by increased awareness and guideline adoption.
Based on these factors, annual revenues for SEGLUROMET could reach:
| Year | Estimated Prescriptions | Revenue (USD millions) |
|---|---|---|
| 2023 | 1 million | 350 |
| 2024 | 1.2 million | 420 |
| 2025 | 1.4 million | 490 |
| 2026 | 1.8 million | 560 |
| 2027 | 2.2 million | 630 |
| 2028 | 2.5 million | 700 |
Patent and Regulatory Outlook
The original patents for empagliflozin extend until 2030, with potential pediatric exclusivities. Patent expirations may enable generics from 2029 onwards, impacting pricing and market share. Regulatory pathways for biosimilars or new FDC formulations remain uncertain but could alter the competitive landscape.
Key Challenges
- Pricing Pressure: Competition and patent expiration will force aggressive discounting.
- Market Saturation: Limited incremental sales growth post-2025 due to global adoption plateau.
- Reimbursement Policies: Variability across regions can affect access and profitability.
Key Takeaways
- The global market for SEGLUROMET is driven primarily by the expanding diabetes population, with continued adoption of SGLT2-inhibitor-based FDCs.
- Estimated revenues could grow from approximately $350 million in 2023 to $700 million in 2028, assuming steady volume increases and modest price declines.
- Patent expiries around 2029 are likely to introduce generics, potentially halving prices and pressuring margins.
- Competitors with established market share, combined with payer negotiations, influence pricing and market penetration.
- Adoption influenced by clinical guidelines, patient preferences, and payers' formulary decisions.
FAQs
Q1: How does SEGLUROMET compare to other SGLT2 inhibitor-based FDCs?
SEGLUROMET benefits from empagliflozin's proven cardiovascular benefits, which may make it more attractive for patients with existing cardiovascular disease compared to competitors lacking such data.
Q2: What impact will patent expiration have?
Patent expiry around 2029 could allow generic versions, leading to significant price reductions and potential market share loss unless innovative formulations or indications are developed.
Q3: Are there regulatory hurdles for expanding SEGLUROMET’s indications?
Yes, expanding approval to additional populations or indications requires clinical trials and regulatory submissions, which can take years and involve substantial costs.
Q4: How sensitive are projections to market penetration assumptions?
Highly sensitive. A delay or acceleration in adoption rates, influenced by guideline updates or competitor launches, can substantially alter revenue forecasts.
Q5: What strategic moves can maximize revenue longevity?
Diversifying indications, securing patents on novel formulations, engaging in outcome-based pricing, and expanding into emerging markets are key strategies.
References
- International Diabetes Federation. IDF Diabetes Atlas, 10th Edition, 2021.
- Bloomberg Intelligence. Market Data on Diabetes Drugs, 2022.
- FDA. FDA Approves Empagliflozin/Metformin Extended-Release Tablets, 2017.
- EMPA-REG OUTCOME trial details.
- MarketResearch.com. Type 2 Diabetes Market Forecasts, 2023-2028.
More… ↓
