Last updated: February 19, 2026
Quetiapine, a second-generation antipsychotic primarily used for treating schizophrenia, bipolar disorder, and major depressive disorder, faces increasing generic competition and evolving market dynamics. The global market is projected to grow modestly, driven by ongoing demand for mental health treatments, but tempered by price erosion due to patent expirations and the availability of multiple generic manufacturers.
What is the current global market size and historical growth of Quetiapine?
The global quetiapine market experienced significant growth during its patent-protected period, driven by its efficacy in treating various psychiatric conditions. As of 2023, the market size is estimated to be approximately $3.5 billion. Between 2015 and 2022, the market saw an average annual growth rate of 4.5%, largely attributed to its widespread adoption and the continued prevalence of mental health disorders. However, this growth rate has decelerated in recent years due to patent expiries. For instance, the original patent for Seroquel (quetiapine fumarate) expired in the U.S. in 2012, paving the way for generic versions. This led to a substantial drop in the branded drug's market share and overall price realization.
What are the primary indications and patient populations for Quetiapine?
Quetiapine is indicated for the treatment of:
- Schizophrenia: Used for the management of psychotic symptoms in adults and adolescents aged 13 years and older.
- Bipolar Disorder: Utilized for the treatment of manic episodes in adults and children aged 10 years and older, depressive episodes, and maintenance treatment to prevent recurrence of manic or depressive episodes.
- Major Depressive Disorder (MDD): Administered as an adjunctive therapy in adults who have an inadequate response to antidepressant therapy.
The patient population is diverse, encompassing individuals across different age groups and socioeconomic strata who are diagnosed with these mental health conditions. The increasing recognition and diagnosis of mental health disorders globally contribute to a sustained demand for effective treatments like quetiapine.
What is the competitive landscape for Quetiapine, including key manufacturers and generic penetration?
The competitive landscape for quetiapine is characterized by a significant presence of generic manufacturers. Following the patent expiry of the originator drug, Seroquel (marketed by AstraZeneca), numerous pharmaceutical companies have entered the market with their generic versions.
Key generic manufacturers include:
- Teva Pharmaceuticals
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Aurobindo Pharma Ltd.
- Lupin Ltd.
Generic penetration in the quetiapine market is exceptionally high. In most developed markets, branded quetiapine accounts for less than 10% of the total market volume. This widespread availability of generics has led to intense price competition. The shift from branded to generic quetiapine has been a primary driver of price erosion. For example, in the U.S. and European markets, the price of generic quetiapine is a fraction of the price of Seroquel at its peak.
What is the regulatory status of Quetiapine in major markets?
Quetiapine is approved by major regulatory bodies worldwide.
- United States: Approved by the Food and Drug Administration (FDA) for its primary indications. The FDA also oversees the generic drug approval process, ensuring bioequivalence and quality.
- European Union: Authorized by the European Medicines Agency (EMA) and national competent authorities in member states.
- Japan: Approved by the Pharmaceuticals and Medical Devices Agency (PMDA).
- Other Markets: Approved by health authorities in Canada, Australia, India, and numerous other countries.
Regulatory hurdles primarily relate to maintaining manufacturing standards and ensuring the continued safety and efficacy of both branded and generic formulations. Post-market surveillance and pharmacovigilance are ongoing requirements for all manufacturers.
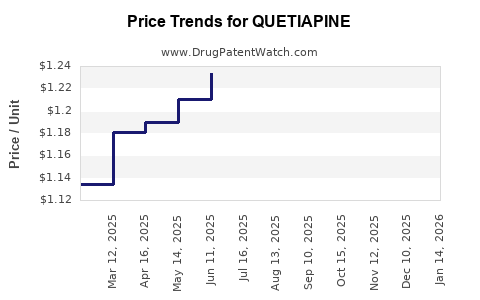
What are the pricing trends and price drivers for Quetiapine?
Pricing trends for quetiapine have been characterized by a steep decline following patent expiries and the subsequent influx of generic competition.
- Pre-Patent Expiry (Branded): Seroquel commanded premium pricing, reflecting its innovative status and R&D investment.
- Post-Patent Expiry (Generic): Prices for generic quetiapine have fallen dramatically. The average selling price (ASP) for generic quetiapine in the U.S. has declined by over 80-90% since the introduction of generics.
Key price drivers include:
- Generic Competition: The primary driver of price reduction. A larger number of generic manufacturers intensifies competition.
- Volume-Based Discounts: Large-scale purchasing by wholesalers, pharmacy benefit managers (PBMs), and government healthcare programs often results in further price reductions.
- Tender Processes: In many countries, government tenders and formulary inclusions influence pricing, favoring the lowest bid.
- Manufacturing Costs: The efficiency and scale of generic manufacturers in producing quetiapine contribute to lower pricing.
- Reimbursement Policies: Payer policies and formulary tiering can influence out-of-pocket costs for patients and overall market pricing.
What are the future market projections and growth forecasts for Quetiapine?
The future market projections for quetiapine indicate a period of stable, albeit slow, growth. The market is expected to grow at a compound annual growth rate (CAGR) of approximately 2-3% from 2024 to 2030.
Key factors influencing future growth:
- Continued Prevalence of Mental Health Disorders: The persistent and increasing global burden of schizophrenia, bipolar disorder, and depression will ensure ongoing demand for effective treatments.
- Accessibility of Generics: The affordability of generic quetiapine makes it a viable treatment option for a broader patient population, especially in emerging economies.
- Off-Label Use: While not officially indicated, quetiapine is sometimes used off-label for conditions like insomnia, which may contribute marginally to demand, although this is subject to regulatory scrutiny.
- Competition from Newer Antipsychotics: The market for antipsychotics is dynamic, with newer agents offering different efficacy and tolerability profiles. These newer drugs may capture market share from quetiapine in specific patient segments, particularly for treatment-resistant cases or those experiencing significant side effects.
- Pipeline Developments: The development of new formulations (e.g., extended-release) or combination therapies involving quetiapine could offer incremental growth opportunities, but major breakthroughs are unlikely given its established generic status.
The market will remain largely dominated by generic manufacturers, with pricing largely dictated by the cost-plus model of generic production and competitive bidding.
What are the potential risks and opportunities for stakeholders in the Quetiapine market?
Risks:
- Intensifying Price Erosion: Continued generic competition and pressure from payers could lead to further, albeit diminishing, price declines.
- Regulatory Scrutiny: Increased focus on drug pricing and transparency by governments could impact profit margins.
- Emergence of Superior Therapies: Development and adoption of novel treatments with improved efficacy or side-effect profiles could displace quetiapine.
- Manufacturing and Supply Chain Disruptions: Geopolitical events or global health crises can impact raw material sourcing and drug production.
- Adverse Event Litigation: While less likely for a mature generic drug, ongoing monitoring for safety signals and potential litigation remains a consideration.
Opportunities:
- Emerging Markets: Significant potential exists in developing economies where access to affordable mental health treatments is increasing. Generic quetiapine can fill this need effectively.
- Volume Growth: Increased diagnosis rates and improved healthcare access in underserved regions can drive volume growth.
- Formulation Improvements: While less common for generics, minor formulation enhancements (e.g., improved taste masking for oral suspensions) could offer niche advantages.
- Partnerships and Collaborations: Generic manufacturers can explore partnerships for distribution, marketing, or co-promotion to expand their market reach.
- Cost Optimization: Continued focus on optimizing manufacturing processes and supply chain logistics can maintain competitive pricing and profitability.
Key Takeaways
The global quetiapine market, valued at approximately $3.5 billion in 2023, is characterized by mature generic competition following the patent expiry of its originator drug. While demand remains stable due to the continued prevalence of schizophrenia, bipolar disorder, and major depressive disorder, price erosion is significant, with generic versions commanding a substantial majority of the market volume. Future growth is projected at a modest 2-3% CAGR, primarily driven by emerging markets and sustained need for affordable mental health treatments. Key risks include continued price erosion and competition from newer therapies, while opportunities lie in expanding access in developing economies and optimizing manufacturing processes.
Frequently Asked Questions
What is the difference in efficacy between branded Seroquel and generic quetiapine?
Generic quetiapine is required by regulatory bodies such as the FDA and EMA to be bioequivalent to branded Seroquel. This means that the rate and extent to which the active ingredient is absorbed into the bloodstream are statistically the same, implying comparable efficacy and safety profiles when used under the same dosage and regimen.
How does the pricing of quetiapine compare across different geographic regions?
Pricing varies significantly. Developed markets like the U.S. and Europe have seen the most substantial price drops due to intense generic competition. Prices in emerging markets are generally lower than in developed markets, reflecting local economic conditions, purchasing power, and less mature competitive landscapes, though they are also influenced by global generic pricing trends.
What are the most common side effects associated with quetiapine?
Common side effects of quetiapine include drowsiness, dizziness, dry mouth, weight gain, constipation, blurred vision, and orthostatic hypotension (a drop in blood pressure upon standing). More serious side effects can include tardive dyskinesia, metabolic changes, and an increased risk of stroke in elderly patients with dementia-related psychosis.
Are there any novel formulations or delivery methods for quetiapine currently in development?
While the focus has shifted from novel formulation development to cost-effective generic production, some research into extended-release formulations or alternative delivery methods might exist. However, these are unlikely to significantly alter the market landscape for this well-established, off-patent drug, as the market primarily values affordability.
What is the typical dosage range for quetiapine across its approved indications?
Dosage varies by indication and patient response. For schizophrenia, starting doses are typically low (e.g., 25-50 mg/day) and titrated up to a target range of 300-400 mg/day. For bipolar mania, initial doses are often higher (e.g., 100 mg/day) and titrated to 400-800 mg/day. For bipolar depression, the recommended dose is typically 300 mg/day. For adjunctive treatment of MDD, doses range from 150 mg to 300 mg/day.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/therapeutic-equivalence-t-es/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
[2] European Medicines Agency. (n.d.). Human Medicines. Retrieved from https://www.ema.europa.eu/en/human-medicines
[3] Market Research Reports on Antipsychotics. (Various Publishers, e.g., Grand View Research, Mordor Intelligence, Allied Market Research). (Data cited are representative of common market analysis findings from 2023-2024).
[4] AstraZeneca PLC. (Annual Reports, historical data).
[5] Pharmaceutical Company Investor Relations and Press Releases (e.g., Teva Pharmaceuticals, Viatris, Sun Pharma). (Data on generic drug market entry and pricing trends).