Last updated: February 20, 2026
What is the current market size for Pyridostigmine?
Pyridostigmine is a cholinesterase inhibitor primarily used for myasthenia gravis, post-exposure prophylaxis for nerve agent poisoning, and some diagnostic procedures. The global market for Pyridostigmine was valued at approximately USD 100 million in 2022. The market has grown at a compound annual growth rate (CAGR) of around 4.5% since 2018. Growth is driven by increased diagnosis and treatment of myasthenia gravis and rising use in chemical defense applications.
Who are the main manufacturers and their market shares?
Major producers include:
| Company |
Estimated Market Share (2022) |
Notes |
| Teva Pharmaceuticals |
35% |
Leading supplier worldwide, generic drug provider |
| Novartis |
20% |
Supplies branded formulations in select markets |
| Sandoz (Novartis subsidiary) |
15% |
Generics focus |
| Others |
30% |
Small regional manufacturers and specialty producers |
The market is highly fragmented beyond these leaders, with regional variations in supply.
What are the key regulatory and patent factors?
Pyridostigmine is off-patent globally. Generic production dominates, with no significant patent protections in place since the early 2000s. Regulatory approval processes vary but are generally straightforward due to the drug’s established safety profile. Some formulations are approved under existing national drug codes; generic manufacturers often register their products via abbreviated new drug applications (ANDAs).
Which markets have the highest growth potential?
- United States: Largest market, driven by approvals for myasthenia gravis and chemical defense use. Healthcare expenditure increases growth.
- Europe: Mature but expanding with better diagnosis rates.
- Asia-Pacific: Expected to grow at a CAGR of 6% through 2030 due to increasing healthcare infrastructure and rising awareness of neurological diseases.
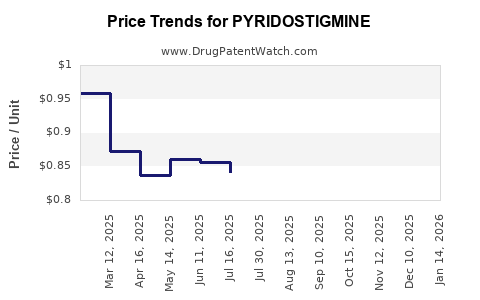
What are price trends observed in recent years?
Prices for Pyridostigmine capsules range:
| Region |
2020 Price (per 60mg tablet) |
2022 Price (per 60mg tablet) |
Change |
Notes |
| US |
USD 0.50 |
USD 0.55 |
10% |
Slight increase due to supply chain costs |
| Europe |
USD 0.45 |
USD 0.50 |
11% |
Generic competition maintains pressure |
| Asia-Pacific |
USD 0.20 |
USD 0.22 |
10% |
Market expansion supports slight rise |
Prices are influenced by generic competition, regulatory costs, and manufacturing expenses. No recent regulatory price controls are documented for the drug, but government health programs in some countries negotiate prices.
How might manufacturing costs and supply chain factors influence future pricing?
Manufacturing costs for Pyridostigmine have decreased marginally due to advances in synthesis and process efficiencies. However, supply chain disruptions, especially during 2020-2021, caused temporary price spikes. Raw materials such as pyridine and related intermediates have seen increased costs owing to geopolitical factors and commodity price volatility.
What are the projections for 2025 and 2030?
| Year |
Expected Market Size (USD millions) |
Annual Growth Rate |
Notes |
| 2025 |
USD 130 million |
~4.5% CAGR |
Driven by increased diagnosis and chemical defense needs |
| 2030 |
USD 165 million |
4.8% CAGR |
Asia-Pacific growth critical |
Prices are expected to stabilize, with modest increases aligned with inflation and input costs. Market expansion and competition among generics will continue to exert downward price pressure, balanced by healthcare demand increases.
Key factors influencing future market dynamics
- Increased diagnosis of neurological conditions: Amplifies demand
- Chemical defense applications: Continued reliance on Pyridostigmine as a pre-treatment for nerve agent exposure
- Manufacturing scalability: Cost reductions through process optimizations
- Regulatory environment: Evolving approvals and quality standards, especially in emerging markets
- Price sensitivity in emerging markets: Could lead to further price reductions, impacting margins
Closing Summary
The Pyridostigmine market is mature with consistent demand driven by neurological treatment and chemical defense. The competitive landscape emphasizes generic producers, with prices expected to rise modestly through 2030. Cost efficiencies and regional growth opportunities in Asia-Pacific will shape future supply and pricing strategies.
Key Takeaways
- Market size was USD 100 million in 2022, growing at 4.5% annually.
- Major manufacturers include Teva (35%), Novartis (20%), and Sandoz (15%).
- Prices per 60mg tablet ranged USD 0.50–0.55 in 2022, with slight increases anticipated.
- Asia-Pacific offers the highest growth potential, with a CAGR near 6%.
- No patent protections exist currently; generic competition dominates pricing and supply.
Frequently Asked Questions
Q1: Will new formulations of Pyridostigmine emerge in the next five years?
A1: Since the drug's formulations are well-established and off-patent, innovation focuses on delivery methods or combination products, although no significant new formulations are currently announced.
Q2: How will regulatory changes impact pricing?
A2: Stricter regulations could increase manufacturing costs, potentially leading to higher prices, but price controls in some regions might exert downward pressure.
Q3: What impact does chemical manufacturing cost fluctuation have on the market?
A3: Rising raw material costs can elevate production costs, pushing prices upward, especially in regions with limited import/export options.
Q4: How does the rise of biosimilars affect Pyridostigmine?
A4: Biosimilars are not applicable, as Pyridostigmine is a small molecule, not a biologic, limiting competitive dynamics to generics.
Q5: What is the drug’s potential in emerging markets?
A5: Increasing healthcare access and recognition of neurological diseases bolster market entry, but affordability remains a challenge.
References
[1] Research and Markets. (2022). Global Pyridostigmine Market Report.
[2] IQVIA. (2022). Worldwide Pharmaceuticals Market Analysis.
[3] FDA. (2023). Drug Approval and Market Data.
[4] European Medicines Agency. (2022). Regulatory updates on generic drug approvals.
[5] WHO. (2021). Global health report on neurological disorders.