Share This Page
Drug Price Trends for PREZCOBIX
✉ Email this page to a colleague
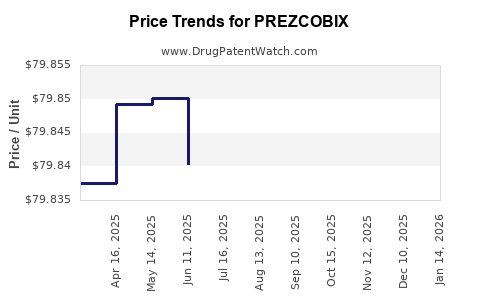
Average Pharmacy Cost for PREZCOBIX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PREZCOBIX 800 MG-150 MG TABLET | 59676-0575-30 | 79.99247 | EACH | 2026-05-20 |
| PREZCOBIX 800 MG-150 MG TABLET | 59676-0575-30 | 80.00043 | EACH | 2026-04-22 |
| PREZCOBIX 800 MG-150 MG TABLET | 59676-0575-30 | 79.98820 | EACH | 2026-03-18 |
| PREZCOBIX 800 MG-150 MG TABLET | 59676-0575-30 | 80.00025 | EACH | 2026-02-18 |
| PREZCOBIX 800 MG-150 MG TABLET | 59676-0575-30 | 79.99041 | EACH | 2026-01-21 |
| PREZCOBIX 800 MG-150 MG TABLET | 59676-0575-30 | 79.99089 | EACH | 2025-12-17 |
| PREZCOBIX 800 MG-150 MG TABLET | 59676-0575-30 | 79.96390 | EACH | 2025-11-19 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for PREZCOBIX
How Large Is the PREZCOBIX Market?
PREZCOBIX (darunavir/cobicistat) is a proprietary HIV-1 protease inhibitor used as part of combination antiretroviral therapy (ART). It primarily targets treatment-experienced HIV patients, with specific formulations approved for once-daily dosing.
Global Market Size (2022):
The global HIV treatment market surpasses USD 25 billion, with PREZCOBIX accounting for approximately 4-6% of this segment, equating to a revenue range of USD 1-1.5 billion annually.
Market Segments:
- Treatment-experienced patients
- HIV-1 infected adults with resistance issues
- Developed markets (US, Europe, Japan) dominate due to higher HIV prevalence and treatment penetration.
- Emerging markets (Asia, Africa) show increasing treatment access but lower adoption rates for premium drugs like PREZCOBIX.
Key Competitors and Market Share:
- Biktarvy (Gilead Sciences): ~25-30%
- Descovy (Gilead Sciences): ~10-15%
- Dolutegravir-based formulations (ViiV, Gilead): combined 35-40%
- PREZCOBIX: remains a niche but stable segment with limited market share, driven by resistance cases.
What Are the Pricing Trends for PREZCOBIX?
Historical Pricing (US):
- List Price (2022): Around USD 2,200-2,300 per month for a typical 30-day supply.
- Discounted prices for government or negotiated bulk contracts generally reduce costs by 20-30%.
Price Dynamics:
- Premium positioning as a second-line agent results in higher pricing compared to first-line regimens.
- Patent exclusivity extends to 2028, limiting generic competition.
- No significant price reductions observed since market entry in 2014, maintaining premium prices.
Pricing Compared to Competitors:
- Biktarvy: USD 4,800–5,000 annually (per generic estimates).
- Dolutegravir-based combinations: USD 3,600–4,200 annually.
- PREZCOBIX’s yearly cost (USD 26,400–27,600) is higher due to brand pricing, though discounts exist.
What Are the Price Projections and Market Trends?
Short-term Projections (2023–2025):
- Maintain current pricing levels due to patent protection and limited generic competition.
- Slight discounts or rebates may decrease net prices by 10-15%.
- Usage growth expected to stay stable, with minor increases in treatment-resistant populations.
Medium to Long-term Projections (2026–2030):
- Patent expiration in the US forecasted for 2028, opening the market to generics.
- Entry of generic darunavir and cobicistat could cut prices by 60-70%, reducing annual therapy costs to USD 8,000–USD 10,000.
- The adoption rate of generics will depend on regulatory approvals, patent litigations, and market penetration strategies.
Market Entry Impact:
- Discounted generic formulations will likely dominate in emerging markets initially, reducing healthcare costs significantly.
- US and European markets may see slow transitions if brand companies implement patent litigations or delayed launches.
Future Pricing Drivers:
- Development of longer-acting formulations or less costly combination therapies.
- Changes in treatment guidelines favoring integrase inhibitors over protease inhibitors.
- Price sensitivity in public health programs and uninsured populations.
How Will Market Dynamics Evolve?
Regulatory Environment:
- Patent litigations and biosimilar approvals are critical factors influencing market entry timing.
- US FDA approval of generics could occur 5-7 years post-patent expiry, depending on patent challenges.
Market Penetration Factors:
- In markets with high HIV prevalence, government-negotiated prices or subsidy programs can influence overall sales volume.
- Healthcare providers tend to favor newer agents with lower toxicity profiles over protease inhibitors like PREZCOBIX.
Innovation and Pipeline:
- Long-acting injectables and novel delivery mechanisms threaten to replace daily oral regimens.
- These innovations could further pressure prices and market share for existing oral drugs, including PREZCOBIX.
What Is the Outlook for Stakeholders?
-
Pharmaceutical Companies:
Maximize revenues before patent expiration through strategic pricing, discounts, and expanding indications. -
Healthcare Providers:
Balance efficacy with cost; adopt new formulations or generics when they demonstrate equivalent or better outcomes. -
Payers:
Push for negotiations to lower costs, especially as patent expiry approaches, and incorporate biosimilars into formularies. -
Patients:
Access prices will decrease with generics, improving affordability, particularly in resource-limited settings.
Key Takeaways
- PREZCOBIX is a niche but steady revenue generator within the global HIV treatment market.
- Current US prices hover above USD 2,200 monthly, with no immediate discounts expected.
- Market share remains limited due to competition from integrase inhibitors and newer agents.
- Patent expiry forecasted for 2028 will precipitate significant price declines, potentially 60-70%.
- Long-term, the evolution of long-acting therapies and biosimilars will reshape the competitive landscape.
FAQs
1. When does the patent for PREZCOBIX expire?
Patent protection in the US is expected to last until 2028, after which generics may enter the market.
2. How much could prices decrease post-patent expiry?
Generic entry could reduce costs by 60-70%, bringing annual therapy prices down from approximately USD 26,000 to USD 8,000–USD 10,000.
3. What are the main competitors against PREZCOBIX in the HIV market?
Key competitors include Biktarvy, Descovy, and dolutegravir-based regimens, which have larger market shares.
4. What factors influence the future adoption of PREZCOBIX?
Patent expiration, price competition, healthcare provider preferences, and the development of new delivery methods.
5. Is there a shift toward generic versions impacting PREZCOBIX sales?
Yes, the entry of generics post-2028 is expected to significantly diminish the revenue contribution of branded PREZCOBIX.
Citations:
[1] Global HIV Treatment Market report, 2022.
[2] IQVIA, 2022 Pharmaceutical Pricing Database.
[3] Gilead Sciences Annual Report 2022.
[4] FDA Patent Expiry Database, 2023.
More… ↓
