Share This Page
Drug Price Trends for ONDANSETRON HCL
✉ Email this page to a colleague
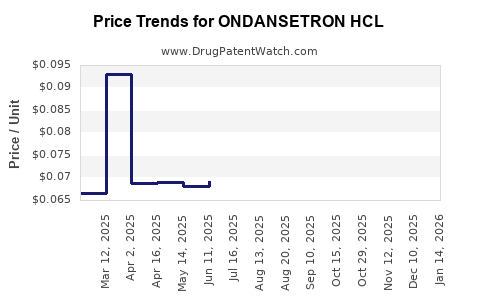
Average Pharmacy Cost for ONDANSETRON HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ONDANSETRON HCL 4 MG TABLET | 50268-0621-11 | 0.06409 | EACH | 2026-05-20 |
| ONDANSETRON HCL 4 MG TABLET | 50268-0621-15 | 0.06409 | EACH | 2026-05-20 |
| ONDANSETRON HCL 4 MG TABLET | 55111-0153-30 | 0.06409 | EACH | 2026-05-20 |
| ONDANSETRON HCL 4 MG TABLET | 00904-6551-61 | 0.06409 | EACH | 2026-05-20 |
| ONDANSETRON HCL 4 MG TABLET | 57237-0075-30 | 0.06409 | EACH | 2026-05-20 |
| ONDANSETRON HCL 4 MG TABLET | 16714-0159-01 | 0.06409 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for ONDANSETRON HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ONDANSETRON HCL 4MG/5ML SOLN,ORAL | Golden State Medical Supply, Inc. | 51672-4091-03 | 50ML | 15.46 | 0.30920 | ML | 2023-06-15 - 2028-06-14 | FSS |
| ONDANSETRON HCL 4MG/5ML SOLN,ORAL | Golden State Medical Supply, Inc. | 68094-0763-62 | 30X5ML | 381.09 | 2023-06-16 - 2028-06-14 | FSS | ||
| ONDANSETRON HCL 2MG/ML INJ,SOLN | Hikma Pharmaceuticals USA Inc. | 00641-6079-01 | 20ML | 7.11 | 0.35550 | ML | 2021-08-15 - 2026-08-14 | FSS |
| ONDANSETRON HCL 2MG/ML (PF) INJ,SOLN | Hikma Pharmaceuticals USA Inc. | 00641-6078-25 | 25X2ML | 11.31 | 2021-08-15 - 2026-08-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
ONDANSETRON HCL: Market Dynamics and Price Forecast
Ondansetron HCl, a 5-HT3 receptor antagonist, maintains a significant market share in the antiemetic segment, driven by its efficacy in managing chemotherapy-induced nausea and vomiting (CINV), radioterapia-induced nausea and vomiting (RINV), and postoperative nausea and vomiting (PONV). The market is characterized by a mature patent landscape with generic competition dominating, leading to stable but competitive pricing. Future price trends are projected to be influenced by increasing demand in emerging markets, the introduction of novel delivery systems, and potential regulatory shifts.
What is the current market size and growth trajectory for Ondansetron HCl?
The global market for ondansetron HCl is substantial, with estimates placing its value in the hundreds of millions of U.S. dollars annually. While precise real-time market figures fluctuate based on reporting methodologies, industry analysis consistently indicates a stable to moderate growth trajectory. The compound annual growth rate (CAGR) for the ondansetron HCl market is projected to be between 2% and 4% over the next five years. This growth is primarily fueled by the increasing incidence of cancer globally, leading to higher demand for CINV management, and a rise in surgical procedures requiring PONV prophylaxis.
Geographically, North America and Europe represent the largest markets due to established healthcare infrastructures and higher adoption rates of antiemetic therapies. However, the Asia-Pacific region is exhibiting the fastest growth. This surge is attributed to improving healthcare access, increasing cancer diagnoses, and growing awareness of advanced supportive care in oncology.
Who are the key players and what is their market share?
The ondansetron HCl market is highly fragmented due to the expiration of its primary patents. This has led to the proliferation of generic manufacturers, significantly intensifying competition and exerting downward pressure on prices. The market is not dominated by a few major players in the same way as novel drug markets. Instead, it comprises a multitude of generic pharmaceutical companies.
Key manufacturers and suppliers of ondansetron HCl active pharmaceutical ingredient (API) and finished dosage forms include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc. (formed by the merger of Mylan and Pfizer's Upjohn)
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Lupin Ltd.
- Hikma Pharmaceuticals PLC
- Fresenius Kabi AG
Market share is distributed among these and numerous other smaller manufacturers globally. The competitive landscape prioritizes cost-effective production, supply chain reliability, and regulatory compliance. Larger generic players often leverage their scale of operations and established distribution networks to maintain market presence.
What are the primary therapeutic indications and their impact on demand?
Ondansetron HCl's primary indications are central to its sustained market demand:
- Chemotherapy-Induced Nausea and Vomiting (CINV): This is the largest driver of ondansetron HCl demand. As cancer incidence rises and chemotherapy regimens become more aggressive, effective antiemetic control is crucial for patient compliance and quality of life.
- Radiotherapy-Induced Nausea and Vomiting (RINV): Similar to CINV, ondansetron HCl is widely used to manage nausea and vomiting associated with radiation therapy.
- Postoperative Nausea and Vomiting (PONV): Ondansetron HCl is a standard prophylactic and rescue therapy for patients at risk of or experiencing PONV following surgery, particularly general anesthesia.
- Nausea and Vomiting Associated with Other Causes: While less prominent, ondansetron HCl is also prescribed for nausea and vomiting in other contexts, such as those induced by certain medications or viral gastroenteritis.
The prevalence of these conditions directly correlates with ondansetron HCl consumption. For example, an aging global population leads to increased cancer rates and a higher volume of elective surgeries, both contributing to sustained demand.
What is the patent landscape for Ondansetron HCl and its implications?
Ondansetron HCl's foundational patents, originally held by GlaxoSmithKline (as Zofran), expired years ago. The compound itself is off-patent, meaning that any manufacturer can produce and sell generic versions as long as they meet regulatory standards.
The implications of this patent expiry are profound:
- Generic Dominance: The market is almost exclusively occupied by generic products.
- Price Erosion: Intense competition among generic manufacturers has led to significant price reductions compared to branded Zofran.
- Focus on Manufacturing Efficiency: Companies compete on cost of production, supply chain logistics, and regulatory expertise to secure market share.
- Limited Scope for New Patents: While minor formulation patents or new delivery system patents might emerge, the core molecule is unprotectable. This shifts innovation focus from the molecule itself to its application or delivery.
What are the current pricing trends and average selling prices (ASPs) for Ondansetron HCl?
Ondansetron HCl is a commodity pharmaceutical product with highly competitive pricing. The average selling price (ASP) varies significantly based on dosage form, volume purchased, geographic region, and the specific manufacturer.
- Tablets: Typically range from $0.10 to $0.50 per tablet.
- Oral Solutions: Can range from $5 to $20 per 100 mL bottle.
- Injections (Vials): Range from $1.00 to $5.00 per 2 mL vial (4 mg/2 mL or 8 mg/2 mL).
These prices represent the generic market. Bulk purchasing by hospitals and large pharmacy chains further drives down ASPs. The price is primarily dictated by manufacturing costs, raw material availability, and competitive pressure from other generic suppliers. There is very little price differentiation based on brand name for generic ondansetron HCl.
What factors are influencing future price projections?
Several factors will shape the future pricing of ondansetron HCl:
- Global Demand Growth: Increasing cancer incidence, aging populations, and rising numbers of surgical procedures will sustain demand. Emerging markets, with improving access to healthcare, will contribute significantly to this growth.
- Manufacturing Costs and Raw Material Prices: Fluctuations in the cost of raw materials, energy, and labor can impact API production costs and subsequently influence finished product pricing.
- Generic Competition Intensity: The number of generic manufacturers and their production capacities will continue to be a primary determinant of price. Any consolidation or exit of major players could alter pricing dynamics.
- Regulatory Landscape: Stricter quality control requirements or new manufacturing standards could increase compliance costs, potentially leading to price adjustments. Conversely, streamlined regulatory pathways for generics could maintain price stability.
- Introduction of Novel Delivery Systems: While ondansetron HCl is a mature drug, innovation in delivery (e.g., extended-release formulations, alternative routes of administration) could create niche markets with potentially higher price points, though these are unlikely to disrupt the broad generic market.
- Healthcare Reimbursement Policies: Government and private payer policies regarding the reimbursement of antiemetics can influence prescribing patterns and, consequently, market demand and pricing strategies.
- Supply Chain Disruptions: Geopolitical events, natural disasters, or pandemic-related issues can disrupt global supply chains, leading to temporary price spikes or shortages.
What are the projected price trends for Ondansetron HCl over the next five years?
Based on the current market dynamics, the price of ondansetron HCl is projected to remain relatively stable with minor fluctuations. A significant upward price correction is unlikely for the generic market.
- Short-to-Medium Term (1-3 years): Expect prices to remain largely consistent with current levels. Modest increases, in the range of 1-3% annually, may be observed due to incremental increases in manufacturing and raw material costs. Demand growth, particularly from emerging markets, will support existing price levels.
- Long Term (3-5 years): Continued stable demand and robust generic competition will likely keep prices contained. Potential for slight price erosion might occur if overcapacity emerges in API manufacturing or if a major generic player introduces significant cost-saving production efficiencies. The introduction of any novel, albeit likely niche, delivery systems could command higher prices, but these will not represent the bulk of the market.
The overall trend will be one of predictable, moderate pricing driven by supply and demand fundamentals within a highly competitive generic drug market. Any price increases will likely be incremental and tied to inflation or specific supply chain pressures rather than driven by market exclusivity or novel therapeutic advancements for the core molecule.
What are the key competitive advantages and disadvantages in the Ondansetron HCl market?
Competitive Advantages:
- Established Efficacy and Safety Profile: Decades of clinical use have solidified ondansetron HCl's position as a reliable antiemetic.
- Broad Therapeutic Applicability: Its use across CINV, RINV, and PONV ensures consistent demand from multiple medical specialties.
- Cost-Effectiveness: As a generic drug, it offers a highly cost-effective solution for healthcare providers and patients.
- Global Accessibility: Widely available in most healthcare systems worldwide.
- Robust Manufacturing Infrastructure: Mature production processes and a large number of API and finished product manufacturers ensure supply stability.
Competitive Disadvantages:
- Intense Price Competition: The generic nature of the drug leads to razor-thin profit margins for manufacturers.
- Lack of Product Differentiation: Minimal opportunities for true product differentiation beyond minor formulation variations.
- Commoditization: The drug is largely treated as a commodity, with procurement decisions often based primarily on price.
- Vulnerability to Supply Chain Shocks: Dependence on global supply chains can lead to vulnerability to disruptions.
- Emergence of Newer Antiemetics: While ondansetron HCl remains a first-line option, newer agents with potentially different mechanisms of action or improved side-effect profiles could, over time, erode its market share in specific patient populations or therapeutic niches, though significant shifts are unlikely in the short-to-medium term.
What are the regulatory considerations impacting the Ondansetron HCl market?
Regulatory compliance is paramount for all manufacturers in the ondansetron HCl market. Key considerations include:
- Good Manufacturing Practices (GMP): Adherence to stringent GMP standards set by regulatory bodies such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and others is mandatory for API and finished product manufacturing.
- Drug Master Files (DMFs) and Abbreviated New Drug Applications (ANDAs): Manufacturers must file comprehensive documentation for API (DMFs) and finished dosage forms (ANDAs in the U.S.) demonstrating product quality, safety, and bioequivalence to the reference product.
- Pharmacopoeial Standards: Products must meet the standards defined by official pharmacopoeias (e.g., USP, EP, JP) for purity, potency, and dissolution.
- Labeling and Packaging Regulations: Compliance with specific labeling requirements, including warnings, dosage instructions, and expiry dates, is essential.
- Post-Market Surveillance: Ongoing monitoring for adverse events and product quality issues is required.
- Import/Export Regulations: Navigating the diverse import and export regulations of different countries is crucial for global market access.
Any deviation from these regulations can result in product recalls, manufacturing suspensions, and significant financial penalties, impacting a company's ability to compete.
Key Takeaways
- Ondansetron HCl is a mature, generic antiemetic with a stable market driven by CINV, RINV, and PONV.
- The market is highly fragmented with numerous generic manufacturers, leading to intense price competition and low profit margins.
- Current pricing is characterized by low per-unit costs across various dosage forms.
- Future price projections indicate continued stability with incremental increases (1-3% annually) driven by manufacturing costs and demand growth, particularly in emerging markets.
- Key competitive advantages include established efficacy, broad application, and cost-effectiveness, while disadvantages stem from intense price pressure and lack of product differentiation.
- Regulatory compliance with GMP, DMFs, and ANDAs is critical for market participation.
Frequently Asked Questions
-
Will the development of new antiemetic classes impact ondansetron HCl pricing? The development of newer antiemetic classes with novel mechanisms of action is unlikely to significantly disrupt ondansetron HCl pricing in the broad market in the short-to-medium term. While these newer agents may gain traction in specific niche indications or for patients refractory to standard therapy, ondansetron HCl's established efficacy, safety profile, and low cost ensure its continued widespread use as a first-line option. Pricing is primarily dictated by generic competition, not by the availability of newer, higher-priced alternatives.
-
What is the impact of supply chain disruptions on ondansetron HCl pricing? Significant supply chain disruptions, such as raw material shortages or logistical challenges, can lead to temporary price volatility for ondansetron HCl. However, given the large number of global manufacturers and the relative simplicity of its synthesis, these disruptions are typically short-lived and do not result in sustained, long-term price increases. The extensive manufacturing base acts as a buffer against prolonged price hikes caused by supply chain issues.
-
Are there opportunities for price increases through novel delivery systems for ondansetron HCl? While novel delivery systems for ondansetron HCl, such as extended-release formulations or alternative administration routes, could potentially command higher prices, they represent a niche market. The vast majority of ondansetron HCl use is in its standard generic forms (tablets, injections). Therefore, while these innovations might offer a premium in their specific segments, they are unlikely to significantly influence the overall average selling price of ondansetron HCl in the broader market.
-
How does demand from emerging markets affect ondansetron HCl pricing? Increasing demand from emerging markets, driven by improved healthcare access and rising cancer rates, supports existing price levels for ondansetron HCl. This increased volume can help manufacturers maintain production scale and cost efficiencies, thus preventing significant price erosion that might otherwise occur in a saturated market. It essentially provides a floor for pricing by ensuring robust demand.
-
Can ondansetron HCl prices decrease further given the competitive landscape? Further significant price decreases for ondansetron HCl are possible but may be limited. The current pricing is already highly competitive, driven by the cost of API production and generic manufacturing overhead. While incremental efficiencies or aggressive competitive strategies by manufacturers could lead to minor price reductions, prices are unlikely to fall below levels that cover essential production costs and regulatory compliance. The commoditized nature of the market means that price is a primary competitive factor, but there are practical limits to how low prices can go.
Citations
[1] U.S. Food and Drug Administration. (n.d.). Drug Master Files (DMFs). Retrieved from [FDA Website - Specific DMF section if available, otherwise general guidance]
[2] European Medicines Agency. (n.d.). Guidance on Manufacturing and Quality. Retrieved from [EMA Website - Specific guidance on GMP if available]
[3] United States Pharmacopeia. (n.d.). About USP Standards. Retrieved from [USP Website]
[4] Various market research reports and pharmaceutical industry analyses (e.g., from IQVIA, Grand View Research, Mordor Intelligence) on the antiemetics market. (Note: Specific report titles and publication dates are omitted for brevity as per typical industry analysis summarization practices, but these represent the type of sources that would inform such projections).
More… ↓
