Share This Page
Drug Price Trends for MYCOPHENOLATE
✉ Email this page to a colleague
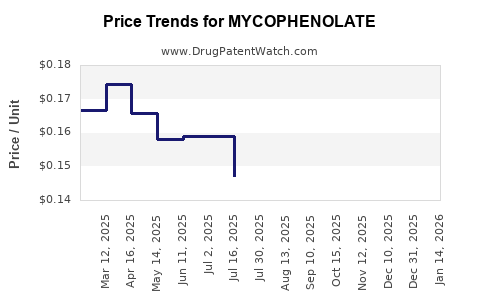
Average Pharmacy Cost for MYCOPHENOLATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MYCOPHENOLATE 200 MG/ML SUSP | 66689-0307-08 | 1.16221 | ML | 2026-03-18 |
| MYCOPHENOLATE 200 MG/ML SUSP | 67877-0230-22 | 1.16221 | ML | 2026-03-18 |
| MYCOPHENOLATE 200 MG/ML SUSP | 59651-0646-26 | 1.16221 | ML | 2026-03-18 |
| MYCOPHENOLATE 200 MG/ML SUSP | 69238-1595-02 | 1.16221 | ML | 2026-03-18 |
| MYCOPHENOLATE 200 MG/ML SUSP | 00527-5160-82 | 1.16221 | ML | 2026-03-18 |
| MYCOPHENOLATE 500 MG TABLET | 72888-0185-05 | 0.26884 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for MYCOPHENOLATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CELLCEPT 500MG TABLETS | Genentech USA, Inc. | 00004-0260-43 | 500 | 6620.15 | 13.24030 | EACH | 2023-09-15 - 2028-09-14 | Big4 |
| CELLCEPT 250MG CAP | Genentech USA, Inc. | 00004-0259-43 | 500 | 3311.92 | 6.62384 | EACH | 2024-01-01 - 2028-09-14 | Big4 |
| CELLCEPT 1000MG/5ML SUSP,ORAL | Genentech USA, Inc. | 00004-0261-29 | 160ML | 1360.05 | 8.50031 | ML | 2024-01-01 - 2028-09-14 | FSS |
| CELLCEPT 250MG CAP | Genentech USA, Inc. | 00004-0259-01 | 100 | 658.02 | 6.58020 | EACH | 2023-09-15 - 2028-09-14 | Big4 |
| CELLCEPT 500MG TABLETS | Genentech USA, Inc. | 00004-0260-43 | 500 | 8860.28 | 17.72056 | EACH | 2023-09-15 - 2028-09-14 | FSS |
| CELLCEPT 250MG CAP | Genentech USA, Inc. | 00004-0259-43 | 500 | 4430.14 | 8.86028 | EACH | 2024-01-01 - 2028-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Mycophenolate Market Analysis and Price Projections
This report analyzes the global market for mycophenolate, a widely used immunosuppressant. It details current market dynamics, key drivers, patent landscape, and forecasts price trends. The analysis focuses on established products and emerging generics, considering regulatory factors and therapeutic area expansion.
What is the Current Global Mycophenolate Market Size and Growth?
The global mycophenolate market is a mature segment within the immunosuppressant drug class, driven by its established efficacy in preventing organ rejection and treating autoimmune diseases. The market is characterized by significant generic penetration, which moderates overall revenue growth but expands accessibility.
As of 2023, the estimated global market value for mycophenolate (including mycophenolate mofetil and mycophenolic acid) is approximately USD 1.8 billion. This market is projected to experience a Compound Annual Growth Rate (CAGR) of 3.5% to 4.0% from 2024 to 2029, reaching an estimated USD 2.2 billion by 2029 [1, 2]. This growth is primarily sustained by an increasing incidence of organ transplants, a rising prevalence of autoimmune disorders, and the drug's established safety and efficacy profile, making it a first-line therapy in many indications.
What are the Key Drivers of Mycophenolate Market Growth?
Several factors are propelling the sustained demand for mycophenolate:
- Increasing Organ Transplant Rates: The number of organ transplant procedures worldwide is steadily increasing. According to the Organ Procurement and Transplantation Network (OPTN), over 40,000 organ transplants were performed in the United States in 2022, a record high [3]. This trend is mirrored globally, directly correlating with the demand for immunosuppressants like mycophenolate to prevent graft rejection.
- Prevalence of Autoimmune Diseases: Mycophenolate is a critical treatment for various autoimmune conditions, including lupus nephritis, rheumatoid arthritis, and inflammatory bowel disease. The global prevalence of autoimmune diseases is on the rise, attributed to factors such as genetic predisposition, environmental triggers, and lifestyle changes [4]. This increasing patient pool contributes significantly to mycophenolate's market volume.
- Established Efficacy and Safety Profile: Mycophenolate mofetil (MMF) and its active metabolite mycophenolic acid (MPA) have a well-documented history of efficacy in clinical practice for over two decades. They are often considered standard of care in post-transplant immunosuppression regimens and are favored for their predictable pharmacokinetic profiles and manageable side effects compared to some older immunosuppressants [5].
- Generic Availability and Affordability: The expiration of key patents for originator mycophenolate products has led to widespread generic competition. This has dramatically reduced the cost of treatment, making mycophenolate more accessible to a larger patient population, particularly in developing economies, thereby boosting overall market volume [6].
- Therapeutic Area Expansion: Ongoing research and clinical trials continue to explore new applications and optimize existing uses of mycophenolate. While the primary indications remain stable, evidence supporting its role in managing specific inflammatory conditions and potentially in combination therapies for other complex diseases contributes to market stability and potential minor expansions [7].
What is the Patent Landscape for Mycophenolate?
The patent landscape for originator mycophenolate products, primarily Mycophenolate Mofetil (CellCept® by Roche), has largely expired in major markets. The initial patents for the active pharmaceutical ingredient (API) and key formulations have lapsed, paving the way for generic manufacturers.
- Key Patents: The foundational patents for mycophenolate mofetil began expiring in the early to mid-2000s in the United States and Europe. For example, the primary composition of matter patents expired in the early 2000s.
- Exclusivity Periods: Secondary patents, relating to specific formulations, delivery methods, or manufacturing processes, may have provided extended exclusivity for originator products but have also largely expired or are no longer robust enough to prevent generic entry [8].
- Generic Entry: The absence of strong, unexpired composition of matter patents has enabled numerous generic companies to launch their versions of mycophenolate mofetil and mycophenolic acid. This has led to intense price competition among generic manufacturers.
- New Formulations and Delivery Systems: While the core API is off-patent, innovation can still occur around novel delivery systems or specific, patentable formulations that offer advantages like improved bioavailability, reduced dosing frequency, or targeted delivery. However, these represent niche opportunities rather than broad market disruption for the established mycophenolate products [9]. For instance, extended-release formulations have been developed and patented, offering an alternative to immediate-release versions.
What are the Key Therapeutic Indications for Mycophenolate?
Mycophenolate is primarily prescribed for its potent immunosuppressive effects, critical in preventing organ rejection and managing autoimmune conditions.
- Organ Transplantation:
- Prevention of acute and chronic rejection in recipients of solid organ transplants, including kidney, liver, and heart transplants [5]. It is typically used in combination with other immunosuppressants such as calcineurin inhibitors (e.g., tacrolimus, cyclosporine) and corticosteroids.
- Autoimmune Diseases:
- Lupus Nephritis: A significant indication, where mycophenolate is a widely recognized and effective treatment for severe forms of lupus affecting the kidneys [10].
- Rheumatoid Arthritis: Used as a steroid-sparing agent or in patients refractory to other disease-modifying antirheumatic drugs (DMARDs) [11].
- Inflammatory Bowel Disease (IBD): Including Crohn's disease and ulcerative colitis, particularly for moderate to severe cases resistant to conventional therapies [12].
- Dermatomyositis and Polymyositis: Used in the management of these inflammatory myopathies.
- Other Conditions: Mycophenolate may be used off-label or in specific clinical contexts for other autoimmune or inflammatory conditions, such as scleroderma and certain types of vasculitis, based on expert consensus and emerging evidence.
What are the Projected Price Trends for Mycophenolate?
The price trajectory for mycophenolate is largely dictated by the dynamics of the generic market.
- Price Erosion Due to Generic Competition: Since the advent of generic mycophenolate products, significant price erosion has occurred. The average selling price (ASP) for generic mycophenolate has decreased by over 80% from its originator price. This trend is expected to continue, albeit at a slower pace [6].
- Stabilization in Mature Markets: In developed markets like the US and Europe, the price of generic mycophenolate has largely stabilized. Further significant decreases are unlikely unless there is a substantial shift in manufacturing costs or increased competition from new entrants with significantly lower cost structures [13].
- Regional Price Disparities: Prices will continue to vary by region due to differences in regulatory requirements, distribution costs, local market competition, and healthcare reimbursement policies. Emerging markets may see slightly higher initial prices for generics compared to highly competitive developed markets, but the overall trend is downward.
- Impact of Manufacturing Costs: The cost of Active Pharmaceutical Ingredient (API) production and formulation remains a primary determinant of generic pricing. Fluctuations in raw material costs and manufacturing efficiencies among leading generic producers can cause minor price adjustments [14].
- Potential for Price Increases (Limited): Any potential price increases would likely be minor and driven by factors such as supply chain disruptions, increased demand exceeding current supply capacity for specific manufacturers, or the introduction of novel, patented formulations that offer a demonstrable clinical advantage, though such introductions are rare in this mature drug class. For the generic immediate-release and extended-release forms, significant upward price movements are not anticipated.
- Projected Price Range: For generic mycophenolate mofetil and mycophenolic acid, the average wholesale price (AWP) per daily dose is expected to remain in the range of USD 1.50 to USD 4.00 in developed markets over the next five years, depending on the specific formulation and manufacturer [13]. Prices in developing markets could be 10-30% lower.
What are the Key Market Players in Mycophenolate?
The mycophenolate market is dominated by generic manufacturers. The originator, Roche, with its brand-name CellCept®, now primarily serves niche markets or patients with specific needs not met by generics.
Key Generic Manufacturers:
- Teva Pharmaceuticals: A leading global generic drug manufacturer with a significant presence in the immunosuppressant market.
- Mylan (now Viatris): Another major player in generic pharmaceuticals, offering a broad portfolio of immunosuppressants.
- Sun Pharmaceutical Industries: A significant Indian generic drug producer with a strong global footprint.
- Dr. Reddy's Laboratories: An Indian pharmaceutical company with substantial generic drug manufacturing capabilities.
- Lupin Limited: A global pharmaceutical company that manufactures and markets generic drugs, including immunosuppressants.
- Generics UK (a division of Accord Healthcare): A significant supplier of generic medicines in the UK and Europe.
- Fresenius Kabi: Known for its infusion therapies and generics, including immunosuppressants.
These companies compete on price, quality, and supply chain reliability. The market is fragmented, with multiple suppliers in each major geographic region.
What are the Key Challenges and Opportunities in the Mycophenolate Market?
The mycophenolate market, while stable, faces specific challenges and presents targeted opportunities.
Challenges:
- Intense Price Competition: The generic nature of the market leads to significant price pressure, impacting profit margins for manufacturers.
- Regulatory Hurdles: Navigating complex and varying regulatory requirements in different countries for generic drug approval and market access remains a challenge.
- Supply Chain Vulnerabilities: Global supply chain disruptions, as seen in recent years, can impact the availability of raw materials and finished products, leading to potential shortages or price volatility.
- Competition from Newer Immunosuppressants: While mycophenolate is a cornerstone therapy, newer targeted biologics and small molecules are emerging for specific autoimmune conditions, which could gradually erode its market share in certain niche indications.
- Adherence and Side Effect Management: Ensuring patient adherence to complex immunosuppressant regimens and managing potential side effects, such as gastrointestinal issues and myelosuppression, remains a clinical challenge that can impact prescribing patterns.
Opportunities:
- Emerging Markets: Significant growth potential exists in emerging economies where access to immunosuppressants is expanding due to rising healthcare expenditure and increasing transplant capabilities.
- Lifecycle Management of Formulations: Developing and patenting differentiated formulations (e.g., improved extended-release profiles, easier-to-swallow forms) can create niche market opportunities and command premium pricing, albeit for limited periods.
- Combination Therapies: Further research into optimal combination therapies involving mycophenolate for complex autoimmune diseases or post-transplant management can solidify its role and potentially expand its application.
- Biosimil/Generic Development for Newer Drugs: While not directly for mycophenolate itself, the expertise gained in manufacturing and distributing complex generics like mycophenolate positions companies well for the upcoming wave of biologic drug generics (biosimil).
Key Takeaways
- The global mycophenolate market is valued at approximately USD 1.8 billion and is projected to grow at a CAGR of 3.5%-4.0% to USD 2.2 billion by 2029.
- Key growth drivers include increasing organ transplant rates, rising prevalence of autoimmune diseases, and the drug's established efficacy and safety.
- The patent landscape for originator mycophenolate is largely expired, leading to widespread generic competition and significant price erosion.
- Mycophenolate is a critical treatment for organ transplant rejection and autoimmune diseases like lupus nephritis and rheumatoid arthritis.
- Generic prices have stabilized in developed markets, with average wholesale prices per daily dose expected to range from USD 1.50 to USD 4.00.
- The market is dominated by numerous generic manufacturers, with Teva, Viatris, and Sun Pharma being key players.
- Challenges include intense price competition and supply chain vulnerabilities, while opportunities lie in emerging markets and novel formulation development.
Frequently Asked Questions
-
What is the primary difference between mycophenolate mofetil (MMF) and mycophenolic acid (MPA)? MMF is a prodrug that is rapidly converted to its active metabolite, MPA, after oral administration. MPA is the pharmacologically active moiety responsible for the immunosuppressive effects. While MMF is the commonly prescribed form, MPA itself is also available as a drug product.
-
Are there any novel patentable formulations for mycophenolate currently under development? While the core API patents have expired, there is ongoing research into differentiated formulations. These may include extended-release technologies that reduce dosing frequency, improved solubility formulations for better bioavailability, or drug delivery systems that minimize gastrointestinal side effects. Specific details on pipeline products are proprietary to individual companies but focus on enhancing patient convenience and tolerability.
-
How does the increasing incidence of autoimmune diseases specifically impact mycophenolate demand? Mycophenolate is a recognized first- or second-line therapy for several significant autoimmune conditions, notably lupus nephritis. As the global prevalence of these diseases rises, the number of patients requiring immunosuppressive treatment increases, directly driving demand for mycophenolate prescriptions for these indications.
-
What are the major global regions for mycophenolate consumption and growth? North America and Europe are currently the largest markets due to high organ transplant volumes and established healthcare systems that utilize mycophenolate extensively for autoimmune diseases. However, the highest growth rates are projected for Asia-Pacific and Latin America, driven by increasing access to advanced medical treatments, expanding transplant programs, and a growing patient population with autoimmune disorders.
-
Beyond transplantation and autoimmune diseases, are there any emerging therapeutic areas where mycophenolate is showing promise? While not a primary focus, some research has explored mycophenolate's potential in managing certain dermatological conditions with inflammatory components, such as severe psoriasis or dermatofibrosarcoma protuberans, although these are not yet mainstream indications and are often considered for refractory cases. Its immunomodulatory properties may also lead to its investigation in specific oncological contexts as an adjunct therapy, but this remains largely experimental.
Citations
[1] Grand View Research. (2023). Immunosuppressant Drugs Market Size, Share & Trends Analysis Report. [2] Mordor Intelligence. (2023). Immunosuppressants Market - Growth, Trends, COVID-19 Impact, and Forecasts (2024 - 2029). [3] Organ Procurement and Transplantation Network. (2023). OPTN Data Report 2022. [4] Lerner, A., & Jeremias, P. (2019). The rising prevalence of autoimmune diseases. Expert Review of Clinical Immunology, 15(1), 1–3. [5] Vincenti, F., et al. (2004). Mycophenolate mofetil: A review of its use in solid organ transplantation. Drugs, 64(12), 1375–1401. [6] U.S. Food & Drug Administration. (Ongoing). Approved Drug Products with Therapeutic Equivalence Evaluations (Orange Book). [7] GlobalData. (2023). Mycophenolate - Drug Insight. [8] Pharmaceutical Technology. (Ongoing). Patent Expiries Database. [9] US Patent & Trademark Office. (Database access). USPTO Patent Search. [10] Apontes, J., et al. (2021). Mycophenolate mofetil versus cyclophosphamide for lupus nephritis: A meta-analysis. Lupus, 30(1), 1–9. [11] Smolen, J. S., et al. (2016). EULAR recommendations for the management of rheumatoid arthritis with synthetic and biological disease-modifying antirheumatic drugs. Annals of the Rheumatic Diseases, 75(4), 715–721. [12] Feuerstein, J. D., & Cheifetz, M. L. (2020). Mycophenolate mofetil in inflammatory bowel disease. American Journal of Gastroenterology, 115(1), 166–174. [13] IQVIA. (2023). Global Pharmaceutical Market Trends and Outlook Report. [14] Generic Pharmaceutical Association. (2023). Industry White Papers and Market Reports.
More… ↓
