Share This Page
Drug Price Trends for METFORMIN ER
✉ Email this page to a colleague
Average Pharmacy Cost for METFORMIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| METFORMIN ER 1,000 MG GASTR-TB | 50228-0446-90 | 0.41915 | EACH | 2026-03-18 |
| METFORMIN ER 1,000 MG GASTR-TB | 27241-0241-90 | 0.41915 | EACH | 2026-03-18 |
| METFORMIN ER 1,000 MG GASTR-TB | 60505-4701-09 | 0.41915 | EACH | 2026-03-18 |
| METFORMIN ER 1,000 MG GASTR-TB | 68180-0339-09 | 0.41915 | EACH | 2026-03-18 |
| METFORMIN ER 1,000 MG GASTR-TB | 42571-0334-90 | 0.41915 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Metformin ER Market Analysis and Price Projections
Metformin extended-release (ER) is a cornerstone therapy for type 2 diabetes. Its market is characterized by broad accessibility, established efficacy, and significant price competition, particularly with the advent of generic versions. Future price trends are expected to remain largely stable, influenced by generic market dynamics, payer negotiations, and limited innovation in novel formulations.
What is the Current Market Landscape for Metformin ER?
The global metformin ER market is mature and highly competitive. The drug remains a first-line treatment option for a large patient population diagnosed with type 2 diabetes.
Key Market Segments and Drivers
- Geographic Distribution: North America and Europe represent the largest markets due to high diabetes prevalence and advanced healthcare infrastructure. Emerging markets in Asia-Pacific and Latin America are experiencing growth driven by increasing diagnosis rates and improving healthcare access.
- Patient Demographics: Metformin ER is prescribed across a wide age spectrum of adults with type 2 diabetes. Its favorable safety profile makes it suitable for many patients, including those with kidney impairment, where other agents might be contraindicated.
- Therapeutic Positioning: Metformin ER is predominantly used as a monotherapy or in combination with other oral antidiabetic agents or injectable therapies. Its effectiveness in lowering A1c levels and its weight-neutral or modest weight-loss effect contribute to its sustained use.
- Competitive Landscape: The market features numerous manufacturers offering generic metformin ER. Branded formulations, such as Glucophage XR (Bristol-Myers Squibb) and Fortamet (Astellas Pharma), maintain a presence but face significant price pressure from generics. Biosimilar development is not applicable to small molecule drugs like metformin.
Market Size and Growth
Estimates for the global metformin ER market size vary but generally place it in the multi-billion dollar range. For instance, the broader metformin market (including immediate-release) was valued at approximately USD 1.2 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of around 4.5% through 2030, with ER formulations comprising a significant portion of this value. [1] This growth is primarily driven by an increasing global diabetes prevalence. The World Health Organization (WHO) estimates that over 422 million adults worldwide have diabetes, with type 2 diabetes accounting for approximately 90% of cases. [2]
What are the Patent and Exclusivity Statuses Affecting Metformin ER?
Metformin hydrochloride itself is an old drug, with its initial patents long expired. However, extended-release formulations have been developed to improve patient adherence and gastrointestinal tolerability, and these specific formulations may have had patent protection.
Key Exclusivity Periods
- Metformin Hydrochloride: The original patents for metformin hydrochloride expired decades ago. It is a well-established, off-patent molecule.
- Extended-Release Formulations: Patents for specific extended-release technologies and formulations have historically provided market exclusivity for branded products. For example, patents related to sustained-release matrix compositions and osmotic release systems have been critical. However, many of these foundational ER patents have also expired.
- Recent and Ongoing Filings: While major patent exclusivities have lapsed for most leading metformin ER products, manufacturers may still hold patents on specific manufacturing processes, novel excipients, or combinations with other drugs. The U.S. Food and Drug Administration (FDA) Orange Book lists approved drug products and their patent information. For Glucophage XR (metformin hydrochloride extended-release), the earliest listed patents are from the late 1990s and early 2000s, with many now expired. [3]
- Generic Entry: The expiration of key formulation patents has led to widespread generic competition. This influx of generic manufacturers has been the primary driver of price reduction for metformin ER.
What are the Pricing Dynamics of Metformin ER?
The pricing of metformin ER is characterized by significant variability, heavily influenced by its generic status and the competitive pressures within the market.
Factors Influencing Price
- Generic Competition: With numerous generic manufacturers producing metformin ER, prices are driven down to very low levels, especially in developed markets. This competition is intense, with many companies vying for market share.
- Payer Contracts and Rebates: Large pharmacy benefit managers (PBMs) and government payers negotiate significant rebates from drug manufacturers. These behind-the-scenes discounts make the net price paid by payers substantially lower than the list price. Metformin ER is often placed on formularies with minimal or no co-pays for patients.
- Wholesale Acquisition Cost (WAC) vs. Net Price: The WAC is a list price that bears little resemblance to the actual transaction price. The net price, after rebates and discounts, is what manufacturers ultimately receive. For metformin ER, WACs can range from USD 0.10 to USD 1.00 per tablet (e.g., 500mg or 1000mg), but net prices are often significantly lower, sometimes pennies per dose. [4]
- Dosage and Formulation: While less pronounced than in novel drugs, slight price variations can exist between different tablet strengths (e.g., 500mg, 750mg, 1000mg) and potentially minor differences in ER technologies if patents still afford some distinction.
- Supply Chain and Distribution: Costs associated with manufacturing, packaging, distribution, and regulatory compliance also factor into pricing, though economies of scale for high-volume generics like metformin ER minimize these per-unit impacts.
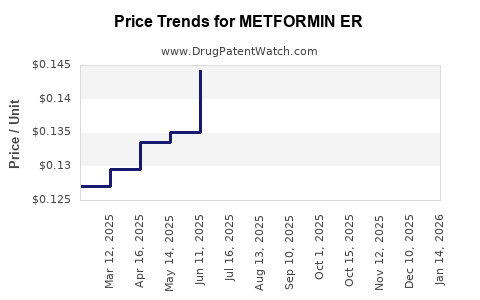
Historical Price Trends
Over the past decade, the average selling price (ASP) of metformin ER has steadily declined due to the aforementioned factors. The market has matured to a point where significant price increases are unlikely unless there are major shifts in manufacturing costs or a sudden consolidation of generic suppliers.
What are the Future Price Projections for Metformin ER?
Future price projections for metformin ER indicate continued stability, with minimal upward pressure and a high likelihood of remaining at low price points.
Projected Price Trajectory
- Continued Generic Dominance: The presence of a large number of generic manufacturers will sustain price competition. This is unlikely to change unless there is a significant consolidation of the market or an unforeseen increase in manufacturing costs.
- Stable Demand: The persistent and growing prevalence of type 2 diabetes ensures a consistent demand for metformin ER. This stable demand underpins the market for generic competition.
- Limited Innovation: While incremental improvements in drug delivery or combinations might emerge, transformative innovations that would command premium pricing are improbable for metformin ER itself. The focus for innovation in diabetes therapy has shifted to newer classes of drugs with distinct mechanisms of action (e.g., GLP-1 receptor agonists, SGLT2 inhibitors).
- Payer Influence: PBMs and payers will continue to exert downward pressure on prices through contract negotiations and formulary management, prioritizing cost-effective treatments.
- Potential for Minor Fluctuations: While the overall trend is stable, minor price fluctuations may occur due to raw material costs, manufacturing capacity adjustments, or individual manufacturer strategies. However, these are expected to be within a narrow band.
Quantifiable Projections
Based on current market dynamics and the historical performance of mature generic drugs, metformin ER prices are projected to remain relatively flat over the next five to seven years. A modest annual decline of 0.5% to 2.0% in real terms might be observed due to ongoing competitive pressures and efficiency gains in manufacturing and distribution. Wholesale prices for commonly prescribed strengths (e.g., 500mg, 1000mg extended-release) are expected to remain in the range of USD 0.10 to USD 0.50 per tablet. [4] The net price to payers will likely be even lower, potentially in the single-digit cents per dose.
What are the Regulatory Considerations for Metformin ER?
Regulatory bodies play a role in ensuring the quality, safety, and efficacy of metformin ER, influencing manufacturing standards and market access.
Key Regulatory Aspects
- FDA Approval and Monitoring: Metformin ER products in the U.S. must receive FDA approval, demonstrating bioequivalence to the reference listed drug for generics. The FDA also monitors for product quality, impurities (e.g., nitrosamine contamination concerns that led to recalls of some metformin products in the past), and adverse events. [5]
- European Medicines Agency (EMA) Oversight: In Europe, the EMA and national competent authorities regulate metformin ER, ensuring compliance with European Pharmacopoeia standards and good manufacturing practices.
- Good Manufacturing Practices (GMP): Manufacturers are subject to strict GMP regulations, which govern all aspects of production and quality control. Compliance is regularly audited by regulatory agencies.
- Generic Drug Applications (ANDA/MAA): Generic manufacturers must submit Abbreviated New Drug Applications (ANDAs) in the U.S. or Marketing Authorisation Applications (MAAs) in Europe, proving that their product is the same as the approved reference product in terms of active ingredient, dosage form, strength, route of administration, quality, performance characteristics, and intended use.
- Post-Market Surveillance: Regulatory agencies conduct post-market surveillance to monitor the safety and effectiveness of approved drugs. This can lead to label changes, warnings, or even market withdrawals if safety issues arise.
Key Takeaways
- Metformin ER is a mature, highly competitive market dominated by generic manufacturers.
- Original patents for metformin have long expired, and key extended-release formulation patents have also lapsed.
- Pricing is driven by intense generic competition and significant payer negotiations, resulting in very low net prices.
- Future price projections indicate continued stability, with minimal upward movement and ongoing downward pressure.
- Regulatory oversight focuses on quality, safety, and bioequivalence, ensuring market access for approved generic products.
Frequently Asked Questions
How does the price of generic metformin ER compare to branded versions?
Generic metformin ER is substantially less expensive than branded versions. The availability of multiple generic manufacturers has driven prices down to a fraction of the cost of branded products.
What is the typical price range for a 30-day supply of metformin ER?
For patients with insurance or access to discount programs, a 30-day supply of metformin ER often has a co-pay of $0 to $10. The wholesale acquisition cost per unit is very low, often in the range of $0.10 to $0.50 per tablet.
Are there any upcoming patent expirations that could impact metformin ER pricing?
As metformin ER is a generic drug with no active patent protection for its core formulation, no significant future patent expirations are expected to directly impact its pricing. The market dynamics are already defined by its off-patent status.
What factors could potentially increase the price of metformin ER?
Significant increases in metformin ER prices are unlikely. Potential, though improbable, factors could include widespread manufacturing disruptions leading to severe supply shortages, a dramatic consolidation of generic manufacturers, or a substantial increase in the cost of raw materials or active pharmaceutical ingredients.
How do payer rebates affect the net price of metformin ER?
Payer rebates are substantial and drive the net price of metformin ER far below its list price. These negotiations between manufacturers and payers significantly reduce the effective cost for insurance providers and, consequently, for patients through lower co-pays or deductibles.
How is the quality of generic metformin ER ensured?
The quality of generic metformin ER is ensured through stringent regulatory oversight by agencies like the FDA and EMA. Manufacturers must adhere to Good Manufacturing Practices (GMP) and demonstrate bioequivalence to the reference product through rigorous testing before approval. Post-market surveillance also monitors for quality and safety.
Citations
[1] Grand View Research. (2023). Metformin Market Size, Share & Trends Analysis Report. [2] World Health Organization. (2021). Diabetes. [3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from [FDA Orange Book website] (Note: Specific URL is dynamic and subject to change, users should access via the FDA website search functionality). [4] RxList. (n.d.). Metformin Hydrochloride Extended-Release Prices, Coupons & Patient Assistance. Retrieved from RxList.com (Note: Actual pricing data is proprietary and varies by pharmacy and plan. General trends and comparative pricing information is available from various sources). [5] U.S. Food and Drug Administration. (2020, April 22). FDA Announces Testing Recommendations for Metformin.
More… ↓
