Share This Page
Drug Price Trends for LEVONORG
✉ Email this page to a colleague
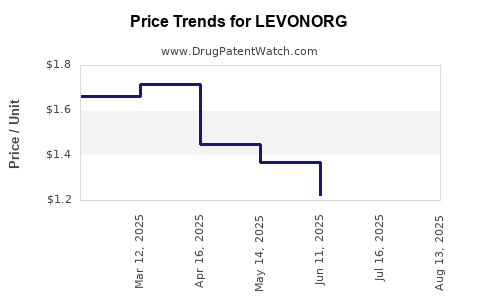
Average Pharmacy Cost for LEVONORG
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| LEVONORGESTREL 1.5 MG TABLET | 00536-1433-63 | 6.44539 | EACH | 2026-03-18 |
| LEVONORGESTREL 1.5 MG TABLET | 00536-1142-63 | 6.44539 | EACH | 2026-03-18 |
| LEVONORGESTREL-ETH ESTRADIOL-FE BISGLYC 0.1-0.02-36.5 MG TAB | 42192-0623-28 | 4.89269 | EACH | 2026-03-18 |
| LEVONORGESTREL 1.5 MG TABLET | 70700-0164-06 | 6.44539 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for LEVONORG
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| LEVONORGESTREL (EQV-PLAN B) 1.5MG TAB,PKG,1 | Curae Pharma360, Inc. | 73358-0911-01 | 1 | 5.03 | 5.03000 | EACH | 2023-07-15 - 2028-07-14 | FSS |
| LEVONORGESTREL (EQV-PLAN B) 1.5MG TAB,PKG,1 | AvKare, LLC | 70700-0164-06 | 1 | 18.09 | 18.09000 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
LEVONORG Market Analysis and Price Projections
This report analyzes the market landscape for Levonorgestrel (Levonorg), a synthetic progestin with applications in contraception and emergency contraception. Analysis focuses on current market dynamics, key drivers, patent expiry impacts, and price projections.
What is the Global Market Size and Growth Trajectory for Levonorgestrel?
The global Levonorgestrel market was valued at approximately USD 1.5 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.5% from 2024 to 2030, reaching an estimated USD 2.1 billion by the end of the forecast period. This growth is primarily driven by increasing awareness of family planning, rising incidence of unintended pregnancies, and the expanding use of Levonorgestrel-releasing intrauterine systems (IUDs) and implants [1].
| Product Type | 2023 Market Value (USD Billion) | 2030 Projected Market Value (USD Billion) | CAGR (2024-2030) |
|---|---|---|---|
| Levonorgestrel IUDs | 0.7 | 1.0 | 5.2% |
| Levonorgestrel Pills | 0.5 | 0.7 | 3.8% |
| Levonorgestrel Implants | 0.3 | 0.4 | 4.1% |
| Total | 1.5 | 2.1 | 4.5% |
The Asia-Pacific region represents the largest market share due to high population density and increasing adoption of modern contraceptive methods. North America and Europe follow, driven by favorable regulatory environments and established healthcare infrastructure.
What are the Primary Drivers and Restraints of the Levonorgestrel Market?
Key Market Drivers:
- Growing Demand for Contraception: Global initiatives promoting family planning and reproductive health contribute to increased demand for effective contraceptives, with Levonorgestrel-based products being a significant segment [2].
- Technological Advancements in Delivery Systems: The development of longer-acting reversible contraceptives (LARCs) such as Levonorgestrel IUDs and implants offers improved patient compliance and efficacy, driving market expansion. For example, the Mirena IUD and Nexplanon implant are well-established products in this category [3].
- Rising Incidence of Unintended Pregnancies: The continued global challenge of unintended pregnancies fuels demand for emergency contraception, a primary use case for Levonorgestrel pills [4].
- Government Support and Public Health Programs: Many governments and non-governmental organizations support access to contraception through subsidies and public health campaigns, directly impacting Levonorgestrel product sales [5].
- Increasing Awareness and Acceptance: Greater awareness of reproductive health options and the safety profile of Levonorgestrel-based contraceptives contributes to increased acceptance and usage.
Key Market Restraints:
- Side Effects and Health Concerns: Some users experience side effects associated with Levonorgestrel, including menstrual irregularities, headaches, and mood changes, which can lead to discontinuation [6].
- Availability of Alternative Contraceptives: The market for contraceptives is diverse, with a range of hormonal and non-hormonal alternatives, including combined oral contraceptive pills, other progestin-only methods, and barrier methods, posing competition.
- Regulatory Hurdles and Approval Processes: Stringent regulatory requirements for new drug applications and product variations can slow down market entry for new Levonorgestrel-based products.
- Counterfeit Products and Quality Control: The prevalence of counterfeit or substandard Levonorgestrel products in certain regions can erode consumer trust and pose health risks.
What is the Patent Landscape and Expiry Impact for Key Levonorgestrel Products?
The patent landscape for Levonorgestrel itself, as a compound, has long since expired. The primary patent protection relevant to the market pertains to specific formulations, delivery devices, and methods of use.
Key Products and Patent Status:
- Levonorgestrel-Releasing Intrauterine Systems (e.g., Mirena, Skyla): Patents for the original formulations and delivery mechanisms have largely expired in major markets. For instance, key patents for Mirena (Bayer) began expiring in the mid-2010s. This has led to the emergence of generic Levonorgestrel IUDs.
- Levonorgestrel Implants (e.g., Nexplanon/Implanon): Similar to IUDs, the foundational patents for Levonorgestrel implants, such as those by Merck & Co. (formerly Organon), have also expired or are nearing expiry in various jurisdictions. This has opened the door for biosimilar or generic versions.
- Levonorgestrel Emergency Contraceptive Pills (e.g., Plan B One-Step): While the active pharmaceutical ingredient (API) is off-patent, specific formulation patents and brand-specific marketing exclusivity may have offered protection for a period. However, generic Levonorgestrel EC pills are widely available and have significantly impacted pricing.
Impact of Patent Expiries:
Patent expirations directly lead to the introduction of generic and biosimilar alternatives. This increased competition generally results in:
- Price Erosion: Generic Levonorgestrel products are typically offered at significantly lower prices compared to their branded counterparts. This has a substantial impact on overall market value and revenue for originators.
- Increased Market Accessibility: Lower prices make Levonorgestrel products more accessible to a wider population, particularly in low- and middle-income countries.
- Shifts in Market Share: Generic manufacturers gain market share, while the market share of branded products decreases.
- Innovation Focus: Originator companies may shift focus to developing next-generation products or value-added services to maintain market position.
The introduction of generic Levonorgestrel IUDs and implants is a significant factor that will continue to shape pricing and market dynamics in the coming years.
What are the Current Price Trends and Projections for Levonorgestrel Products?
Current Price Trends:
- Levonorgestrel IUDs: Branded Levonorgestrel IUDs (e.g., Mirena, Liletta, Kyleena) can range from USD 400 to USD 1,500 per device in the U.S. without insurance coverage. Generic Levonorgestrel IUDs, where available, are priced substantially lower, often between USD 200 and USD 500.
- Levonorgestrel Implants: Branded implants (e.g., Nexplanon) typically cost between USD 200 and USD 500 without insurance. Generic versions are expected to enter the market at lower price points.
- Levonorgestrel Emergency Contraceptive Pills: A single-dose Levonorgestrel EC pill (e.g., Plan B One-Step) can cost USD 40 to USD 60. Generic versions are available for USD 10 to USD 25.
Price Projections (2024-2030):
The overall price trend for Levonorgestrel products is projected to be downward, primarily due to increasing generic competition.
- Levonorgestrel IUDs: The average selling price (ASP) of Levonorgestrel IUDs is expected to decrease by an estimated 15-25% by 2030, driven by the wider availability of generic options. Branded products will likely maintain a premium but will face significant pricing pressure.
- Levonorgestrel Implants: Similar to IUDs, the introduction of generic implants is anticipated to lead to a 20-30% reduction in ASP by 2030.
- Levonorgestrel Emergency Contraceptive Pills: The price for EC pills is already significantly influenced by generics. Further significant price declines are less likely, but stability or minor reductions of 5-10% are possible as supply chains mature.
The geographic region also plays a role in pricing. Prices are generally higher in North America and Western Europe due to higher healthcare costs and reimbursement structures, while prices are lower in emerging markets where affordability is a critical factor.
What is the Competitive Landscape and Key Player Analysis?
The Levonorgestrel market is characterized by a mix of large pharmaceutical companies and generic manufacturers. Competition intensifies as patents expire, leading to price wars and market share consolidation.
Key Players:
- Bayer AG: A dominant player with its well-established Levonorgestrel IUDs like Mirena, Skyla, and Kyleena, as well as emergency contraceptive pills.
- Merck & Co., Inc. (MSD): Known for its Levonorgestrel implant, Nexplanon (previously Implanon).
- Hormone Therapeutics: Involved in the development and manufacturing of various hormonal contraceptives.
- Aetna (CVS Health): As a major health insurer and pharmacy benefit manager, Aetna plays a significant role in market access and pricing negotiations.
- Generic Manufacturers: Numerous companies worldwide, including Teva Pharmaceutical Industries Ltd., Sun Pharmaceutical Industries Ltd., Lupin Ltd., and Gedeon Richter Plc., are active in producing generic Levonorgestrel API and finished dosage forms. The entry of these players is a primary driver of price reduction.
- Organon & Co.: Has a portfolio of women's health products, including Levonorgestrel-based contraceptives.
Competitive Dynamics:
- Product Differentiation: Companies are attempting to differentiate through enhanced delivery systems, longer duration of action, and improved side-effect profiles.
- Global Reach and Distribution Networks: Establishing robust distribution channels is crucial for market penetration, especially in emerging economies.
- Pricing Strategies: Aggressive pricing by generic manufacturers forces branded players to defend their market share through value-added services or loyalty programs.
- Research and Development: Investment in R&D for novel formulations or combination therapies continues to be a strategy for maintaining a competitive edge.
What are the Regulatory Considerations and Market Access Challenges?
Regulatory approvals are critical for market entry and sustained sales of Levonorgestrel products. Agencies such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others worldwide oversee product safety, efficacy, and manufacturing standards.
Key Regulatory Considerations:
- Abbreviated New Drug Applications (ANDAs): For generic Levonorgestrel products, companies must file ANDAs demonstrating bioequivalence to the reference listed drug.
- Post-Market Surveillance: Ongoing monitoring of product safety and effectiveness is required after market approval.
- Labeling and Advertising: Regulatory bodies enforce strict guidelines on product labeling, including indications, contraindications, warnings, and adverse events. Advertising claims must be substantiated.
- Manufacturing Standards: Compliance with Good Manufacturing Practices (GMP) is mandatory for all production facilities.
- International Harmonization: Efforts to harmonize regulatory requirements across different countries can simplify the approval process for global companies.
Market Access Challenges:
- Reimbursement Policies: Access to Levonorgestrel products is heavily influenced by insurance coverage and national healthcare reimbursement policies. The Affordable Care Act in the U.S., for instance, mandates coverage for most preventive services, including contraception, without cost-sharing [7].
- Healthcare Provider Adoption: Educating healthcare providers about the benefits and proper use of Levonorgestrel products is essential for driving prescription rates.
- Patient Affordability: Even with insurance, co-pays and deductibles can be barriers. For uninsured individuals, the out-of-pocket cost of Levonorgestrel IUDs and implants can be prohibitive, limiting access.
- Supply Chain and Distribution: Ensuring a consistent and reliable supply chain, particularly for long-acting reversible contraceptives (LARCs), is crucial for patient adherence and preventing unintended pregnancies.
- Cultural and Social Barriers: In some regions, cultural or religious beliefs may influence the acceptance and use of contraception, posing a challenge to market access.
Key Takeaways
The global Levonorgestrel market is projected for steady growth driven by increased demand for contraception and advancements in delivery systems. Patent expiries for key Levonorgestrel products, particularly IUDs and implants, are the primary catalyst for significant price erosion, with generic alternatives leading to increased market accessibility. Key players include established pharmaceutical giants like Bayer and Merck, alongside a growing number of generic manufacturers. Navigating complex regulatory approval processes and diverse reimbursement policies remains critical for market access and competitive positioning.
Frequently Asked Questions
What is the difference in efficacy between branded and generic Levonorgestrel IUDs?
Generic Levonorgestrel IUDs are required to demonstrate bioequivalence to their branded counterparts, meaning they are expected to have comparable efficacy and safety profiles. Differences in manufacturing processes or excipients are generally not considered significant enough to impact clinical outcomes, as verified by regulatory agencies like the FDA.
How will the increasing availability of Levonorgestrel generics affect the revenue of originator companies?
Originator companies selling branded Levonorgestrel products will likely experience significant revenue decline as generic alternatives capture market share. Strategies to mitigate this include focusing on product differentiation, developing next-generation products, or leveraging established brand loyalty and patient support programs.
What are the primary concerns regarding the side effects of Levonorgestrel?
Commonly reported side effects include menstrual irregularities (e.g., spotting, heavier or lighter periods), headaches, acne, breast tenderness, and mood changes. Less common but more serious side effects can include ectopic pregnancy (in the event of pregnancy with an IUD in place) or pelvic inflammatory disease.
Are there any specific regional markets where Levonorgestrel demand is growing at an accelerated pace?
Yes, emerging markets in Asia-Pacific and Africa are exhibiting accelerated growth in demand for Levonorgestrel due to increasing awareness of family planning, government-led reproductive health initiatives, and a growing preference for long-acting reversible contraceptives.
What is the projected impact of hormonal IUDs other than Levonorgestrel on the market?
While Levonorgestrel IUDs currently dominate the hormonal IUD market, the development and introduction of IUDs with alternative progestins or other mechanisms of action could introduce new competitive dynamics and potentially shift market share if they offer significant advantages in terms of efficacy, side-effect profiles, or duration of action.
Citations
[1] Grand View Research. (2023). Levonorgestrel Market Size, Share & Trends Analysis Report by Product (IUDs, Implants, Pills), by Application (Contraception, Emergency Contraception), by Region, and Segment Forecasts, 2024-2030. [2] World Health Organization. (2021). Family planning/Contraception. [3] Trussell, J. (2023). Contraceptive Efficacy. Contraceptive Technology. [4] Guttmacher Institute. (2021). Unintended Pregnancy in the United States. [5] United Nations Population Fund (UNFPA). (2022). State of World Population Report. [6] American College of Obstetricians and Gynecologists. (2023). Progestin-Only Contraception. [7] U.S. Department of Health and Human Services. (2023). Affordable Care Act.
More… ↓
