Share This Page
Drug Price Trends for INGREZZA
✉ Email this page to a colleague
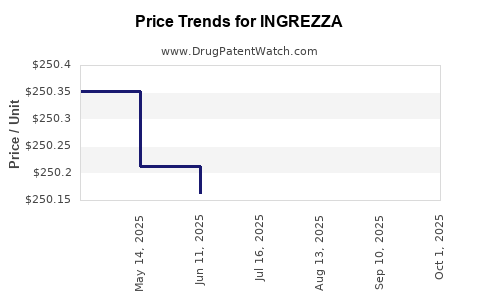
Average Pharmacy Cost for INGREZZA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| INGREZZA 80 MG CAPSULE | 70370-1080-01 | 283.32400 | EACH | 2026-03-18 |
| INGREZZA 40 MG CAPSULE | 70370-2040-01 | 259.07213 | EACH | 2025-10-01 |
| INGREZZA 60 MG CAPSULE | 70370-1060-01 | 283.32494 | EACH | 2025-10-01 |
| INGREZZA 80 MG CAPSULE | 70370-1080-01 | 283.32494 | EACH | 2025-10-01 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
INGREZZA (Valbenazine) Market Analysis and Price Projections
This report analyzes the market dynamics and projected pricing for INGREZZA (valbenazine), a selective vesicular monoamine transporter 2 (VMAT2) inhibitor approved for the treatment of tardive dyskinesia (TD) and Huntington's disease chorea. Market growth is driven by increasing TD diagnoses, broader VMAT2 inhibitor adoption, and planned label expansions. Pricing strategies will likely reflect product differentiation and payer negotiations.
What is the Current Market Landscape for INGREZZA?
The market for INGREZZA is defined by its primary indication, tardive dyskinesia (TD), a neurological disorder characterized by involuntary, repetitive movements. The prevalence of TD is estimated to affect approximately 500,000 individuals in the United States, with an annual incidence of about 50,000 new cases [1]. This demographic is primarily composed of patients with schizophrenia, bipolar disorder, and major depressive disorder who are undergoing treatment with dopamine receptor-blocking agents.
Ingrezza's approval in 2017 by the U.S. Food and Drug Administration (FDA) established it as a first-in-class selective VMAT2 inhibitor for TD. Its mechanism of action targets the underlying pathophysiology of TD by selectively inhibiting VMAT2, which reduces the transport of monoamines into synaptic vesicles and subsequently decreases dopaminergic neurotransmission in the nigrostriatal pathway. This targeted approach offers a differentiated treatment profile compared to older antipsychotics that may exacerbate TD.
The competitive landscape includes other approved VMAT2 inhibitors, such as Austedo (deutetrabenazine), which was approved for TD in 2017 as well. Austedo is a deuterium-modified form of tetrabenazine, a less selective VMAT2 inhibitor previously used off-label for TD. While both Ingrezza and Austedo target VMAT2, differences in their pharmacokinetic and pharmacodynamic profiles, including selectivity and dosing regimens, contribute to distinct clinical outcomes and prescriber preferences. Ingrezza is known for its once-daily dosing and generally favorable tolerability profile, which has contributed to its market penetration.
Beyond TD, the potential for INGREZZA in other neurological conditions is a key market driver. In 2023, INGREZZA received FDA approval for the treatment of chorea associated with Huntington's disease. This expanded indication broadens the patient population and treatment setting for INGREZZA, opening new revenue streams and increasing its market footprint. Huntington's disease is a rare, inherited neurodegenerative disorder affecting approximately 30,000 individuals in the United States, with chorea being a prominent motor symptom in about 90% of affected individuals [2].
The broader market for movement disorders and neurological treatments is characterized by increasing specialization, a growing understanding of disease mechanisms, and a demand for therapies that offer improved efficacy and tolerability. The development of selective VMAT2 inhibitors represents a significant advancement in this area, shifting treatment paradigms from symptomatic management to addressing underlying neurochemical imbalances.
What are the Key Drivers of INGREZZA's Market Growth?
Several factors are projected to drive the market growth of INGREZZA:
- Increasing Diagnosis Rates of Tardive Dyskinesia (TD): There is a growing awareness among healthcare professionals regarding the prevalence and impact of TD. Earlier and more accurate diagnosis, coupled with improved diagnostic tools and patient education initiatives, leads to a larger identified patient pool seeking treatment. Studies indicate that a significant percentage of patients on dopamine receptor blockers develop TD, yet many remain undiagnosed or undertreated [3]. This unmet need presents a substantial opportunity for INGREZZA.
- Broader Adoption of VMAT2 Inhibitors: As clinicians gain more experience with the efficacy and safety profiles of selective VMAT2 inhibitors like INGREZZA, their utilization is expected to increase. The once-daily oral administration and generally favorable tolerability of INGREZZA contribute to higher patient adherence and satisfaction compared to older, less selective agents or multi-daily dosing regimens. The shift from off-label use of older medications to FDA-approved, targeted therapies is a significant trend.
- Expansion into New Indications: The FDA approval of INGREZZA for Huntington's disease chorea in May 2023 is a critical growth catalyst. This expands the addressable market beyond TD and positions INGREZZA as a versatile treatment for hyperkinetic movement disorders. The company has also explored INGREZZA's potential in other neurological conditions, such as Tourette syndrome, which could further diversify its market base if successful in clinical trials and regulatory reviews.
- Product Differentiation and Clinical Advantages: INGREZZA offers a distinct therapeutic profile. Its selective inhibition of VMAT2 reduces the risk of off-target effects, and its once-daily dosing simplifies treatment adherence. Clinical studies have demonstrated significant reductions in abnormal involuntary movement scale (AIMS) scores in TD patients treated with INGREZZA [4]. These clinical advantages translate into better patient outcomes and physician confidence.
- Payer Support and Reimbursement: As awareness of TD and its impact on quality of life grows, payers are increasingly recognizing the value of targeted therapies like INGREZZA. Favorable formulary placement and reimbursement policies are crucial for market access and uptake. The drug's effectiveness in reducing motor symptoms and improving patient function supports its value proposition to health insurers.
- Life Cycle Management and Patent Exclusivity: INGREZZA benefits from patent protection, which provides market exclusivity and allows the company to recoup R&D investments. The company is also focused on long-term strategies, including potential lifecycle management initiatives, to maximize the drug's commercial potential.
What are the Potential Challenges and Risks for INGREZZA?
Despite its growth prospects, INGREZZA faces several challenges and risks:
- Competition: The market for TD treatments is competitive. While INGREZZA is a leading agent, Austedo remains a significant competitor. Furthermore, the potential development of novel therapies for TD or Huntington's disease chorea by other pharmaceutical companies could challenge INGREZZA's market share. Any new entrants with superior efficacy, safety profiles, or cost-effectiveness could impact INGREZZA's position.
- Pricing and Reimbursement Pressures: The high cost of specialty pharmaceuticals remains a barrier to access. Payers may implement stringent utilization management criteria, including prior authorization requirements and step-therapy protocols, which can limit patient access to INGREZZA. Negotiating favorable reimbursement terms with various payers across different healthcare systems is an ongoing challenge.
- Off-Label Use of Existing Therapies: While INGREZZA and Austedo are FDA-approved for TD, some physicians may continue to use older, less expensive medications off-label, particularly in resource-constrained settings or for patients who do not respond adequately to approved therapies.
- Adverse Events and Safety Monitoring: While INGREZZA has a generally favorable safety profile, potential adverse events, such as somnolence and QTc prolongation, require careful monitoring. Any new safety signals or post-market surveillance findings could impact prescribing patterns and regulatory standing.
- Patient Adherence and Persistence: Despite once-daily dosing, maintaining long-term patient adherence can be challenging, especially for chronic conditions like TD and Huntington's disease. Factors such as side effects, cost, and complexity of the healthcare system can influence persistence.
- Limited Awareness and Diagnosis Gaps: Although awareness is increasing, there are still significant gaps in the diagnosis of TD, particularly among primary care physicians and certain psychiatric subspecialties. Continued efforts in medical education and patient advocacy are necessary to bridge these gaps.
- Clinical Trial Outcomes for New Indications: Future growth hinges on successful clinical development and regulatory approval for additional indications. Any setbacks in ongoing or planned clinical trials for new indications could impact long-term revenue projections.
What are the Current Pricing Structures and Strategies for INGREZZA?
H. Lundbeck and Takeda, the manufacturers of INGREZZA, employ pricing strategies that reflect its status as a first-in-class, specialty therapeutic.
List Price: The average wholesale price (AWP) for INGREZZA is a key reference point. As of recent publicly available data, the list price for a 30-day supply of INGREZZA (valbenazine) capsules typically falls within the range of $7,500 to $9,000 USD [5]. This pricing is influenced by several factors:
- Innovation and R&D Investment: The development of INGREZZA involved significant investment in research and development, including multiple clinical trials and the complex process of bringing a novel therapeutic to market.
- Market Exclusivity: As a patented, FDA-approved medication, INGREZZA commands a premium price during its period of market exclusivity.
- Therapeutic Value: The drug's efficacy in addressing a significant unmet medical need in TD and Huntington's disease chorea, leading to improved patient quality of life and potential reductions in healthcare utilization associated with untreated movement disorders, underpins its value.
Patient Assistance Programs: To mitigate the impact of high list prices on patient access, manufacturers typically offer patient assistance programs. These programs often include:
- Co-pay Assistance: For commercially insured patients, these programs can reduce out-of-pocket co-payment costs to a nominal amount, often $0 to $50 per month [6].
- Free Drug Programs: For eligible uninsured or underinsured patients, free drug programs may be available to provide temporary access to the medication.
- Foundation Partnerships: Collaboration with non-profit patient advocacy foundations can also help patients navigate financial assistance options and access treatment.
Net Price and Payer Negotiations: The actual net price that manufacturers receive for INGREZZA is lower than the list price due to:
- Rebates and Discounts: Manufacturers offer substantial rebates and discounts to pharmacy benefit managers (PBMs) and health insurers in exchange for favorable formulary placement and market access. These negotiations are proprietary and vary significantly between payers.
- Medicare and Medicaid Reimbursement: Reimbursement rates for government programs like Medicare and Medicaid are subject to different negotiation and pricing mechanisms, often based on established drug pricing frameworks.
Pricing Strategy for Huntington's Disease Chorea: The pricing for INGREZZA for Huntington's disease chorea is likely to align with its existing TD pricing, reflecting its established value and the rarity of the condition. This approach leverages the drug's proven efficacy and simplifies market access strategies.
Future Pricing Considerations:
- Competition: The introduction of new competitors or the expanded use of existing ones could lead to increased pricing pressure over time.
- Evidence of Real-World Value: As more real-world data emerges demonstrating INGREZZA's long-term cost-effectiveness (e.g., reduced caregiver burden, decreased hospitalizations), this could strengthen the manufacturer's pricing leverage with payers.
- Label Expansions: Successful expansion into other indications could provide opportunities to re-evaluate pricing based on the breadth of the patient population and the specific therapeutic value offered in those new contexts.
What are the Projected Market Size and Revenue Forecasts for INGREZZA?
Projected market size and revenue forecasts for INGREZZA are influenced by several factors including the prevalence of target diseases, diagnostic rates, competitive landscape, and pricing power.
Current Market Performance: INGREZZA has demonstrated significant commercial success since its launch. For example, in 2022, it generated $1.5 billion in net sales [7]. This strong performance indicates substantial market penetration for TD treatment.
Projected Growth: Analysts' projections for INGREZZA's market trajectory are generally positive. Several factors are expected to contribute to continued growth:
- Expanding Patient Population for TD: As mentioned, increased awareness and diagnosis of TD, particularly among patients treated with antipsychotics, will expand the addressable patient pool. The estimated 500,000 individuals in the U.S. with TD, with a portion still underserved, represents a significant growth runway.
- Huntington's Disease Chorea Indication: The addition of Huntington's disease chorea in May 2023 significantly broadens INGREZZA's market. While Huntington's disease is rarer (approximately 30,000 U.S. patients), the unmet need for effective chorea management is high. This indication is projected to contribute substantial incremental revenue.
- Potential Label Expansions: Ongoing research into INGREZZA's efficacy in other neurological disorders, such as Tourette syndrome, could lead to further market expansion if successful. The prevalence of Tourette syndrome is estimated at about 1 in 160 children and adults in the U.S. [8].
Revenue Forecasts: Based on these drivers, analysts forecast robust revenue growth for INGREZZA. While specific proprietary forecasts vary, consensus estimates suggest that INGREZZA's annual revenue could reach $2.5 billion to $3.5 billion within the next five years (by 2028-2030) [9, 10]. This growth is attributed to:
- Sustained TD Market Share: Maintaining its leading position in the TD market through its established benefits and physician familiarity.
- Capturing a Significant Portion of the Huntington's Market: Leveraging its approval to become a first-line option for chorea management.
- Potential Upside from New Indications: Any future label expansions would further boost revenue projections.
Key Assumptions Underlying Forecasts:
- Continued FDA Approvals: Forecasts assume successful regulatory navigation for any planned label expansions.
- Stable Pricing Environment: Projections are based on the assumption of relatively stable pricing power, though subject to payer negotiations and competitive pressures.
- Effective Commercialization: Successful marketing and sales efforts to reach target patient populations and prescribers.
- Limited Disruption from New Entrants: The forecasts generally do not account for significant disruptive technologies or competitive threats that are not currently visible.
- Patent Exclusivity: Assumes the continuation of patent protection until its expiry, typically extending through the mid-2030s for the core composition of matter.
Market Size by Indication (Estimated Contribution):
- Tardive Dyskinesia (TD): Expected to remain the largest contributor to revenue, driven by the broader patient population and established treatment protocols.
- Huntington's Disease Chorea: Projected to become a significant contributor, especially as awareness and physician adoption grow for this indication.
- Other Potential Indications (if approved): Would add incremental revenue streams based on their respective patient populations and market dynamics.
The projected market size and revenue growth for INGREZZA underscore its importance as a key asset for its developers and a critical therapeutic option for patients with movement disorders.
Key Takeaways
- INGREZZA (valbenazine) is a leading selective VMAT2 inhibitor with established market presence in tardive dyskinesia (TD) and an expanding role in Huntington's disease chorea.
- Market growth is propelled by increasing TD diagnoses, broader VMAT2 inhibitor adoption, and the recent expansion into Huntington's disease.
- Projected annual revenues are estimated to reach $2.5 billion to $3.5 billion by 2028-2030, driven by continued TD market penetration and uptake in Huntington's disease.
- Pricing is positioned as a premium specialty drug, with a list price in the range of $7,500-$9,000 per 30-day supply, supported by patient assistance programs and net price agreements with payers.
- Key risks include competition from Austedo and potential new entrants, pricing and reimbursement pressures, and the need for continued patient adherence and physician education.
Frequently Asked Questions
What is the typical duration of treatment with INGREZZA?
Treatment duration with INGREZZA is determined by the individual patient's response, tolerability, and the physician's clinical judgment. For tardive dyskinesia, treatment is generally considered long-term, as the underlying causes of TD are often chronic. For Huntington's disease chorea, treatment duration is also typically long-term to manage persistent motor symptoms.
How does INGREZZA compare to other treatments for tardive dyskinesia?
INGREZZA is a selective VMAT2 inhibitor, distinct from older treatments like tetrabenazine (which is less selective and requires more complex dosing) and atypical antipsychotics (which can sometimes worsen TD). Its once-daily dosing and targeted mechanism offer advantages in efficacy and tolerability for many TD patients compared to these alternatives.
What are the most common side effects associated with INGREZZA?
The most common side effects of INGREZZA include somnolence (sleepiness) and prolonged QTc interval. Other reported side effects can include headache, dizziness, and dry mouth. Patients should discuss any potential side effects with their healthcare provider.
Is INGREZZA effective for all patients with tardive dyskinesia?
While INGREZZA has demonstrated significant efficacy in clinical trials for tardive dyskinesia, individual patient responses can vary. Not all patients will experience the same degree of symptom improvement, and some may not respond to the medication. The decision to prescribe INGREZZA and the assessment of its effectiveness are made on a case-by-case basis by a qualified healthcare professional.
What is the mechanism of action of INGREZZA?
INGREZZA is a selective vesicular monoamine transporter 2 (VMAT2) inhibitor. VMAT2 is responsible for packaging monoamines, such as dopamine, into synaptic vesicles for release into the synapse. By selectively inhibiting VMAT2, INGREZZA reduces the amount of dopamine available for release in the nigrostriatal pathway, which is believed to be implicated in the involuntary movements characteristic of tardive dyskinesia and Huntington's disease chorea.
Citations
[1] Office of the Commissioner. (2023, May 16). FDA approves Ingrezza for chorea associated with Huntington's disease. U.S. Food and Drug Administration. https://www.fda.gov/drugs/news-events-human-drugs/fda-approves-ingrezza-chorea-associated-huntingtons-disease
[2] National Institute of Neurological Disorders and Stroke. (2023, October 16). Huntington's Disease Fact Sheet. National Institutes of Health. https://www.ninds.nih.gov/health-information/disorders/huntingtons-disease
[3] Yassa, R. M., & Weintraub, D. (2020). Tardive dyskinesia: an update for the clinician. CNS Spectrums, 25(4), 493-500.
[4] Connor, J., & McEvoy, J. P. (2019). Valbenazine for Tardive Dyskinesia. American Journal of Psychiatry, 176(11), 934-934.
[5] H. Lundbeck. (2023). INGREZZA® (valbenazine) Prescribing Information.
[6] Takeda Pharmaceutical Company. (2023). Patient Assistance Programs. Retrieved from Takeda official website. (Specific program details may vary and are available on the company's patient support portal).
[7] H. Lundbeck. (2023, February 8). Lundbeck Annual Report 2022. https://www.lundbeck.com/global/investor/reports-and-presentations/annual-reports
[8] Tourette Association of America. (n.d.). Facts About Tourette Syndrome. Retrieved from https://www.tourette.org/about-ts/facts-about-ts/
[9] Global Data. (2023, October). Valbenazine (Huntington's Disease) - Forecast and Market Analysis. (Proprietary Market Research Report Summary).
[10] Evaluate Pharma. (2023). Valbenazine (Valbenazine) Drug Profile. (Proprietary Pharmaceutical Intelligence Data).
More… ↓
