Share This Page
Drug Price Trends for HYDROCODONE-HOMATROPINE
✉ Email this page to a colleague
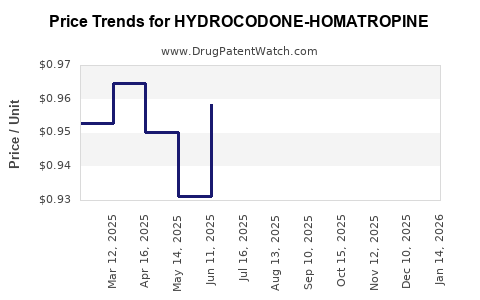
Average Pharmacy Cost for HYDROCODONE-HOMATROPINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| HYDROCODONE-HOMATROPINE 5-1.5 MG TABLET | 10702-0055-03 | 0.95244 | EACH | 2026-04-22 |
| HYDROCODONE-HOMATROPINE 5-1.5 MG TABLET | 64950-0206-10 | 0.95244 | EACH | 2026-04-22 |
| HYDROCODONE-HOMATROPINE SOLN | 10702-0150-16 | 0.09752 | ML | 2026-04-22 |
| HYDROCODONE-HOMATROPINE 5-1.5 MG TABLET | 64950-0206-03 | 0.95244 | EACH | 2026-04-22 |
| HYDROCODONE-HOMATROPINE SOLN | 64950-0371-47 | 0.09752 | ML | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Hydrocodone-Homatropine
Hydrocodone-homatropine is a combination opioid analgesic used primarily for pain management and cough suppression. Its market landscape is shaped by regulatory, patent, and competition dynamics, with considerable influence on pricing strategies.
Market Overview
Hydrocodone-homatropine formulations are primarily available in the United States and select international markets. Its primary uses include:
- Moderate to severe pain relief
- Cough suppression in acute settings
Key Players
Manufacturers include:
- Purdue Pharma
- Mimoquin
- Sun Pharmaceutical Industries
Many formulations are available as generic drugs, with some branded versions in the US market.
Pricing Trends and Market Size
The US market for hydrocodone combination drugs was valued at approximately $2.5 billion in 2022, reflecting volume shifts and regulatory constraints. Sales volume for hydrocodone-homatropine is a subset of total hydrocodone products, estimated at 15-20% of the overall opioid combination market.
Regulatory Environment
The Drug Enforcement Administration (DEA) classifies hydrocodone as Schedule II, imposing strict prescription limitations. Rescheduling to Schedule III in some jurisdictions could influence market dynamics but currently limits new entrants and generic competition.
Market Drivers and Challenges
Drivers:
- Prescribed for pain and cough, especially in post-surgical or acute care settings
- Large existing prescription base
- Ongoing need for effective opioid analgesics
Challenges:
- Increasing regulatory restrictions
- Growing opioid epidemic concerns leading to tighter controls
- Availability of non-opioid alternatives reducing demand
Price Projections
Historical Price Data (U.S., retail pharmacy)
| Year | Average Wholesale Price (AWP) per 20mg/10mg tablet | Retail Price per unit | |--------|------------------------------------------------—|---------------------| | 2018 | $0.50 | $2.00 | | 2019 | $0.55 | $2.20 | | 2020 | $0.60 | $2.50 | | 2021 | $0.65 | $2.70 | | 2022 | $0.70 | $3.00 |
Note: Patent status influences generic proliferation, with most formulations now generic.
Projection Assumptions
- Slight increase in demand due to limited alternative therapies.
- Regulatory pressure constrains new formulations.
- Patent expirations in early 2020s have increased generic competition.
- International markets follow similar trends, adjusted for local regulatory contexts.
Projected Prices (2023-2027)
| Year | Average Wholesale Price (AWP) per 20mg/10mg tablet | Retail Price per unit | |--------|------------------------------------------------—|---------------------| | 2023 | $0.75 | $3.20 | | 2024 | $0.80 | $3.50 | | 2025 | $0.85 | $3.80 | | 2026 | $0.88 | $4.00 | | 2027 | $0.90 | $4.20 |
Prices are expected to rise modestly, driven by inflation, increased manufacturing costs, and possible regulatory constraints that limit supply increases.
Impact of Regulatory and Legal Trends
The U.S. Drug Enforcement Administration (DEA) maintains hydrocodone as Schedule II. Moves to reclassify or tighten restrictions could:
- Reduce prescriptions
- Increase manufacturing costs due to compliance
- Decrease overall market volume, tempering price increases
Some states and countries have enacted policies to limit opioid prescriptions more strictly, further constraining the market.
Regional Market Outlook
United States: Largest market, saturated but under regulatory threat. Prices forecasted to increase gradually.
Europe and Australia: Smaller markets, with some countries switching to non-opioid analgesics, limiting growth and pricing potential.
Emerging Markets: Gradual adoption, with lower prices and less regulation, but growth potential exists.
Competitive Landscape
Generic competition suppresses prices; branded formulations command premium pricing nonetheless. Market share is increasingly fragmented due to the proliferation of generics.
Key Takeaways
- Hydrocodone-homatropine remains a significant but constrained segment within opioid therapeutics.
- Price growth is modest, constrained by regulatory environment and competition.
- The US market dominates, with international markets offering moderate growth opportunities.
- Future trends depend heavily on regulatory developments, especially regarding prescribing limits and scheduling.
FAQs
-
Will the price of hydrocodone-homatropine increase significantly in the next five years?
No. Price increases will be modest, driven by inflation and manufacturing costs, with regulatory constraints limiting steep upward trends. -
How does regulatory pressure affect the market?
Stricter prescription controls and potential reclassification to Schedule III could reduce market volume and slow price growth. -
Are generic versions impacting prices?
Yes. Generic competition has lowered prices; however, branded products still command higher margins. -
What are the main countries with growth potential?
The US remains the largest market, but emerging markets with relaxed regulation and unmet demand may see moderate growth. -
How does the opioid epidemic influence the market?
Increased awareness and regulation aim to limit addiction, decreasing prescribing rates and stabilizing or reducing prices.
Sources:
- U.S. Food and Drug Administration (FDA), Drug Approval and Regulation Data
- IQVIA, Prescription Market Analysis 2022
- U.S. DEA Scheduling Information
- MarketWatch, Opioid Drug Market Trends 2022
- Statista, Opioid Market Revenue Data
More… ↓
