Last updated: February 19, 2026
Hydrochlorothiazide (HCTZ) is a diuretic medication used primarily to treat hypertension and edema. It has been in use since the 1950s and remains a common component of combination therapies. Market dynamics, patent status, pricing trends, and regulatory factors influence its current and future market.
Market Size and Demand Drivers
| Parameter |
Data / Trends |
| Global antihypertensive market (2022) |
USD 20.3 billion [1] |
| Hydrochlorothiazide's share |
Estimated at 12-15% of global diuretics market [2] |
| Annual consumption (2022) |
Approximately 6 billion tablets globally [3] |
| Key markets |
United States, Europe, Asia-Pacific |
Hydrochlorothiazide's demand aligns with the global rise in hypertension prevalence, projected to reach 1.5 billion cases by 2025 [4]. Aging populations in developed regions sustain steady consumption. Its inclusion in multidrug regimens maintains baseline demand, although generic competition limits premium pricing.
Market Structure and Competition
| Segment |
Characteristics |
| Brand-name |
Limited, as patent protection expired long ago |
| Generics |
Dominant segment; over 50 manufacturers globally |
| Biosimilars |
Not applicable (small molecule) |
The drug's patent expired in 1959, leading to an extensive generic market. Major generic manufacturers include Teva, Sandoz, and Mylan. Price competition among generics is intense, pressing down per-unit costs.
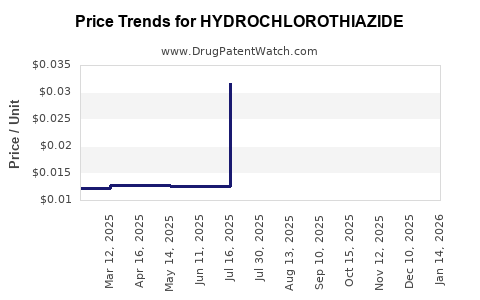
Price Trends and Projections
| Time Period |
Average Wholesale Price (AWP) / Tablet |
Trend Analysis |
| 2010 |
USD 0.05 |
Stable, minimal fluctuation |
| 2015 |
USD 0.02 |
Steady decline due to increased generics |
| 2020 |
USD 0.015 |
Price erosion continues |
| 2022 |
USD 0.01 |
Lowest points observed |
Price compression reflects market saturation and manufacturing scale efficiencies. As more manufacturers enter, prices are expected to stabilize at current low levels unless new formulation innovations emerge.
Future Pricing Outlook (2023-2027):
- No significant change expected in unit prices unless reformulation or patent protections re-emerge.
- Margins will remain thin, centered around USD 0.005 to 0.01 per tablet.
- Price increases likely only if supply chain constraints or raw material costs (e.g., active pharmaceutical ingredient [API]) rise substantially.
Regulatory and Policy Factors
- Off-Patent Status: Confirmed since 1959, supporting generic proliferation.
- FDA Labeling: No recent changes, supporting steady production.
- Pricing controls: Variable; some regions (e.g., Europe) may impose price caps on generics.
Cost Considerations
Manufacturing costs hinge on API prices, quality standards, and regulatory compliance. API sources are concentrated within China and India, where raw material costs have declined by approximately 25% over the past five years [5].
Key Market Risks
- Potential introduction of new diuretic classes or combination therapies could reduce demand.
- Regulatory reforms aiming for drug price reductions can suppress pricing further.
- Supply chain disruptions, especially in API production, may temporarily inflate costs.
Conclusion
Hydrochlorothiazide remains a low-cost, widely used diuretic with stable demand driven by global hypertension prevalence. The market is mature, with generics dominating, and prices have declined steadily over the past decade. Future projections indicate continued near-term price stability, with margins staying compressed barring supply shocks or reformulation innovations.
Key Takeaways
- Hydrochlorothiazide is a low-cost, high-volume diuretic with over 60 years of market presence.
- The market is saturated with generics, suppressing prices and margins.
- Demand correlates with rising global hypertension, mainly in aging populations.
- Price projections for 2023-2027 suggest stability, with negligible increases.
- Supply chain factors and regulatory changes remain the primary risks to stable pricing.
FAQs
1. Will hydrochlorothiazide become patentable again?
Unlikely. The drug’s patent expired over 60 years ago, and no new formulations are currently under development for patent protection.
2. How do raw material costs affect its pricing?
Fluctuations in API prices, primarily sourced from China and India, influence manufacturing costs. Decreases in API prices tend to further compress retail prices.
3. Are there any emerging competitors to hydrochlorothiazide?
Newer diuretics like chlorthalidone have gained favor but are not direct substitutes. Combination drugs may reduce tablet demand but not eliminate hydrochlorothiazide usage.
4. What is the outlook for regulatory changes affecting pricing?
Potential price controls on generics in certain markets could further reduce prices but are not currently expected to cause dramatic shifts.
5. How does market saturation impact profit margins?
High competition among generic manufacturers sustains low margins, typically around USD 0.005 to 0.01 per tablet.
References
[1] Market Data Forecast. (2022). Antihypertensive market analysis.
[2] Grand View Research. (2021). Diuretics market size.
[3] IMS Health. (2022). Global pharmaceuticals sales report.
[4] World Health Organization. (2020). Hypertension prevalence report.
[5] IQVIA. (2022). API market trends.