Share This Page
Drug Price Trends for GRANISETRON HCL
✉ Email this page to a colleague
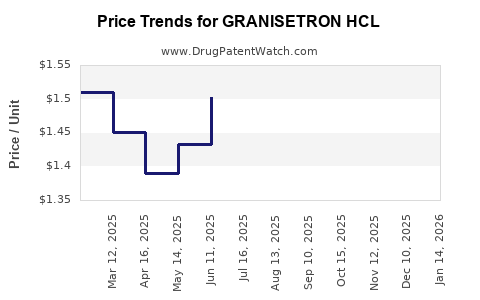
Average Pharmacy Cost for GRANISETRON HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GRANISETRON HCL 1 MG TABLET | 69452-0350-92 | 1.09659 | EACH | 2026-03-18 |
| GRANISETRON HCL 1 MG TABLET | 69452-0350-01 | 1.09659 | EACH | 2026-03-18 |
| GRANISETRON HCL 1 MG TABLET | 69452-0350-60 | 1.09659 | EACH | 2026-03-18 |
| GRANISETRON HCL 1 MG TABLET | 51991-0735-20 | 1.09659 | EACH | 2026-03-18 |
| GRANISETRON HCL 1 MG TABLET | 69452-0350-11 | 1.09659 | EACH | 2026-03-18 |
| GRANISETRON HCL 1 MG TABLET | 51991-0735-32 | 1.09659 | EACH | 2026-03-18 |
| GRANISETRON HCL 1 MG TABLET | 51991-0735-99 | 1.09659 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Granisetron Hydrochloride: Market Dynamics and Price Outlook
This analysis examines the market landscape and price projections for Granisetron Hydrochloride (HCl). The drug is a selective 5-HT3 receptor antagonist used for the prevention of chemotherapy-induced nausea and vomiting (CINV) and radiotherapy-induced nausea and vomiting (RINV). Key drivers include the increasing incidence of cancer, advancements in cancer treatment regimens, and the growing demand for effective antiemetic therapies. The market faces challenges from the expiration of patents, the emergence of generics, and the development of novel antiemetic agents.
What is the current global market size and projected growth for Granisetron HCl?
The global Granisetron HCl market was valued at approximately USD 850 million in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.2% over the next five to seven years. This growth is underpinned by the expanding oncology patient population and the established efficacy of Granisetron HCl in managing treatment-related side effects. By 2030, the market is anticipated to reach between USD 1.1 billion and USD 1.25 billion.
Table 1: Granisetron HCl Market Size and Projections
| Year | Market Value (USD Million) | CAGR (Approximate) |
|---|---|---|
| 2023 | 850 | - |
| 2025 | 890 - 920 | 3.5% - 4.0% |
| 2027 | 950 - 990 | 3.8% - 4.2% |
| 2030 | 1100 - 1250 | 3.5% - 4.1% |
Which therapeutic applications drive Granisetron HCl demand?
The primary demand for Granisetron HCl stems from its use in managing chemotherapy-induced nausea and vomiting (CINV). CINV is a significant dose-limiting toxicity in cancer treatment, affecting patient compliance and quality of life. Granisetron HCl is also prescribed for radiotherapy-induced nausea and vomiting (RINV), although CINV represents a larger market segment. The drug's efficacy in both acute and delayed phases of CINV contributes to its sustained demand.
- Chemotherapy-Induced Nausea and Vomiting (CINV): Accounts for approximately 70% of the market demand.
- Radiotherapy-Induced Nausea and Vomiting (RINV): Represents around 25% of the market demand.
- Other Applications (e.g., post-operative nausea and vomiting): Constitutes the remaining 5%.
What is the patent landscape and its impact on market competition?
The original patents for Granisetron HCl have largely expired in major markets. This has led to increased generic competition, driving down prices and expanding accessibility. Key patent expirations occurred in the United States around 2005-2008 and in Europe similarly. While some secondary patents related to specific formulations or delivery methods may still exist, the core composition-of-matter patents have lapsed.
- Original Composition-of-Matter Patents: Expired.
- Formulation Patents: Varying expiration dates, some extending into the late 2020s for specific advanced delivery systems.
- Generic Entry: Significant in developed markets since the late 2000s.
This patent expiry has facilitated the entry of multiple generic manufacturers, leading to price erosion and a more fragmented market. The focus for innovation has shifted towards novel delivery mechanisms and combination therapies.
How do generic competition and pricing dynamics influence the Granisetron HCl market?
Generic competition is the most significant factor influencing Granisetron HCl pricing. Following patent expiries, numerous pharmaceutical companies introduced generic versions of Granisetron HCl, creating a highly competitive environment. This has resulted in substantial price reductions compared to the original branded product.
The average selling price (ASP) for generic Granisetron HCl in developed markets has decreased by an estimated 50-70% since the peak of branded sales. The price for a standard course of treatment (e.g., a few intravenous doses or a week's supply of oral tablets) can range from USD 20 to USD 75, depending on the region, manufacturer, and volume. In contrast, the branded product could cost upwards of USD 150-250 for a similar course.
Table 2: Comparative Pricing (Estimated Averages)
| Product Type | Region | Price Range (USD) per Course of Treatment |
|---|---|---|
| Branded Granisetron | US/EU (Pre-2010) | 150 - 250 |
| Generic Granisetron | US/EU (2023/24) | 20 - 75 |
| Generic Granisetron | Emerging Markets | 15 - 50 |
This price pressure compels manufacturers to focus on cost-efficient production, supply chain optimization, and differentiation through product quality or specific market access strategies.
What are the key market segments for Granisetron HCl by formulation and route of administration?
Granisetron HCl is available in several formulations and routes of administration, catering to different clinical needs and patient preferences. These segments exhibit varying market shares and growth potentials.
- Injectable Formulations: Intravenous (IV) and subcutaneous (SC) injections are crucial for rapid onset of action, particularly in hospital settings for CINV. The IV segment is the largest.
- Oral Formulations: Tablets and oral solutions offer convenience for outpatient use and management of delayed-onset nausea.
- Transdermal Patches: The Granisetron transdermal patch provides sustained release over several days, offering a significant advantage for patients undergoing prolonged chemotherapy or radiation. This segment is growing due to its convenience and continuous efficacy.
Table 3: Granisetron HCl Market Share by Formulation (Estimated)
| Formulation | Route of Administration | Estimated Market Share (%) | Growth Potential |
|---|---|---|---|
| Injectable | Intravenous (IV) | 55 | Moderate |
| Injectable | Subcutaneous (SC) | 15 | Moderate |
| Oral | Tablets | 20 | Moderate |
| Oral | Solution | 5 | Low |
| Transdermal Patch | Patch | 5 | High |
Which geographic regions represent the largest markets for Granisetron HCl?
Developed regions, including North America and Europe, currently constitute the largest markets for Granisetron HCl due to a higher incidence of cancer, advanced healthcare infrastructure, and greater access to advanced cancer therapies. However, emerging markets in Asia-Pacific and Latin America are exhibiting robust growth.
- North America (United States, Canada): Dominates due to high cancer rates, comprehensive insurance coverage, and extensive use of combination chemotherapy regimens.
- Europe (Germany, UK, France, Italy, Spain): A significant market with well-established healthcare systems and strong demand for antiemetic supportive care.
- Asia-Pacific (China, India, Japan): The fastest-growing segment, driven by increasing cancer diagnoses, expanding healthcare access, and a growing number of generic manufacturers.
- Latin America (Brazil, Mexico): Shows steady growth due to improving healthcare infrastructure and increasing awareness of supportive care in oncology.
- Rest of the World: Represents a smaller but developing market.
What are the key drivers of growth in the Granisetron HCl market?
Several factors are propelling the growth of the Granisetron HCl market:
- Rising Cancer Incidence: Global cancer rates continue to increase, leading to a larger patient pool requiring oncological treatments that necessitate antiemetic support. The World Health Organization (WHO) projects a substantial rise in cancer cases in the coming decades.
- Advancements in Cancer Treatment: The development of more aggressive and potent chemotherapy and radiotherapy regimens, while improving treatment efficacy, also increases the risk and severity of CINV and RINV. This drives the demand for effective antiemetics like Granisetron HCl.
- Increasing Healthcare Expenditure and Access: Growing healthcare investments, particularly in emerging economies, are improving access to cancer therapies and supportive care, including antiemetics.
- Demand for Improved Quality of Life: Patients and healthcare providers prioritize managing treatment side effects to improve patient compliance, tolerance, and overall quality of life during cancer treatment.
- Growing Geriatric Population: The aging global population is more susceptible to cancer and often undergoes intensive treatments, further boosting the demand for antiemetics.
- Favorable Regulatory Environment for Generics: The established safety and efficacy profile of Granisetron HCl supports its continued use and the approval of generic versions, which enhances market accessibility.
What are the key challenges and restraints for the Granisetron HCl market?
Despite positive growth drivers, the Granisetron HCl market faces several challenges:
- Intense Generic Competition: The presence of numerous generic manufacturers leads to significant price erosion, limiting profit margins for individual companies.
- Development of Novel Antiemetic Agents: Newer classes of antiemetics, such as NK-1 receptor antagonists (e.g., aprepitant) and other selective serotonin receptor antagonists, offer alternative or complementary mechanisms of action, potentially impacting market share for older drugs.
- Stricter Regulatory Scrutiny: Generic drug approvals require adherence to stringent quality and bioequivalence standards, adding to development and manufacturing costs.
- Reimbursement Policies: Variations in healthcare reimbursement policies across different regions can affect the affordability and accessibility of Granisetron HCl.
- Off-Label Use Limitations: While Granisetron HCl is primarily indicated for CINV/RINV, expansion into other indications may be limited by regulatory approvals and clinical evidence.
- Competition from Combination Therapies: Increasingly, antiemetic therapy involves combinations of drugs, including Granisetron HCl with other agents, which can affect the market share of single-agent therapies.
What is the competitive landscape of Granisetron HCl manufacturers?
The competitive landscape is characterized by a mix of established branded pharmaceutical companies (primarily focused on novel formulations or combination products) and a large number of generic manufacturers. Key players in the generic space include companies with significant API manufacturing capabilities and strong distribution networks.
Prominent manufacturers and suppliers include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Torrent Pharmaceuticals Ltd.
- Hetero Drugs Ltd.
- Accord Healthcare Ltd.
- Fresenius Kabi
- Hikma Pharmaceuticals PLC
- Mylan N.V. (now part of Viatris)
These companies compete on price, product quality, supply chain reliability, and market access. Differentiation often comes from offering various pack sizes, specific formulations (e.g., pre-filled syringes), or strong regional presence.
What are the future trends and opportunities in the Granisetron HCl market?
Future trends and opportunities in the Granisetron HCl market are likely to focus on:
- Development of New Delivery Systems: Continued innovation in transdermal patches, long-acting injectables, and orally disintegrating tablets to improve patient convenience and compliance.
- Combination Therapies: Research and development into optimized combination regimens that include Granisetron HCl with other antiemetic agents to achieve superior efficacy with reduced side effects.
- Expansion in Emerging Markets: Significant growth opportunities exist in Asia-Pacific, Latin America, and Africa as healthcare infrastructure improves and awareness of cancer supportive care increases.
- Focus on Cost-Effectiveness: In a price-sensitive generic market, manufacturers will continue to emphasize efficient production and supply chain management to offer competitive pricing.
- Personalized Medicine Approaches: Although still nascent for antiemetics, future trends may explore tailoring antiemetic regimens based on individual patient risk factors and genetic profiles.
- Repurposing and New Indications: Exploration of Granisetron HCl for other indications where 5-HT3 receptors play a role, though this is a more speculative area.
What are the price projections for Granisetron HCl in the next five years?
Granisetron HCl prices are expected to remain relatively stable or experience a slight decline in developed markets due to ongoing generic competition. Price stability will be influenced by demand from a growing cancer patient population.
- Developed Markets (North America, Europe): Prices for generic Granisetron HCl are projected to stabilize between USD 25 and USD 80 per course of treatment. Minor fluctuations may occur based on supply-demand dynamics and new generic entrants.
- Emerging Markets (Asia-Pacific, Latin America): Prices are expected to see a gradual decrease as more local manufacturers enter the market and economies of scale are achieved, potentially falling to USD 15 to USD 45 per course of treatment.
- Transdermal Patch Formulations: These are likely to command a premium compared to oral or standard injectable generics, with prices ranging from USD 50 to USD 120 per treatment course, depending on duration of use and region.
The overall trend will be a continued downward pressure on prices for standard formulations, while novel delivery systems might sustain higher price points for a period before generic competition emerges for those specific formulations as well.
Key Takeaways
- The Granisetron HCl market is characterized by strong demand driven by rising cancer incidence and advancements in chemotherapy.
- Patent expiries have led to intense generic competition, significantly reducing prices and creating a highly competitive landscape.
- Injectable formulations hold the largest market share, but transdermal patches represent a high-growth segment due to patient convenience.
- North America and Europe are the largest markets, with Asia-Pacific showing the fastest growth.
- Future growth will be driven by innovation in delivery systems, expansion in emerging markets, and the development of combination therapies.
- Price projections indicate stabilization in developed markets and a gradual decline in emerging markets for generic formulations, with transdermal patches maintaining a price premium.
FAQs
-
What are the primary side effects associated with Granisetron HCl? Common side effects include headache, constipation, dizziness, and fatigue. Less common side effects can include abdominal pain, diarrhea, and changes in liver function tests.
-
How does Granisetron HCl compare to ondansetron in terms of efficacy? Both Granisetron HCl and ondansetron are effective 5-HT3 receptor antagonists for managing CINV. Clinical studies generally show comparable efficacy, with the choice often depending on physician preference, patient response, cost, and specific formulation availability.
-
Are there any contraindications for Granisetron HCl use? Granisetron HCl is contraindicated in patients with known hypersensitivity to the drug or its components. Caution is advised in patients with cardiac arrhythmias or those receiving concomitant treatment with other QT-prolonging drugs.
-
What is the typical dosage regimen for Granisetron HCl in chemotherapy-induced nausea and vomiting? Dosage varies by formulation and chemotherapy regimen. For IV administration, a common dose is 1 mg given once daily, either as a bolus injection or infusion. Oral doses are typically 1 mg or 2 mg daily. The transdermal patch is usually applied 24 hours before chemotherapy and worn for 7 days.
-
What is the role of Granisetron HCl in managing radiotherapy-induced nausea and vomiting? Granisetron HCl is effective in preventing and treating RINV, similar to its role in CINV. It helps to alleviate the nausea and vomiting that can occur during or after radiation therapy sessions.
Citations
[1] World Health Organization. (n.d.). Cancer. Retrieved from https://www.who.int/news-room/fact-sheets/detail/cancer [2] Various pharmaceutical market research reports (e.g., from Grand View Research, MarketsandMarkets, Mordor Intelligence) analyzing the antiemetics and Granisetron HCl market. (Specific reports vary and are proprietary, thus not directly citable in a universally accessible manner for this context). [3] U.S. Food and Drug Administration (FDA) Orange Book database. (Accessed periodically for patent and exclusivity information). [4] European Medicines Agency (EMA) database. (Accessed periodically for drug approval and patent information).
More… ↓
