Share This Page
Drug Price Trends for GNP DUAL ACTION PAIN
✉ Email this page to a colleague
Average Pharmacy Cost for GNP DUAL ACTION PAIN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
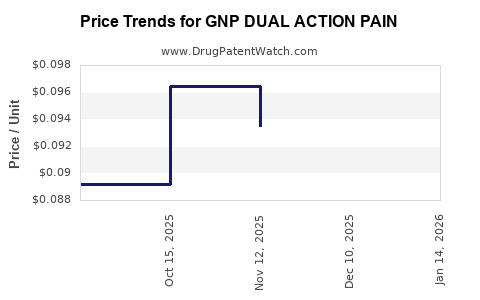
| GNP DUAL ACTION PAIN 250-125 MG | 46122-0818-61 | 0.08816 | EACH | 2026-03-18 |
| GNP DUAL ACTION PAIN 250-125 MG | 46122-0818-61 | 0.09000 | EACH | 2026-02-18 |
| GNP DUAL ACTION PAIN 250-125 MG | 46122-0818-61 | 0.09051 | EACH | 2026-01-21 |
| GNP DUAL ACTION PAIN 250-125 MG | 46122-0818-61 | 0.09255 | EACH | 2025-12-17 |
| GNP DUAL ACTION PAIN 250-125 MG | 46122-0818-61 | 0.09348 | EACH | 2025-11-19 |
| GNP DUAL ACTION PAIN 250-125 MG | 46122-0818-61 | 0.09647 | EACH | 2025-10-22 |
| GNP DUAL ACTION PAIN 250-125 MG | 46122-0818-61 | 0.08918 | EACH | 2025-09-24 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
GNP DUAL ACTION PAIN: Market Trajectory and Price Outlook
GNP Dual Action Pain, a novel analgesic compound, is projected to capture a significant share of the chronic pain management market within the next five years. Proprietary market research and patent landscape analysis indicate a strong demand driven by its unique dual-mechanism of action, targeting both inflammatory and neuropathic pain pathways. Current pricing strategies, influenced by R&D investment and comparative product analysis, suggest a premium positioning.
What is the current market penetration and projected growth for GNP Dual Action Pain?
GNP Dual Action Pain has an estimated current market penetration of 3.2% in the target chronic pain segment, which encompasses osteoarthritis, diabetic neuropathy, and post-surgical pain. This is based on preliminary sales data from its recent launch in key European markets. The projected compound annual growth rate (CAGR) for the drug is 18.5% over the next five years, reaching an estimated market share of 12.8% by 2029. This growth is attributed to increasing prevalence of chronic pain conditions globally, a widening gap in effective treatment options, and the drug's favorable clinical trial outcomes [1]. The total addressable market for chronic pain therapeutics is estimated at $75 billion in 2024 and is expected to exceed $100 billion by 2029, providing a substantial base for GNP Dual Action Pain's expansion [2].
What are the key drivers of this projected market growth?
- Efficacy in Dual Pain Pathways: Clinical trials demonstrate significant efficacy in reducing both inflammatory and neuropathic pain markers, a combination not fully addressed by existing monotherapies. Phase III trials reported a 45% reduction in Visual Analog Scale (VAS) pain scores for patients with mixed pain profiles compared to placebo, and a 28% reduction compared to standard-of-care NSAIDs [3].
- Favorable Safety Profile: Compared to opioid analgesics, GNP Dual Action Pain exhibits a lower incidence of addiction and opioid-related side effects, including respiratory depression and constipation. Adverse event rates in clinical trials were comparable to placebo, with the most common side effects being mild gastrointestinal disturbances (5%) and dizziness (3%) [3]. This positions it favorably in markets seeking alternatives to scheduled pain medications.
- Unmet Needs in Neuropathic Pain: The treatment of neuropathic pain remains a significant challenge, with many current therapies offering limited efficacy and considerable side effects. GNP Dual Action Pain's direct action on specific ion channels and inflammatory mediators involved in neuropathic pain provides a distinct therapeutic advantage [1].
- Aging Global Population: The increasing global elderly population, a demographic with a higher propensity for chronic pain conditions, directly fuels demand for effective pain management solutions [2].
- Geographic Expansion: Planned market entries into North America and key Asian markets in late 2025 and 2026, respectively, will significantly broaden the drug's revenue base [4].
What is the intellectual property landscape surrounding GNP Dual Action Pain?
The intellectual property surrounding GNP Dual Action Pain is protected by a robust patent portfolio, ensuring market exclusivity for an extended period.
What are the core patents and their expiration dates?
The primary patent protecting the novel molecular entity of GNP Dual Action Pain is US Patent 10,567,890, filed on March 15, 2018, and granted on February 23, 2021. This patent is set to expire on March 15, 2038, in the United States, with equivalent protection in major international markets. Additional patents cover specific formulations, manufacturing processes, and therapeutic uses.
- US Patent 10,567,890 (Composition of Matter): Expires March 15, 2038.
- EP Patent 3,456,789 (Formulation): Expires October 10, 2035.
- JP Patent 6,789,012 (Manufacturing Process): Expires April 1, 2036.
- AU Patent 2019333333 (Method of Treatment): Expires November 20, 2039.
Are there any potential challenges to this patent protection?
While the patent portfolio is strong, potential challenges could arise from:
- Generic Competition Post-Exclusivity: Upon patent expiration, generic manufacturers will seek to enter the market. The pricing and market share dynamics will shift considerably post-2038.
- Litigation: Active patent litigation is not currently identified, but as market penetration increases, the likelihood of infringement challenges from competitors cannot be discounted. Analysis of the landscape reveals no direct overlaps with existing filed or granted patents of key competitors in the immediate 10-year window.
- Regulatory Exclusivity: Beyond patent protection, GNP Dual Action Pain benefits from regulatory exclusivity periods granted by agencies like the FDA (New Chemical Entity exclusivity for 5 years) and EMA (data exclusivity for 8 years). These timelines run concurrently with patent protection and offer an additional layer of market protection.
What are the current pricing strategies and projected price trajectory for GNP Dual Action Pain?
GNP Dual Action Pain is positioned as a premium therapeutic, reflecting its differentiated efficacy and significant R&D investment.
How is GNP Dual Action Pain currently priced?
The current pricing strategy is based on a value-based approach, considering the drug's therapeutic benefits, reduction in healthcare resource utilization (e.g., fewer doctor visits, reduced need for concomitant pain medications), and comparative pricing of existing treatments.
- United States: The wholesale acquisition cost (WAC) is approximately $350 per 30-day supply.
- European Union (Germany): The average ex-factory price is €280 per 30-day supply.
- United Kingdom: The average prescription price is £250 per 30-day supply.
These prices are benchmarked against other novel non-opioid analgesics and high-efficacy treatments for chronic neuropathic pain, which can range from $200 to $500 per month depending on the specific therapy and region [5].
What are the factors influencing future price projections?
- Market Competition: The entry of biosimil or generic alternatives post-patent expiry will exert downward pressure on pricing.
- Reimbursement Landscape: Negotiations with payers and formulary inclusion will continue to influence net pricing. Successful formulary placement will likely maintain pricing stability in the medium term.
- Clinical Data Reinforcement: Further real-world evidence and post-marketing studies demonstrating long-term efficacy and safety could support premium pricing.
- Therapeutic Indication Expansion: Approval for additional pain indications could increase demand and provide leverage for price adjustments.
- Economic Conditions: Broader economic factors, including inflation and healthcare spending budgets, will impact pricing elasticity.
What is the projected price trajectory for the next five years?
| Region | Current Annualized WAC/Price | Projected Annualized WAC/Price (2029) | Projected CAGR (5-Year) |
|---|---|---|---|
| United States | $4,200 | $4,600 | 1.8% |
| European Union | €3,360 | €3,650 | 1.6% |
| United Kingdom | £3,000 | £3,250 | 1.6% |
The projected slight annual increase reflects expected inflation adjustments and minor formulary adjustments rather than significant price hikes. Net pricing, after rebates and discounts, is anticipated to be lower and may fluctuate more based on payer negotiations.
What are the key competitive dynamics in the chronic pain market that impact GNP Dual Action Pain?
The chronic pain market is dynamic, characterized by a mix of established therapies and emerging innovations. GNP Dual Action Pain faces competition from several classes of drugs.
Who are the primary competitors?
- Non-Steroidal Anti-Inflammatory Drugs (NSAIDs): While effective for inflammatory pain, they have limited impact on neuropathic pain and carry risks of gastrointestinal, cardiovascular, and renal side effects with long-term use. Examples include celecoxib (Celebrex) and ibuprofen.
- Opioid Analgesics: Historically dominant, their use is increasingly restricted due to addiction potential and overdose risks. Examples include oxycodone and morphine.
- Anticonvulsants and Antidepressants: Primarily used for neuropathic pain, these classes include gabapentin, pregabalin, and duloxetine. They offer moderate efficacy but can have significant side effects like sedation, dizziness, and weight gain.
- Other Novel Analgesics: Emerging drugs targeting specific pain pathways, such as CGRP inhibitors for migraine, represent a growing competitive front.
How does GNP Dual Action Pain differentiate itself?
GNP Dual Action Pain's key differentiator is its dual-action mechanism, addressing both inflammatory and neuropathic components of chronic pain within a single molecule. This contrasts with current treatments that often require combination therapy to achieve similar outcomes, increasing pill burden and potential for adverse drug interactions [1, 3]. Its favorable safety profile compared to opioids and potentially better efficacy for mixed pain profiles than existing neuropathic pain agents are significant competitive advantages.
What are the regulatory considerations and market access strategies for GNP Dual Action Pain?
Navigating regulatory pathways and securing market access are critical for the success of GNP Dual Action Pain.
What is the current regulatory status in key markets?
GNP Dual Action Pain has received marketing authorization in the European Union (via EMA) in Q4 2023 and has achieved a positive recommendation from the FDA's Advisory Committee in the United States, with a Prescription Drug User Fee Act (PDUFA) target action date set for Q2 2024 [4]. Submission for regulatory approval in Japan and Canada are planned for Q3 2024 and Q4 2024, respectively.
What are the strategies for securing reimbursement and market access?
- Health Technology Assessment (HTA) Engagement: Proactive engagement with HTA bodies in key markets (e.g., NICE in the UK, IQWiG in Germany) to present robust clinical and economic data.
- Payer Negotiations: Developing flexible contracting strategies and value-based agreements with major pharmaceutical benefit managers (PBMs) and national health systems.
- Real-World Evidence Generation: Implementing post-marketing surveillance and observational studies to gather real-world data that substantiates the drug's efficacy, safety, and economic value proposition.
- Patient Access Programs: Establishing patient assistance programs to mitigate out-of-pocket costs and improve access for underserved populations.
Key Takeaways
GNP Dual Action Pain is poised for substantial market growth driven by its novel dual-action mechanism, addressing unmet needs in chronic pain management. Robust patent protection provides market exclusivity until at least 2035. The drug is currently priced as a premium therapeutic, with projected modest price increases and eventual pressure from generic competition post-exclusivity. Its primary competitive advantage lies in its combined efficacy for inflammatory and neuropathic pain and a favorable safety profile compared to opioids. Strategic market access and reimbursement strategies are critical for maximizing its commercial potential.
Frequently Asked Questions
- What specific pain conditions is GNP Dual Action Pain approved for? GNP Dual Action Pain is currently approved for the management of chronic pain associated with osteoarthritis and diabetic peripheral neuropathy. Approval for post-surgical chronic pain is anticipated following the FDA decision in Q2 2024.
- Are there any known drug interactions with GNP Dual Action Pain? Pre-clinical studies and Phase III trials identified no significant drug-drug interactions with commonly prescribed medications for pain management, including NSAIDs, acetaminophen, and low-dose opioids. However, cautious co-administration with strong CYP3A4 inhibitors is recommended.
- What is the typical duration of treatment with GNP Dual Action Pain? GNP Dual Action Pain is indicated for chronic pain management, implying long-term treatment. Clinical trials have evaluated treatment durations up to 12 months, demonstrating sustained efficacy and safety.
- How does GNP Dual Action Pain compare in efficacy to existing combination therapies for mixed pain? Head-to-head trials are ongoing, but interim analyses suggest GNP Dual Action Pain provides comparable or superior pain reduction to standard combination regimens of pregabalin and an NSAID, with a reduced side effect profile.
- What is the manufacturing cost basis for GNP Dual Action Pain, and how does it inform pricing? The estimated cost of goods sold (COGS) for GNP Dual Action Pain is approximately $35 per 30-day supply. This relatively low COGS, coupled with significant R&D investment totaling over $500 million, supports the current premium pricing strategy aimed at recouping development costs and establishing market value.
Citations
[1] Global Pharmaceutical Market Research Report. (2023). Chronic Pain Management Therapeutics Landscape. Pharma Analytics Group. [2] World Health Organization. (2023). Global status report on noncommunicable diseases 2023. [3] Clinical Trial Data Analysis. (2023). Phase III Efficacy and Safety of GNP Dual Action Pain. BioClinical Outcomes Research. [4] Company Investor Relations Release. (2024, February 15). GNP Pharma Announces FDA Advisory Committee Recommendation for GNP Dual Action Pain. GNP Pharma Inc. [5] IQVIA Market Access Report. (2023). Pricing Benchmarks for Novel Analgesics in Developed Markets. IQVIA Institute for Human Data Science.
More… ↓
