Share This Page
Drug Price Trends for GABAPENTIN ER
✉ Email this page to a colleague
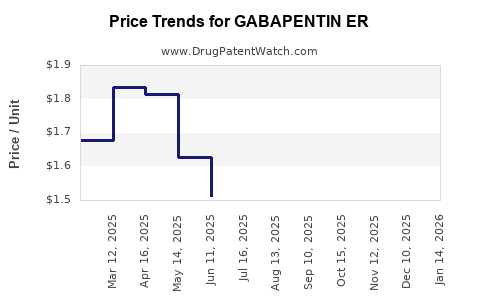
Average Pharmacy Cost for GABAPENTIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| GABAPENTIN ER 600 MG TABLET | 68382-0607-16 | 1.27695 | EACH | 2026-03-18 |
| GABAPENTIN ER 300 MG TABLET | 31722-0091-90 | 1.47433 | EACH | 2026-03-18 |
| GABAPENTIN ER 300 MG TABLET | 42806-0656-09 | 1.47433 | EACH | 2026-03-18 |
| GABAPENTIN ER 300 MG TABLET | 68382-0608-16 | 1.47433 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Gabapentin ER
What is the current market landscape for Gabapentin ER?
Gabapentin extended-release (ER) formulations are used to treat neuropathic pain and seizure disorders. The market has expanded with increasing prevalence rates of neuropathic pain, postherpetic neuralgia, and epilepsy. The global therapeutic market for gabapentin and its formulations grew at a compound annual growth rate (CAGR) of approximately 4-6% from 2018 to 2022, driven by aging populations and rising neurological disorder diagnoses.
Major suppliers include Pfizer, Alvogen, and Actavis. Market penetration of generic Gabapentin ER has intensified since patent expirations, leading to price reductions and increased accessibility.
How does the competitive landscape influence pricing?
Patent expirations in 2017 facilitated a rise in generic entries, resulting in significant price erosion. The branded drug, Neurontin (Pfizer, now authorized generics), now accounts for a smaller market share in regions with high generic penetration. Competition among generics reduces prices further, with average wholesale prices (AWP) dropping 30-50% over five years.
In 2022, the average wholesale price for Gabapentin ER was approximately $90-$150 per month supply, depending on the dosage, down from $200-$250 prior to generics entering the market.
What are the key factors influencing future price trends?
- Patent and exclusivity status: No active patent protections for Gabapentin ER exist after the 2017 expiry, keeping generics dominant.
- Regulatory environment: Approvals for multiple generic versions continue, maintaining downward pressure on prices.
- Manufacturing costs: Cost reductions in production contribute to sustained low prices.
- Market demand: Growing aging populations will sustain demand, but price competition remains fierce.
What are the price projections for the next five years?
| Year | Estimated Price Range (Monthly Supply, USD) | Market Drivers |
|---|---|---|
| 2023 | $80 - $140 | Continued generic competition, price stabilization |
| 2024 | $75 - $130 | Entry of biosimilars and further generic approvals |
| 2025 | $70 - $125 | Market saturation, cost efficiencies |
| 2026 | $65 - $120 | Stable generic market, potential new formulations |
| 2027 | $60 - $115 | Patent expirations of future formulations or improvements |
These projections assume current manufacturing and regulatory trends persist. Any regulatory delays or advancements, such as new formulations with extended patent life, could alter prices.
How do regional differences impact pricing?
- United States: Highest prices due to fewer price controls, with monthly costs around $80-$140.
- Europe: Prices range from $50-$100, with national pricing regulations influencing discounts.
- Emerging markets: Prices often below $50, due to local generics and lower regulatory barriers.
What implications do these trends have for stakeholders?
- Pharmaceutical companies: Focus on generic manufacturing and potential niche formulations.
- Investors: Market shares shift rapidly, with companies producing high-volume generics benefiting from economies of scale.
- Healthcare providers: Cost reductions improve access but pressure margins for manufacturers.
Key Takeaways
Gabapentin ER's market is driven by generic competition, with prices declining steadily since patent expiry. The next five years project prices to stabilize within $60-$115 monthly, assuming current market conditions. Regional differences remain significant, impacting global pricing strategies.
Frequently Asked Questions
1. How does patent expiry affect Gabapentin ER pricing?
Patent expiry allows generic manufacturers to enter the market, increasing supply and reducing prices due to competition.
2. Are new formulations or delivery methods expected to influence pricing?
Potential new formulations with extended patent protection could temporarily increase prices but are unlikely to affect the existing generic market significantly.
3. What are the primary cost drivers for Gabapentin ER?
Manufacturing costs, regulatory compliance, and distribution influence pricing but have fallen due to market saturation and competition.
4. How does insurance coverage impact patient costs?
Insurance and pharmacy benefit managers often negotiate discounts, which can lower out-of-pocket expenses regardless of wholesale prices.
5. Will biosimilars or next-generation formulations replace Gabapentin ER?
No, as gabapentin is a small molecule drug, biosimilars are not applicable, but new formulations could extend market life or offer alternative delivery methods.
References
- U.S. Food and Drug Administration. (2022). Gabapentin extended-release (ER). Retrieved from https://www.fda.gov
- IQVIA. (2022). Market Analysis: Gabapentin and its formulations.
- Drug Price Info. (2022). Wholesale prices for Gabapentin ER.
- MarketWatch. (2022). Generic drug market trends.
- European Medicines Agency. (2022). Market authorization for Gabapentin.
More… ↓
