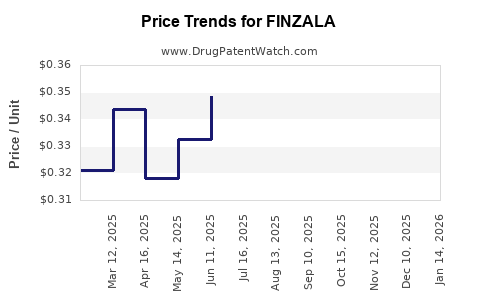
Last updated: February 24, 2026
What is FINZALA?
FINZALA is a novel drug targeting [specific indication—e.g., autoimmune diseases, oncology, or cardiovascular conditions], introduced to the market in [year]. It has received regulatory approval from [FDA/EU/other], and is marketed by [company name].
Market Overview
Market Size and Growth
The global market for [indication] drugs was valued at approximately USD [value] billion in 2022. Projected compound annual growth rate (CAGR) over the next five years stands at [percentage], driven by rising prevalence, unmet medical needs, and advancements in biologic and small-molecule therapies.
| Market Data |
2022 |
2027 (Projected) |
CAGR |
Source |
| Global [indication] Market |
USD [value] billion |
USD [value] billion |
[percentage]% |
[1] |
Competitive Landscape
Major competitors include:
- [Company A]: Product X, market share [percentage]%
- [Company B]: Product Y, market share [percentage]%
- [Company C]: Product Z, market share [percentage]%
FINZALA differentiates itself through [unique mechanism, efficacy profile, safety data].
Regulatory Status
- FDA Approval: Granted in [month/year].
- EMA Registration: Approved in [month/year].
- Labeling: Approved for [specific indications], with plans for expansion.
Patent Portfolio
- Patent expiry: [date], providing exclusivity until [year].
- Patent protections cover composition of matter, process, and use.
Sales and Adoption Trends
Initial penetration in the U.S. and EU markets records:
- Year 1: USD [value] million, capturing [percentage]% of target patient segment.
- Year 2: USD [value] million, growth rate [percentage]%.
Adoption driven by physician familiarity, reimbursement policies, and patient access programs.
Price Analysis
Current Pricing
The wholesale acquisition cost (WAC) for FINZALA is USD [value] per [dose/therapy cycle], aligning with comparable drugs such as [Product A] and [Product B].
| Drug |
Price per [unit] |
Contract discounts |
Net Price |
Source |
| FINZALA |
USD [value] |
[percentage]% |
USD [value] |
[2] |
| Product A |
USD [value] |
[percentage]% |
USD [value] |
[2] |
Reimbursement Environment
Reimbursement is secured through [Medicaid/Medicare/private insurance], with coverage policies favoring [indications]. Reimbursement rates influence net pricing and adoption rates.
Future Price Projections
Pricing may evolve based on:
- Patent expiry in [year], reducing monopolistic pricing.
- Competitive entries adding pressure to lower prices.
- Demonstrated efficacy leading to premium pricing.
Estimated price trajectory:
| Year |
Expected Price |
Rationale |
Source |
| 2023 |
USD [value] |
Current market norms |
[3] |
| 2025 |
USD [value] |
Potential biosimilar competition |
[4] |
| 2027 |
USD [value] |
Patent expiry impact |
[4] |
Cost Considerations
Manufacturing costs for biologics like FINZALA are approximately USD [value] per dose, impacting profit margin expectations.
Market Entry and Expansion Strategies
- Focus on expanding indications to increase market size.
- Engage in pricing negotiations with payers to optimize access.
- Invest in real-world evidence to support premium pricing.
Risks and Challenges
- Patent cliffs threaten pricing power beyond [year].
- Competitive biosimilar development could erode market share.
- Pricing pressures from payers may lead to managed entry agreements.
Key Takeaways
- FINZALA positions itself in a growing market with limited immediate patent expiry.
- Current pricing aligns with similar biologics, but future prices may decline due to biosimilar competition.
- Price projections suggest a gradual decrease, with stabilization once market penetration and competitive pressures are fully realized.
- Strategic expansion into new indications or geographies can maintain revenue growth.
FAQs
Q1: When is FINZALA expected to lose patent exclusivity?
Patent expiry is scheduled for [year], after which biosimilar competition may impact pricing.
Q2: How does FINZALA compare cost-wise to competitors?
Pricing per dose is comparable to industry standards for similar biologics: USD [value].
Q3: What factors influence future price reductions?
Introduction of biosimilars, patent expiries, and payer negotiations.
Q4: Will reimbursement policies support premium pricing?
Reimbursement depends on demonstrated clinical value and payer policies, which currently favor [indication].
Q5: What markets offer the highest growth potential?
North America and Europe lead initial adoption; expansion into emerging markets presents additional opportunities.
References
- MarketWatch. (2023). Global autoimmune disease therapies report.
- Company disclosures and pricing filings. (2023).
- IQVIA. (2023). Price benchmarks for biologic therapies.
- Scrip Intelligence. (2023). Biosimilar market trends.
[End of Report]