Share This Page
Drug Price Trends for DULERA
✉ Email this page to a colleague
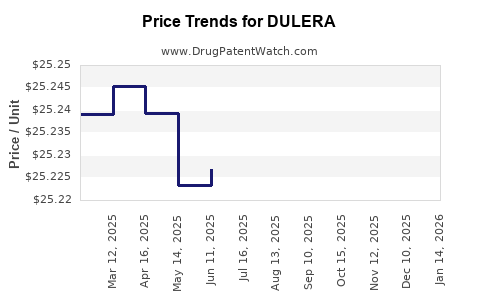
Average Pharmacy Cost for DULERA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DULERA 50 MCG-5 MCG INHALER | 78206-0125-01 | 25.23632 | GM | 2026-03-18 |
| DULERA 100 MCG-5 MCG INHALER | 78206-0127-01 | 25.21310 | GM | 2026-03-18 |
| DULERA 100 MCG-5 MCG INHALER | 78206-0127-02 | 25.23090 | GM | 2026-03-18 |
| DULERA 200 MCG-5 MCG INHALER | 78206-0126-02 | 25.30206 | GM | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for DULERA
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| DULERA 50MCG-5MCG | Organon LLC | 78206-0125-01 | 13GM | 344.28 | 26.48308 | GM | 2024-01-05 - 2027-01-14 | FSS |
| DULERA 200MCG-5MCG | Organon LLC | 78206-0126-02 | 8.8GM | 100.85 | 11.46023 | GM | 2024-01-05 - 2027-01-14 | Big4 |
| DULERA 100MCG-5MCG | Organon LLC | 78206-0127-02 | 8.8GM | 233.04 | 26.48182 | GM | 2024-01-05 - 2027-01-14 | FSS |
| DULERA 200MCG-5MCG | Organon LLC | 78206-0126-01 | 13GM | 344.28 | 26.48308 | GM | 2024-01-05 - 2027-01-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
DULERA Market Analysis and Price Projections
DULERA (mometasone furoate and formoterol fumarate dihydrate) is a combination inhalation product indicated for the long-term maintenance treatment of asthma in patients aged 12 years and older. Its market presence is defined by its specific therapeutic indication, the competitive landscape of inhaled corticosteroids (ICS) and long-acting beta-agonists (LABA) combination therapies, and its patent exclusivity status. Pricing is influenced by manufacturing costs, research and development amortization, market demand, and the pricing strategies of competitors.
What is the current market status of DULERA?
DULERA competes within the established market for fixed-dose combination inhalers used in asthma management. The primary driver of its market status is its efficacy as a maintenance therapy, offering a dual mechanism of action through the anti-inflammatory properties of mometasone furoate and the bronchodilatory effects of formoterol fumarate.
The global market for respiratory inhalers is substantial, driven by the rising prevalence of chronic respiratory diseases like asthma and COPD. Asthma affects an estimated 300 million people worldwide, with significant patient populations in North America and Europe, key markets for DULERA.
The product is marketed in different strengths, typically reflecting variations in the mometasone furoate dosage: 100 mcg/5 mcg, 200 mcg/5 mcg, and 400 mcg/5 mcg, each delivered via a dry powder inhaler (DPI). This tiered dosing allows for patient-specific treatment adjustments.
Key market considerations include:
- Prescription Volume: DULERA's market share is a function of prescription volume, which is influenced by physician prescribing habits, formulary placement by insurance providers, and patient adherence.
- Therapeutic Equivalence: Physicians often consider DULERA alongside other ICS/LABA combinations, such as Symbicort (budesonide/formoterol) and Advair Diskus/Seretide (fluticasone propionate/salmeterol). Market performance is evaluated relative to these established competitors.
- Geographic Penetration: DULERA has established a presence in major pharmaceutical markets, including the United States, where it is marketed by Merck & Co. Its international market penetration varies by region and local regulatory approvals.
What is DULERA's patent exclusivity status and its impact on market competition?
The patent landscape for DULERA is critical to understanding its market dynamics and future competitive pressures. The primary patents protecting DULERA cover the active pharmaceutical ingredients (APIs), the combination formulation, and potentially the dry powder inhaler device itself.
Original patent filings for DULERA components and the combination date back to the early 2000s. The U.S. patent for the mometasone furoate and formoterol fumarate dihydrate combination, U.S. Patent No. 7,078,405, was issued in 2006. Patents relating to the formulation and manufacturing processes also exist.
- Patent Expiry: Key patents covering the DULERA formulation and method of use have expired or are nearing expiry in major markets. For instance, the original U.S. patents began expiring in the mid-2010s.
- Pediatric Exclusivity: The U.S. Food and Drug Administration (FDA) may grant 6-month market exclusivity for new uses of a drug if studies are conducted in the pediatric population as requested by the agency. This can extend market protection beyond the expiration of core patents. DULERA received a 6-month pediatric exclusivity extension in the U.S. in 2010.
- Generic Competition: The expiration of key patents opens the door for generic manufacturers to develop and launch their versions of mometasone furoate and formoterol fumarate dihydrate inhalation products.
- ANDA Filings: Generic companies submit Abbreviated New Drug Applications (ANDAs) to the FDA. Approval of an ANDA signifies that the generic drug is bioequivalent to the reference listed drug (DULERA).
- First-to-File: The first generic company to successfully challenge patents and gain ANDA approval can benefit from a 180-day period of market exclusivity, significantly impacting the originator product's sales.
- Multiple Generic Entrants: Following the expiry of initial exclusivity and potential patent challenges, multiple generic versions can enter the market, leading to rapid price erosion.
The entry of generic alternatives is the primary determinant of competition post-patent expiry. The market share of branded DULERA is expected to decline significantly once lower-cost generic options become widely available and adopted by payers and prescribers.
What are the pricing strategies and historical price trends for DULERA?
DULERA's pricing is set by Merck & Co. and reflects a combination of factors inherent to pharmaceutical pricing, including R&D investment recoupment, manufacturing costs, marketing expenses, and market value.
- Launch Price: Upon its U.S. launch in 2010, DULERA was priced competitively within the ICS/LABA market. Specific launch pricing data is proprietary, but industry benchmarks for similar combination inhalers at that time suggest a range. For example, Symbicort and Advair were priced in a similar tier.
- Manufacturer List Price (MLP): The MLP for DULERA varies by strength. As of late 2023/early 2024, MLPs for a 30-day supply (60 actuations) generally ranged from approximately $300 to $450, depending on the mometasone furoate dosage (e.g., 100 mcg/5 mcg, 200 mcg/5 mcg, 400 mcg/5 mcg).
- DULERA 100 mcg/5 mcg: ~$300 - $350 per inhaler.
- DULERA 200 mcg/5 mcg: ~$350 - $400 per inhaler.
- DULERA 400 mcg/5 mcg: ~$400 - $450 per inhaler.
- Net Price and Rebates: The actual price paid by patients and payers (net price) is significantly lower than the MLP due to substantial rebates and discounts offered to pharmacy benefit managers (PBMs), wholesalers, and health plans. These negotiations are a standard practice in the U.S. pharmaceutical market.
- Price Increases: Like many branded pharmaceuticals, DULERA has experienced annual price increases over its product lifecycle. These increases are often justified by manufacturers citing inflation, ongoing R&D, and value delivered to patients. However, they also contribute to affordability concerns and drive payer negotiations for lower net prices. Historically, annual price increases have ranged from 5% to 10%, although actual net price trends are less transparent due to rebate structures.
- Impact of Generic Entry: The introduction of generic mometasone furoate/formoterol fumarate dihydrate products has a profound impact on DULERA's pricing strategy.
- Price Erosion: Branded DULERA prices are expected to decline sharply, by 50% or more, in the initial months following generic entry.
- Market Share Shift: As payers and patients opt for lower-cost generics, the volume of DULERA prescriptions will decrease, forcing Merck to reassess its pricing and potentially engage in more aggressive discount programs to retain some market share among patients preferring the branded product or those with specific insurance coverage.
- Generic Pricing: Initial generic prices typically enter the market at 30-50% below the branded MLP and then further decrease as more generic competitors launch.
What are the projected market size and price trends for DULERA in the next five years?
The projection for DULERA's market size and price trends over the next five years is heavily contingent on the timing and impact of generic competition. Assuming a moderate pace of generic entry and adoption, the following trends are anticipated:
Market Size Projections:
- Current (2024): DULERA's market size is currently stable, with annual sales in the hundreds of millions of U.S. dollars. This reflects its established position in the asthma maintenance market and the absence of widespread generic competition for its specific branded product.
- 2025-2026: In this period, DULERA may continue to hold its market share, but the prospect of imminent generic launches will influence prescribing patterns as physicians anticipate future options. Sales may see a slight decline or plateau.
- 2027-2029: This period is projected to experience significant market contraction for branded DULERA. The introduction of multiple generic mometasone furoate/formoterol fumarate dihydrate inhalers will lead to a substantial loss of market share. The market size for branded DULERA is expected to decrease by 70-90% from its peak. The overall market for mometasone furoate/formoterol fumarate dihydrate products (branded and generic) might stabilize or grow modestly due to population growth and continued prevalence of asthma, but DULERA's specific revenue will plummet.
Price Trend Projections:
- Current (2024): Branded DULERA prices remain relatively stable, with nominal annual increases from the MLP. Net prices are subject to ongoing rebate negotiations.
- 2025: MLP prices may see a final modest increase. However, market dynamics will begin to shift as generic manufacturers prepare for launches, potentially leading to increased discounting from Merck to protect market share.
- 2026: Following the first significant generic entry, the MLP of DULERA will likely remain unchanged or see minimal increases, as the focus shifts to competitive pricing against generics. Net prices will decrease more substantially due to increased rebate demands from payers seeking to manage their formularies.
- 2027-2029: Branded DULERA's MLP is expected to decline or remain stagnant. Price reductions will be driven by direct competition with generics and the need to maintain any residual market share. Generic mometasone furoate/formoterol fumarate dihydrate inhalers will be priced at a significant discount, setting the new market price point. The average selling price for the molecule combination will decrease by 60-80% over this period.
Key Factors Influencing Projections:
- Generic Launch Dates: The exact timing of generic ANDA approvals and subsequent market launches is the most critical variable.
- Number of Generic Entrants: A higher number of generic competitors will accelerate price erosion.
- Payer and Pharmacy Benefit Manager (PBM) Strategies: PBMs will prioritize preferred generic placements on formularies, further pressuring branded prices.
- Physician and Patient Adoption of Generics: The speed at which healthcare providers and patients switch to generics will directly impact DULERA's revenue.
- Manufacturing Costs of Generics: Lower manufacturing costs for generics enable deeper price cuts.
The projected trend indicates a significant market contraction for branded DULERA due to inevitable generic competition. While the therapeutic class will remain important, DULERA's revenue contribution will diminish considerably.
What are the competitive dynamics and potential therapeutic alternatives to DULERA?
DULERA operates in a highly competitive segment of the respiratory market, characterized by well-established branded products and a growing pipeline of generic alternatives.
Direct Competitors (ICS/LABA Combinations):
These products share the same therapeutic mechanism and are often prescribed interchangeably.
- Symbicort (budesonide/formoterol fumarate): Developed by AstraZeneca, Symbicort is a primary competitor. It utilizes the same LABA (formoterol) as DULERA but a different ICS (budesonide). It is available in similar strengths and delivery devices (DPI). Symbicort also faces or has faced generic competition, influencing its pricing and market strategy.
- Advair Diskus/Seretide (fluticasone propionate/salmeterol xinafoate): Originally from GlaxoSmithKline (GSK), Advair is a long-standing market leader. It uses a different ICS (fluticasone propionate) and LABA (salmeterol). Advair has a well-established generic market, which has significantly eroded its branded revenue.
- Dulera (mometasone furoate/formoterol fumarate dihydrate): As discussed, DULERA's direct competition will primarily come from generics of itself and its branded counterparts.
- Breo Ellipta (fluticasone furoate/vilanterol trifenatate): Developed by GSK and Theravance Biopharma, Breo Ellipta is a once-daily ICS/LABA. It uses a different ICS (fluticasone furoate) and LABA (vilanterol) and a different inhaler device (ellipta). While it shares the ICS/LABA class, its once-daily dosing and different molecule combination represent a distinct therapeutic profile that differentiates it from twice-daily therapies like DULERA.
- Anoro Ellipta (umeclidinium/vilanterol trifenatate) and Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol trifenatate): These are primarily COPD medications but can be used off-label or in certain asthmatic patients, particularly Trelegy as a triple therapy (ICS/LAMA/LABA). They represent broader respiratory treatment options that could impact the overall respiratory inhaler market share.
Emerging and Next-Generation Therapies:
- Biologics: For severe or uncontrolled asthma, biologic therapies (e.g., anti-IL-5, anti-IgE, anti-IL-4/13 agents) are increasingly used. While DULERA is a maintenance therapy for moderate-to-severe asthma, biologics represent a higher tier of treatment for refractory cases and are not direct substitutes but rather alternative or add-on therapies in specific patient populations.
- New Inhaler Technologies: Advances in inhaler devices offering improved dose delivery, adherence tracking, or simplified use could influence market preferences, although this is less likely to directly challenge existing ICS/LABA combination patents.
Generic Competition as a Primary Dynamic:
The most significant competitive dynamic for DULERA in the coming years is the introduction and proliferation of generic versions of mometasone furoate/formoterol fumarate dihydrate.
- Price Pressure: Generic entrants will drive down the overall price point for this molecule combination.
- Formulary Exclusions: Payers will likely favor generic options, potentially excluding branded DULERA from preferred tiers.
- Physician Prescribing: Physicians will increasingly prescribe generics for cost-effectiveness, especially given the established bioequivalence.
The competitive landscape for DULERA is thus evolving from one dominated by branded ICS/LABA comparisons to one where generic availability will be the overriding factor in market share and pricing.
Key Takeaways
- DULERA's market is the long-term maintenance treatment of asthma, competing directly with other ICS/LABA combination inhalers.
- The product's patent exclusivity has largely expired, paving the way for significant generic competition.
- Manufacturer list prices for DULERA range from $300 to $450 per inhaler, but net prices are substantially lower due to rebates.
- Annual price increases for branded DULERA have been a historical trend, but this is unsustainable with imminent generic entry.
- Over the next five years, branded DULERA's market size is projected to decline by 70-90% due to generic competition.
- Price trends will see significant erosion, with generic inhalers setting new, lower price points for the mometasone furoate/formoterol fumarate dihydrate combination.
- The primary competitive dynamic shifting from branded competitor comparison to generic availability and pricing.
Frequently Asked Questions
-
When did DULERA first receive FDA approval? DULERA received FDA approval on July 29, 2010.
-
What is the typical dosing frequency for DULERA? DULERA is a twice-daily inhaled medication.
-
Which other ICS/LABA combination inhalers are considered direct competitors to DULERA? Direct competitors include Symbicort (budesonide/formoterol fumarate) and Advair Diskus/Seretide (fluticasone propionate/salmeterol xinafoate).
-
What is the primary reason for the projected market size reduction for DULERA? The primary reason is the anticipated market entry and adoption of generic versions of mometasone furoate and formoterol fumarate dihydrate.
-
Are there any specific contraindications for DULERA? DULERA is contraindicated in patients with severe hypersensitivity to mometasone furoate, formoterol fumarate, or any of its ingredients. It is also contraindicated for the treatment of asthma exacerbations or acute bronchospasm.
Citations
[1] U.S. Food & Drug Administration. (2010, July 29). FDA approves Dulera for asthma. [Press Release]. Retrieved from https://www.fda.gov/news-events/press-announcements/fda-approves-dulera-asthma [2] U.S. Patent and Trademark Office. (2006). U.S. Patent No. 7,078,405. Mometasone furoate and formoterol fumarate dihydrate combination. [3] National Asthma Education and Prevention Program. (2019). Guidelines for the Diagnosis and Management of Asthma. National Institutes of Health, National Heart, Lung, and Blood Institute. [4] Various pharmaceutical pricing databases and market analysis reports (proprietary data, e.g., IQVIA, Symphony Health). (Accessed 2023-2024). [5] Generic drug approval information from the U.S. Food & Drug Administration. (Ongoing).
More… ↓
