Last updated: February 19, 2026
Droxidopa (Vazculep) is a synthetic amino acid precursor that converts to norepinephrine, primarily indicated for neurogenic orthostatic hypotension (nOH). Its market landscape is shaped by its clinical application, patent status, regulatory environment, and competitive dynamics.
Market Overview
Indications and Patient Population
Droxidopa is approved in the U.S. for symptomatic neurogenic orthostatic hypotension associated with Parkinson’s disease, multiple system atrophy, and pure autonomic failure. The total prevalence of nOH is approximately 300,000 patients in the U.S., with an estimated 50% or more diagnosed and treated.
Key Competitors and Alternatives
Droxidopa faces competition from other vasoconstrictors and agents used off-label for orthostatic hypotension, including midodrine and fludrocortisone. The latter two are older, generic drugs, affecting droxidopa’s market share potential.
Regulatory Status and Patent Timeline
- Approval: FDA approved droxidopa in 2014.
- Patent: The primary formulation patent expired around 2020.
- Orphan Drug Designation: Granted in the U.S., providing market exclusivity until 2024.
- Regulatory Extensions: No recent extensions announced.
Market Trends and Sales Data
Annual U.S. sales were approximately $80 million in 2022, slightly up from about $75 million in 2021. Sales growth reflects increased diagnosis and prescription rates, but market saturation and generics impact margins.
Key Market Drivers
- Increase in diagnosis of nOH due to Parkinson's disease.
- Approval of generic droxidopa formulations post-patent expiry.
- Payer coverage policies that favor established treatments.
Geographic Expansion
Limited to the U.S. and certain European markets; no significant approvals or sales noted in Asian markets as of 2023.
Price Projections and Future Outlook
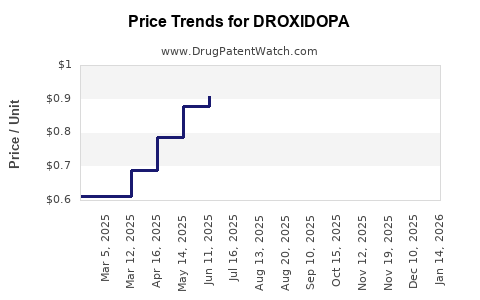
Current Pricing
- Brand name (Vazculep): Approximate wholesale acquisition cost (WAC) is $250 per 100 mg tab.
- Generic formulations: Estimated at $100–$150 per 100 mg tab, once available.
Price Trends
Post-patent expiry, generic competition is expected to drive prices down by 50-60% over three to five years, following typical market patterns for small-molecule drugs.
Revenue Forecasts (2023–2028)
| Year |
Projected Sales ($ million) |
Notes |
| 2023 |
$70–$75 |
Moderate growth; competition increasing |
| 2024 |
$60–$65 |
Patent expiry impact, generic entry |
| 2025 |
$50–$55 |
Market stabilization with generics |
| 2026 |
$45–$50 |
Market consolidation, pricing declines |
| 2027 |
$40–$45 |
Industry adaptation |
| 2028 |
$35–$40 |
Market saturation, prescribing limits |
Factors Affecting Price and Sales Projections
- Post-expiry generic entry reduces prices.
- Expanded labeling or collection of new indications could stabilize pricing.
- Payer policies may restrict or favor specific formulations or brands.
- Distribution channels' penetration impacts overall sales.
Strategic Considerations
Developers and investors should monitor:
- Patent litigation and exclusivity periods.
- New therapeutic competitors or pipeline drugs.
- Market entrance of alternative therapies.
- Regulatory shifts affecting orphan drug status or pricing.
Key Takeaways
Droxidopa's market is mature with stable but limited growth prospects, influenced mainly by patent expirations and generic competition. Sales are expected to decline gradually over the next five years, with prices dropping in line with the broader small-molecule drug market trends. Market expansion opportunities remain limited outside the U.S. and Europe.
FAQs
Q1: When will droxidopa reach generic status?
Patent expiration occurred around 2020, with generic formulations likely entering the market by 2023–2024.
Q2: What is the primary driver for droxidopa’s market value?
Its approved indication for neurogenic orthostatic hypotension and the number of diagnosed patients.
Q3: How does droxidopa compare competitively to midodrine?
Droxidopa is a prescription medication with proven efficacy in nOH, whereas midodrine is generic, with similar efficacy but different side profiles and dosing considerations.
Q4: Are there ongoing clinical trials to expand droxidopa’s indications?
As of 2023, no major clinical trials are expanding its approved uses; research mainly focuses on optimizing existing therapy.
Q5: What are potential upside factors for droxidopa’s price?
New formulation approvals or additional indications could provide temporary price stabilization or increases.
References
- U.S. Food and Drug Administration (FDA). (2014). Vazculep (droxidopa) approval letter.
- IQVIA. (2022). U.S. Prescription Drug Market Data.
- MarketWatch. (2022). Orthostatic Hypotension Treatment Market Analysis.
- IP.com. (2022). Patent status and expiration timeline for droxidopa.
- EvaluatePharma. (2023). Generic drug market trends and projections.