Share This Page
Drug Price Trends for DIMETHYL FUMARATE DR
✉ Email this page to a colleague
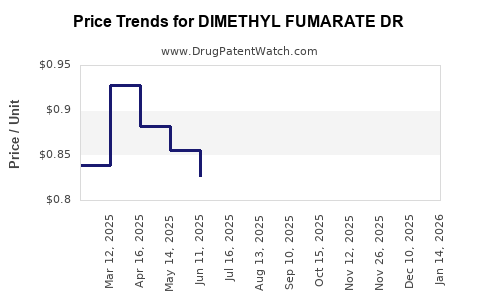
Average Pharmacy Cost for DIMETHYL FUMARATE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| DIMETHYL FUMARATE DR 120 MG CP | 43598-0429-52 | 0.89031 | EACH | 2026-03-18 |
| DIMETHYL FUMARATE DR 120 MG CP | 31722-0657-31 | 0.89031 | EACH | 2026-03-18 |
| DIMETHYL FUMARATE DR 120 MG CP | 16729-0416-04 | 0.89031 | EACH | 2026-03-18 |
| DIMETHYL FUMARATE DR 240 MG CP | 82249-0747-60 | 0.51814 | EACH | 2026-03-18 |
| DIMETHYL FUMARATE DR 120 MG CP | 33342-0349-94 | 0.89031 | EACH | 2026-03-18 |
| DIMETHYL FUMARATE DR 120 MG CP | 24979-0127-21 | 0.89031 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Dimethyl Fumarate Drug Market Analysis and Price Projections
Dimethyl fumarate (DMF) is a small molecule drug approved for the treatment of relapsing forms of multiple sclerosis (MS) and has seen consistent market growth driven by its efficacy and established safety profile. Current market forecasts indicate sustained expansion, with price projections reflecting an evolving competitive landscape and manufacturing cost dynamics.
What is the Current Market Size and Growth Trajectory for Dimethyl Fumarate?
The global market for dimethyl fumarate is substantial and projected to continue its upward trend. Valued at approximately USD 3.8 billion in 2023, the market is forecast to expand at a compound annual growth rate (CAGR) of 5.2% through 2030, reaching an estimated USD 5.3 billion (1). This growth is primarily attributed to the increasing prevalence of multiple sclerosis worldwide and the drug's established position as a first-line treatment option.
Key market drivers include:
- Rising MS Incidence: Global estimates suggest that over 2.3 million people worldwide live with multiple sclerosis, with new diagnoses occurring annually (2). This growing patient population directly translates to increased demand for effective therapies like DMF.
- Treatment Guidelines: Leading neurological associations continue to recommend DMF as a foundational therapy for relapsing-remitting MS (RRMS) due to its favorable benefit-risk profile (3).
- Generic Competition: The availability of multiple generic versions of DMF since its patent expiration has increased accessibility and affordability, contributing to broader market penetration.
- Research and Development: Ongoing research exploring DMF’s potential in other inflammatory and autoimmune conditions, such as psoriasis and rheumatoid arthritis, could further expand its therapeutic applications and market size.
What are the Key Market Segments for Dimethyl Fumarate?
The dimethyl fumarate market can be segmented by application and distribution channel.
By Application
- Multiple Sclerosis (MS): This is the dominant application segment, accounting for over 90% of the current market. DMF is approved for relapsing forms of MS, including relapsing-remitting MS (RRMS), secondary progressive MS (SPMS) with active relapse, and clinically isolated syndrome (CIS) (4).
- Psoriasis: DMF is also approved in some regions for the treatment of moderate to severe plaque psoriasis. While a smaller segment, its contribution to market revenue is growing.
- Other Inflammatory Conditions: Emerging research is investigating DMF's efficacy in other autoimmune diseases, such as Crohn's disease and rheumatoid arthritis. These represent potential future growth avenues.
By Distribution Channel
- Hospital Pharmacies: A significant channel, particularly for initial treatment initiation and inpatient care.
- Retail Pharmacies: The primary channel for ongoing patient prescription fulfillment and chronic disease management.
- Online Pharmacies: Growing in prominence, offering convenience and potentially competitive pricing.
What is the Competitive Landscape for Dimethyl Fumarate Products?
The competitive landscape for dimethyl fumarate is characterized by the presence of both originator brands and a robust generic market.
Key Market Players and Their Products:
| Brand Name | Manufacturer(s) | Indication(s) |
|---|---|---|
| Tecfidera® | Biogen Inc. | Relapsing forms of MS |
| Vumerity® (Diroximel Fumarate) | Alkermes plc | Relapsing forms of MS (a prodrug of monomethyl fumarate, closely related to DMF's mechanism) |
| Dimethyl Fumarate (Various) | Numerous generic manufacturers (e.g., Mylan, Teva, Sun Pharma, Dr. Reddy's) | Relapsing forms of MS, Plaque Psoriasis (depending on specific product approval) |
Market Dynamics:
- Orphan Drug Status: Initially, DMF benefited from orphan drug designation for MS in various regions, providing market exclusivity.
- Patent Expirations and Generic Entry: The expiry of key patents for Tecfidera® has led to the widespread availability of generic DMF. This has significantly increased market competition and driven down prices for the generic formulations.
- Brand Loyalty vs. Cost-Effectiveness: While originator brands may retain some market share due to established physician and patient relationships, the cost-effectiveness of generics is a major driver for market adoption.
- Emergence of Prodrugs: The development of prodrugs like diroximel fumarate (Vumerity®), which aim to improve tolerability by reducing gastrointestinal side effects, introduces another layer of competition within the DMF therapeutic class.
What are the Price Projections for Dimethyl Fumarate?
Price projections for dimethyl fumarate are influenced by the interplay of generic competition, manufacturing costs, and evolving market demand.
Current Pricing Trends:
- Originator Brand (Tecfidera®): Before generic entry, Tecfidera® was priced in the range of USD 70,000 to USD 80,000 annually per patient.
- Generic DMF: Following generic launches, the annual cost for generic DMF formulations has significantly decreased, typically ranging from USD 7,000 to USD 20,000 per patient, depending on the manufacturer, dosage, and insurance coverage. This represents a reduction of over 80% from the originator brand's price.
- Diroximel Fumarate (Vumerity®): As a newer product with specific advantages, Vumerity® is priced competitively, generally within the range of USD 75,000 to USD 85,000 annually per patient, similar to the initial pricing of Tecfidera®.
Price Projections (2024-2030):
The market for DMF is expected to stabilize in terms of price for generic formulations, with incremental adjustments rather than dramatic shifts.
- Generic DMF: Prices for generic DMF are anticipated to remain relatively stable through 2030, likely within the USD 7,000 to USD 18,000 annual per-patient range. Minor fluctuations may occur due to intense competition among generic manufacturers, economies of scale in production, and raw material cost variations. Any significant price decreases would likely be marginal, driven by further manufacturing efficiencies.
- Originator Brand (Tecfidera®): Biogen may maintain a premium for Tecfidera® based on brand recognition and established patient support programs, but its market share is expected to continue to be challenged by generics. Its pricing is unlikely to see substantial increases, given the competitive environment.
- Diroximel Fumarate (Vumerity®): Pricing for Vumerity® will likely remain at a premium compared to generic DMF, reflecting its unique formulation and perceived benefits. However, it may face downward pricing pressure as it matures in the market and faces potential future generic competition (if its patent portfolio differs significantly from original DMF).
Factors Influencing Future Pricing:
- Manufacturing Efficiency: Advancements in synthesis and formulation techniques can reduce production costs, potentially leading to lower wholesale prices for generics.
- Supply Chain Dynamics: Global supply chain stability and the cost of active pharmaceutical ingredients (APIs) will impact production costs.
- Regulatory Policies: Government pricing regulations and negotiation strategies by national health systems can influence the effective prices paid.
- Competition from New Therapies: The development and market entry of novel MS treatments with superior efficacy or tolerability profiles could exert downward pressure on DMF prices.
- Geographic Market Variations: Pricing will continue to vary significantly by region based on local market conditions, reimbursement policies, and the intensity of generic penetration.
What are the Key Manufacturing and Supply Chain Considerations?
The manufacturing of dimethyl fumarate involves established chemical synthesis processes. Key considerations include:
- API Sourcing: The primary raw material is fumaric acid, which is widely available. However, the quality and consistency of the specific intermediates and the final API are critical for regulatory approval and product efficacy.
- GMP Compliance: All manufacturing facilities must adhere to strict Good Manufacturing Practices (GMP) standards set by regulatory bodies like the FDA and EMA to ensure product quality, safety, and consistency.
- Formulation Technologies: DMF is typically formulated into delayed-release capsules. Manufacturers may employ proprietary technologies to ensure the optimal release profile and patient tolerability.
- Scale of Production: With the significant demand, manufacturers operate at scale to meet global needs. Efficiency in large-scale production is crucial for cost competitiveness, especially in the generic market.
- Regulatory Approvals: Manufacturers require individual country approvals for their DMF products, necessitating extensive documentation and adherence to local regulatory requirements.
- Supply Chain Security: Ensuring a robust and secure supply chain is paramount to prevent drug shortages, particularly for chronic disease medications. This involves managing relationships with API suppliers, contract manufacturing organizations (CMOs), and distribution networks.
What are the Regulatory and Patent Landscape Dynamics?
The regulatory and patent landscape has been a critical factor in the market evolution of dimethyl fumarate.
Key Regulatory Approvals:
- United States (FDA): Tecfidera® was first approved by the FDA in March 2013 for relapsing forms of MS. Generic versions of DMF were subsequently approved.
- European Union (EMA): DMF received marketing authorization from the EMA in December 2013 for relapsing-remitting MS and later for plaque psoriasis.
- Other Jurisdictions: Approvals in Canada, Australia, and other major markets followed, with varying timelines.
Patent Expirations and Litigation:
- The primary patents for Biogen's Tecfidera® have expired in major markets, paving the way for generic manufacturers to enter.
- Patent litigation has been a common feature of the market, with originator companies often defending their intellectual property against generic challenges. These legal battles can influence the timing of generic launches.
- The patent expiry dates for Tecfidera® generally occurred between 2020 and 2023 in key markets, leading to the significant increase in generic availability observed since then.
Future Patent Considerations:
- While the core DMF patents have expired, manufacturers may hold patents on specific polymorphic forms, improved formulations, or novel manufacturing processes that could extend market exclusivity for certain niche products.
- Prodrugs like diroximel fumarate have their own distinct patent portfolios, providing them with a period of market exclusivity separate from the original DMF patents.
Key Takeaways
The dimethyl fumarate market is characterized by strong demand driven by the prevalence of multiple sclerosis and its established therapeutic role. The market has transitioned from a branded monopoly to a highly competitive generic landscape, leading to substantial price reductions for generic formulations. Future price projections indicate stability for generics, with continued competition among manufacturers. The regulatory environment, particularly patent expirations, has been the primary catalyst for the market's evolution.
Frequently Asked Questions
-
What is the typical daily dosage of generic dimethyl fumarate for multiple sclerosis? Generic dimethyl fumarate is typically prescribed as a delayed-release capsule. The standard maintenance dosage for relapsing forms of MS is 240 mg twice daily (5). Patients often start with a lower dose to assess tolerability.
-
Are there significant differences in efficacy between branded dimethyl fumarate (Tecfidera®) and its generic versions? No. Generic drugs are required to demonstrate bioequivalence to their branded counterparts, meaning they deliver the same amount of active ingredient into the bloodstream over the same period. Therefore, generic DMF is expected to have the same efficacy and safety profile as Tecfidera®.
-
What are the most common side effects associated with dimethyl fumarate therapy? The most common side effects include flushing, gastrointestinal issues (nausea, diarrhea, abdominal pain), and lymphopenia (a decrease in the number of lymphocytes). These are generally manageable, and dose titration can help mitigate them (4).
-
How does diroximel fumarate (Vumerity®) differ from dimethyl fumarate in terms of tolerability? Diroximel fumarate is a prodrug that is rapidly converted to monomethyl fumarate (MMF), the active metabolite of DMF. Clinical trials suggest that diroximel fumarate may be associated with a lower incidence of gastrointestinal side effects and flushing compared to DMF, potentially due to its different metabolic pathway and absorption characteristics (6).
-
Will the price of generic dimethyl fumarate continue to decrease significantly in the coming years? While marginal decreases may occur due to ongoing manufacturing efficiencies and intense competition, significant further price reductions are unlikely. The market has already experienced the primary price compression associated with the shift from branded to generic status. Future price movements will likely be influenced more by production costs, supply chain stability, and broader market demand.
Citations
[1] Grand View Research. (2024). Dimethyl Fumarate Market Size, Share & Trends Analysis Report By Application (Multiple Sclerosis, Psoriasis), By Distribution Channel, By Region, And Segment Forecasts, 2024 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/dimethyl-fumarate-market
[2] National MS Society. (n.d.). About MS. Retrieved from https://www.nationalmssociety.org/What-is-MS/About-MS
[3] National Institute for Health and Care Excellence. (2020). Multiple sclerosis in adults: management. NICE guideline [NG145]. Retrieved from https://www.nice.org.uk/guidance/ng145
[4] U.S. Food and Drug Administration. (2013, March 27). FDA approves Tecfidera (dimethyl fumarate) for relapsing forms of multiple sclerosis. FDA News Release. Retrieved from https://www.fda.gov/drugs/postmarket-drug-safety-information-for-patients-and-providers/drug-safety-)))))
[5] Mylan (Viatris). (2023). Dimethyl Fumarate Delayed Release Capsules, USP 120 mg and 240 mg. Prescribing Information.
[6] Alkermes plc. (2019). Vumerity (diroximel fumarate) Prescribing Information. Retrieved from https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/211970s000lbl.pdf
More… ↓
