Share This Page
Drug Price Trends for CLOCORTOLONE PIVALATE
✉ Email this page to a colleague
Average Pharmacy Cost for CLOCORTOLONE PIVALATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
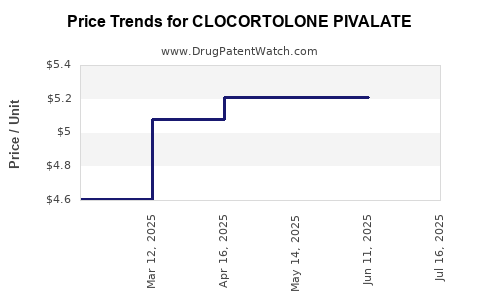
| CLOCORTOLONE PIVALATE 0.1% CRM | 51672-4166-06 | 5.21049 | GM | 2025-07-23 |
| CLOCORTOLONE PIVALATE 0.1% CRM | 51672-4166-06 | 5.21049 | GM | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CLOCORTOLONE PIVALATE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CLOCORTOLONE PIVALATE 0.1% CREAM,TOP | Golden State Medical Supply, Inc. | 51672-4166-06 | 45GM | 128.06 | 2.84578 | GM | 2023-06-15 - 2028-06-14 | FSS |
| CLOCORTOLONE PIVALATE 0.1% CREAM,TOP | Golden State Medical Supply, Inc. | 51672-4166-06 | 45GM | 131.12 | 2.91378 | GM | 2023-06-23 - 2028-06-14 | FSS |
| CLOCORTOLONE PIVALATE 0.1% CREAM,TOP | Golden State Medical Supply, Inc. | 51672-4166-08 | 90GM | 264.99 | 2.94433 | GM | 2023-06-15 - 2028-06-14 | FSS |
| CLOCORTOLONE PIVALATE 0.1% CREAM,TOP | Golden State Medical Supply, Inc. | 51672-4166-08 | 90GM | 271.32 | 3.01467 | GM | 2023-06-23 - 2028-06-14 | FSS |
| CLOCORTOLONE PIVALATE 0.1% CREAM,TOP | Prasco, LLC | 66993-0947-48 | 45GM | 248.80 | 5.52889 | GM | 2021-07-01 - 2026-06-30 | FSS |
| CLOCORTOLONE PIVALATE 0.1% CREAM,TOP | Prasco, LLC | 66993-0947-93 | 90GM | 288.40 | 3.20444 | GM | 2021-07-01 - 2026-06-30 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
CLOCORTOLONE PIVALATE: PATENT LANDSCAPE AND PRICING PROJECTIONS
Clocortolone pivalate is a potent topical corticosteroid indicated for the treatment of inflammatory skin conditions. This analysis examines its current patent landscape, market performance, and future pricing trajectory.
WHAT ARE THE KEY PATENT PROTECTIONS FOR CLOCORTOLONE PIVALATE?
The patent landscape for clocortolone pivalate is characterized by an original composition of matter patent and subsequent formulation and method of use patents. The initial patent, U.S. Patent 3,940,474, filed on October 21, 1974, and granted on February 24, 1976, claimed the compound clocortolone pivalate itself. This foundational patent has long since expired, opening the door for generic competition.
Subsequent patent filings focused on specific formulations and delivery methods. For instance, patents related to specific cream or ointment bases, or combinations with other active ingredients, have provided extended protection for specific product iterations. These secondary patents often target improved stability, efficacy, or patient compliance. The expiration dates of these later patents are critical for understanding the timeline of enhanced market exclusivity.
A review of the U.S. Patent and Trademark Office (USPTO) database reveals several patents that mention clocortolone pivalate. Key patent families and their general expiration timelines illustrate the historical protection:
- Composition of Matter: U.S. Patent 3,940,474 (Expired). This patent covered the chemical entity clocortolone pivalate.
- Formulation Patents: Various patents filed throughout the 1980s and 1990s addressed specific pharmaceutical compositions (e.g., creams, ointments) containing clocortolone pivalate. These patents generally expired between the late 1990s and mid-2000s.
- Method of Use Patents: Patents claiming specific therapeutic applications or treatment regimens for clocortolone pivalate have also existed. Their expiration timelines vary based on their filing and grant dates.
While the core compound is off-patent, a manufacturer can still achieve market differentiation and extend exclusivity through innovative formulations or delivery systems, provided these are covered by unexpired patents. The absence of significant new patent filings specifically related to novel clocortolone pivalate derivatives suggests a mature patent lifecycle for the active pharmaceutical ingredient (API).
WHAT IS THE CURRENT MARKET STATUS OF CLOCORTOLONE PIVALATE?
Clocortolone pivalate is marketed globally under various brand names and as a generic product. Its primary indication is for the relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
Key Market Segments:
- Prescription Dermatology: This is the primary market for clocortolone pivalate, where it competes with other topical corticosteroids of varying potencies.
- Generic Market: Following patent expiries, generic versions of clocortolone pivalate formulations are widely available, significantly impacting pricing dynamics.
Market Performance Indicators:
- Sales Volume: While precise global sales figures for clocortolone pivalate alone are not readily available due to proprietary data and its aggregation within broader corticosteroid market reports, its continued availability as a prescription and generic option indicates consistent demand.
- Competitive Landscape: Clocortolone pivalate is positioned within the mid-to-high potency corticosteroid class. It competes with drugs such as triamcinolone acetonide, betamethasone valerate, and mometasone furoate. The efficacy and side effect profile of clocortolone pivalate influences its market share against these competitors.
- Prescription Trends: Analysis of prescription data from sources like IQVIA or national prescription databases would reveal prescribing patterns, growth rates, and geographical variations. Generic penetration in the U.S. market, for example, has historically led to a decline in brand-name sales for older drugs once exclusivity lapses.
The market for topical corticosteroids is mature, with high levels of genericization for many older APIs. The primary drivers for market share in this segment are price, physician preference, and payer formularies.
HOW ARE CLOCORTOLONE PIVALATE FORMULATIONS PRICED?
The pricing of clocortolone pivalate is bifurcated, reflecting its status as both a branded and a generic pharmaceutical.
Branded Product Pricing:
Historically, branded clocortolone pivalate products commanded premium pricing, reflecting R&D investment, manufacturing costs, marketing, and the exclusivity period granted by patents. Manufacturer-set wholesale acquisition costs (WAC) and net prices after rebates and discounts to payers and pharmacies determine the final patient or insurer cost. Brand pricing strategies aim to capture value based on perceived efficacy, convenience, and patient outcomes.
Generic Product Pricing:
The introduction of generic clocortolone pivalate formulations leads to significant price erosion. Generic manufacturers, leveraging bioequivalence to the branded product and operating with lower overheads, offer products at substantially reduced prices. The number of generic manufacturers in the market directly influences price competition. A fragmented generic market with multiple suppliers typically results in lower prices due to intense competition.
Factors Influencing Pricing:
- Potency and Formulation: While clocortolone pivalate is a potent corticosteroid, its specific formulation (e.g., cream, ointment, lotion, specific concentration) influences its cost. More complex or novel formulations might carry a higher price point, even in generic form, if they offer specific advantages.
- Competition: The number of therapeutic alternatives, both corticosteroids and non-corticosteroids, directly impacts pricing power. A crowded therapeutic space generally leads to downward price pressure.
- Payer Influence: Pharmacy benefit managers (PBMs) and insurance companies negotiate significant discounts and rebates with manufacturers. These negotiations are critical in determining the formulary status of a drug and its effective price paid by payers. Tier placement on formularies (e.g., preferred brand, generic, non-preferred) significantly influences prescribing patterns and out-of-pocket costs for patients.
- Manufacturing Costs: The cost of API synthesis, formulation, packaging, and distribution are fundamental cost drivers. Economies of scale in manufacturing can lead to lower per-unit costs.
- Regulatory Environment: Pricing is also influenced by regulatory policies, such as those governing drug approval, manufacturing standards, and reimbursement.
Price Projections:
Given the mature patent lifecycle of clocortolone pivalate and the established presence of generic alternatives, significant price increases for the API or standard generic formulations are unlikely.
- Branded Products: For any remaining branded formulations, pricing will likely remain stable or subject to modest annual increases aligned with general pharmaceutical inflation, subject to payer negotiations. However, the market share for branded versions is expected to continue to decline as generic options dominate.
- Generic Products: Generic clocortolone pivalate pricing will continue to be driven by competitive market dynamics. Prices are expected to remain low, with potential for further marginal decreases as new generic manufacturers enter the market or existing ones optimize their production costs. Prices for generics typically fall within a narrow range, often differing by only a few percentage points between suppliers. The average wholesale price (AWP) for a 30-gram tube of 0.05% clocortolone pivalate cream, for example, might range from $15 to $30 in the generic market, depending on the manufacturer and pharmacy.
Example Pricing Comparison (Hypothetical Generic Market Data):
| Manufacturer | Formulation | Strength | Pack Size | Average WAC (USD) |
|---|---|---|---|---|
| Generic Pharma A | Cream | 0.05% | 30g | 22.50 |
| Generic Pharma B | Ointment | 0.05% | 30g | 25.00 |
| Generic Pharma C | Cream | 0.05% | 60g | 38.00 |
| Generic Pharma D | Lotion | 0.05% | 60mL | 42.00 |
Note: WAC represents Wholesale Acquisition Cost and is a reference price. Actual prices paid by pharmacies and patients vary based on discounts, rebates, and insurance coverage.
The pricing trajectory for clocortolone pivalate will be characterized by the continued dominance of the generic market, with stable or declining prices for standard formulations, and any remaining branded products facing pressure to justify their premium through differentiated value.
WHAT ARE THE KEY THERAPEUTIC ADVANTAGES AND DISADVANTAGES OF CLOCORTOLONE PIVALATE?
Clocortolone pivalate, as a synthetic corticosteroid, offers specific therapeutic benefits and inherent limitations when compared to other agents in its class and alternative treatment modalities.
Therapeutic Advantages:
- Potency: Clocortolone pivalate is classified as a high-potency corticosteroid. This means it provides potent anti-inflammatory, immunosuppressive, and vasoconstrictive effects. This high potency is beneficial for treating severe or resistant inflammatory skin conditions such as psoriasis, eczema, and dermatitis.
- Efficacy in Specific Conditions: Studies have demonstrated its efficacy in reducing erythema, edema, and pruritus associated with various dermatoses. Its vasoconstrictive properties can lead to rapid visual improvement in skin lesions.
- Formulation Variety: It is available in various formulations, including creams, ointments, and lotions. This allows for tailored treatment based on the nature of the skin lesion (e.g., ointments for dry, scaly lesions; lotions for hairy areas or weeping lesions).
- Established Safety Profile (when used appropriately): As a widely used corticosteroid, its general safety profile, including potential side effects, is well-documented. When used for short durations and in appropriate areas of the body, it is generally safe.
Therapeutic Disadvantages:
- Risk of Systemic Absorption and Side Effects: Due to its high potency, there is a risk of systemic absorption, particularly with prolonged use over large surface areas or under occlusion. This can lead to hypothalamic-pituitary-adrenal (HPA) axis suppression, Cushing's syndrome, hyperglycemia, and other systemic effects.
- Local Side Effects: Topical application can cause local side effects, including skin atrophy (thinning), striae (stretch marks), telangiectasias (dilated blood vessels), acneiform eruptions, and increased susceptibility to infections (bacterial, fungal, viral).
- Rebound Phenomenon: Discontinuation of potent topical corticosteroids can sometimes lead to a rebound flare of the underlying condition. Gradual tapering of the medication is often recommended.
- Limited Use in Certain Areas: High-potency corticosteroids like clocortolone pivalate are generally used with caution or avoided on the face, genitals, and intertriginous areas (skin folds) due to the higher risk of adverse effects.
- Need for Prescription: Clocortolone pivalate is a prescription-only medication, requiring physician oversight for its appropriate use.
Compared to lower-potency corticosteroids, clocortolone pivalate offers faster and more robust symptom relief for severe conditions but carries a higher risk profile. Its use necessitates careful risk-benefit assessment by the prescribing physician, considering the patient's condition, location of lesions, duration of treatment, and potential for systemic exposure.
WHAT ARE THE FUTURE MARKET PROSPECTS FOR CLOCORTOLONE PIVALATE?
The future market prospects for clocortolone pivalate are largely defined by its current market position as a genericized, high-potency topical corticosteroid.
Market Trends:
- Dominance of Generic Market: The market will continue to be dominated by generic formulations. The demand will be driven by cost-effectiveness and physician familiarity with its efficacy.
- Stable, Low-Volume Demand: Demand for clocortolone pivalate is expected to remain stable but likely at low volumes compared to blockbuster drugs. It serves a specific niche for moderate to severe corticosteroid-responsive dermatoses where higher potency is required.
- Competition from Newer Agents: The development of novel therapies for inflammatory skin conditions, including biologics, small molecules targeting specific inflammatory pathways, and non-steroidal topical agents, could gradually erode the market share of traditional topical corticosteroids, including clocortolone pivalate, in some patient populations. However, these newer agents are often significantly more expensive and may not be suitable for all patients or all conditions.
- Limited Innovation Potential: Given the mature API patent status, significant innovation in terms of new clocortolone pivalate derivatives or novel drug delivery systems is unlikely. Any future product enhancements would likely focus on optimizing existing formulations for stability, patient compliance, or combination therapy, which would require new intellectual property protection.
- Impact of Payer Policies: Formulary decisions by payers will continue to influence prescribing patterns. Generic clocortolone pivalate is likely to remain a preferred option for many payers due to its low cost, provided it meets efficacy benchmarks against alternatives.
Growth Projections:
- Low Single-Digit Growth (or Decline): The market for clocortolone pivalate is not anticipated to experience significant growth. A low single-digit percentage growth, or even a slight decline, is more probable as newer treatment paradigms emerge and generic competition remains intense. Growth, if any, will likely be driven by an aging population with increasing prevalence of dermatological conditions and cost-conscious healthcare systems that favor established generics.
- Geographic Variations: Growth rates may vary by region, influenced by local healthcare policies, reimbursement structures, and the availability of alternative treatments. Emerging markets might see some growth as access to basic dermatological treatments expands.
Risks and Opportunities:
- Risks:
- Increased competition from other generic corticosteroids.
- Adoption of alternative, potentially safer or more targeted therapies for chronic inflammatory skin diseases.
- Stricter regulatory guidelines regarding corticosteroid use and potential for adverse events.
- Opportunities:
- Potential for fixed-dose combinations with other topical agents (e.g., antifungals, antibiotics) if novel IP can be secured and clinical benefits demonstrated.
- Continued use in specific patient populations where cost and established efficacy are paramount.
- Leveraging manufacturing efficiencies to maintain a competitive edge in the generic market.
Overall, the market for clocortolone pivalate is expected to be stable and largely driven by its utility as a cost-effective, potent topical corticosteroid for specific dermatological indications. Future growth is unlikely to be substantial, and the market will remain competitive and influenced by broader trends in dermatological treatment and healthcare economics.
Key Takeaways
Clocortolone pivalate's patent protection for the active pharmaceutical ingredient has long expired, enabling widespread generic availability. While formulation and method-of-use patents may have extended some market exclusivity for specific branded products, these protections are also largely expired or nearing expiry. The market is dominated by generics, leading to highly competitive pricing. Branded clocortolone pivalate products command a premium, but face significant price erosion and declining market share against generics. Generic formulations are priced aggressively, with prices expected to remain low and stable due to intense competition among multiple manufacturers. Clocortolone pivalate is a high-potency corticosteroid effective for severe inflammatory skin conditions but carries risks of local and systemic side effects, necessitating careful physician oversight. Future market prospects are characterized by continued generic dominance, stable but low-volume demand, and potential erosion from newer, more targeted dermatological therapies.
Frequently Asked Questions
-
What is the primary therapeutic indication for clocortolone pivalate? Clocortolone pivalate is indicated for the relief of inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses.
-
What is the potency classification of clocortolone pivalate compared to other topical corticosteroids? Clocortolone pivalate is classified as a high-potency topical corticosteroid.
-
Are there any active patents that currently provide exclusivity for clocortolone pivalate? The original composition of matter patent has long expired. While some formulation or method-of-use patents may have existed, their exclusivity period is largely over, allowing for generic competition.
-
What are the main factors influencing the price of generic clocortolone pivalate? The price of generic clocortolone pivalate is primarily influenced by the number of competing manufacturers, manufacturing costs, and payer negotiations.
-
What is the expected market growth trajectory for clocortolone pivalate over the next five years? The market for clocortolone pivalate is expected to experience low single-digit growth or a slight decline, driven by generic competition and the emergence of alternative therapies.
Citations
[1] U.S. Patent 3,940,474. (1976). Corticosteroid esters. Retrieved from USPTO database.
More… ↓
