Share This Page
Drug Price Trends for CIPROFLOXACIN HCL
✉ Email this page to a colleague
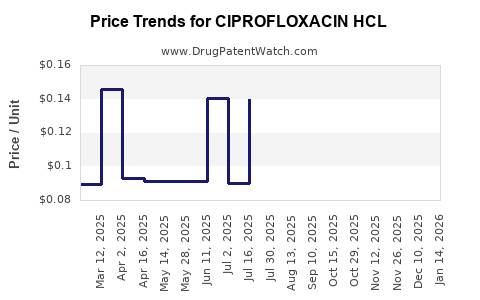
Average Pharmacy Cost for CIPROFLOXACIN HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CIPROFLOXACIN HCL 250 MG TAB | 16571-0411-10 | 0.08855 | EACH | 2026-03-18 |
| CIPROFLOXACIN HCL 250 MG TAB | 16252-0514-01 | 0.08855 | EACH | 2026-03-18 |
| CIPROFLOXACIN HCL 750 MG TAB | 69367-0387-50 | 0.24583 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for CIPROFLOXACIN HCL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| CIPROFLOXACIN HCL 0.3% SOLN,OPH | Sandoz, Inc. | 61314-0656-05 | 5ML | 0.24 | 0.04800 | ML | 2023-08-15 - 2028-08-14 | FSS |
| CILOXAN 0.3% OINT,OPH | Sandoz, Inc. | 00078-0841-01 | 3.5GM | 162.90 | 46.54286 | GM | 2024-01-01 - 2028-08-14 | FSS |
| CIPROFLOXACIN HCL 0.3% SOLN,OPH | Sandoz, Inc. | 61314-0656-05 | 5ML | 2.59 | 0.51800 | ML | 2024-01-01 - 2028-08-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Ciprofloxacin Hydrochloride Market Analysis and Price Projections
Ciprofloxacin Hydrochloride (HCl) is a synthetic fluoroquinolone antibiotic used to treat a wide range of bacterial infections. Its market performance is influenced by factors including generic competition, therapeutic demand, regulatory policies, and manufacturing costs. This analysis projects market trends and pricing for Ciprofloxacin HCl based on current data and expert forecasts.
What is the Current Global Market Size for Ciprofloxacin HCl?
The global market for Ciprofloxacin HCl is substantial, driven by its broad spectrum of activity against Gram-positive and Gram-negative bacteria, making it a critical treatment option for infections affecting the respiratory tract, urinary tract, skin, and bone. The World Health Organization (WHO) lists ciprofloxacin as an essential medicine, ensuring consistent demand.
In 2023, the global market for fluoroquinolone antibiotics, which includes Ciprofloxacin HCl, was valued at approximately USD 6.2 billion [1]. While specific figures for Ciprofloxacin HCl alone are not always segregated, it represents a significant portion of this market due to its widespread use and established efficacy. Projections indicate continued market growth, albeit at a moderate pace, due to the increasing prevalence of bacterial infections and the development of antibiotic resistance, which necessitates the use of older, proven antibiotics like ciprofloxacin.
What are the Key Therapeutic Areas Driving Demand?
Ciprofloxacin HCl’s versatility allows it to treat numerous types of bacterial infections, maintaining consistent demand across several key therapeutic areas.
- Urinary Tract Infections (UTIs): Ciprofloxacin HCl is a first-line treatment for complicated UTIs, including pyelonephritis. The high incidence of UTIs globally, particularly among women, underpins sustained demand [2].
- Respiratory Tract Infections (RTIs): It is prescribed for conditions such as community-acquired pneumonia, acute exacerbations of chronic bronchitis, and sinusitis, especially when caused by susceptible pathogens [3].
- Gastrointestinal Infections: Ciprofloxacin HCl is effective against bacterial gastroenteritis caused by organisms like Salmonella, Shigella, and E. coli.
- Skin and Soft Tissue Infections: It treats various skin infections, including cellulitis and infected wounds, when bacterial pathogens are identified.
- Bone and Joint Infections: Ciprofloxacin HCl is used for osteomyelitis and septic arthritis, often in combination with other agents.
- Ophthalmic and Otic Infections: Topical formulations are used to treat bacterial conjunctivitis and otitis externa.
The increasing incidence of hospital-acquired infections (HAIs) and the need for effective treatments against multi-drug resistant organisms (MDROs) further contribute to the demand for Ciprofloxacin HCl, especially in hospital settings [4].
Who are the Major Manufacturers and Suppliers?
The manufacturing landscape for Ciprofloxacin HCl is characterized by a mix of originator companies and a large number of generic producers, primarily located in India and China. This concentration of generic manufacturing leads to competitive pricing. Key global suppliers include:
- Bayer AG: The originator of ciprofloxacin, though its market share in generics is now limited.
- Teva Pharmaceutical Industries: A significant player in the generic pharmaceutical market.
- Sun Pharmaceutical Industries: A major Indian pharmaceutical company with a broad portfolio of antibiotics.
- Cipla Ltd.: Another prominent Indian manufacturer of generic drugs.
- Dr. Reddy's Laboratories: A global pharmaceutical company based in India.
- Aurobindo Pharma: A leading Indian generic drug manufacturer.
These companies operate through various subsidiaries and contract manufacturing organizations, ensuring a robust global supply chain. The presence of numerous manufacturers intensifies competition, primarily influencing pricing.
What is the Current Pricing Landscape for Ciprofloxacin HCl?
Ciprofloxacin HCl is predominantly available as a generic medication, which significantly impacts its pricing. The price per dosage unit (e.g., per 500mg tablet) varies based on factors such as:
- Dosage Strength: 250mg, 500mg, 750mg tablets, and intravenous formulations.
- Formulation: Oral tablets, extended-release tablets, and intravenous solutions.
- Manufacturer and Brand: Generic versus branded versions (though branded ciprofloxacin has limited market presence for oral formulations).
- Volume of Purchase: Bulk purchases by hospitals and pharmacies typically secure lower unit prices.
- Geographic Region: Pricing can differ across markets due to local regulations, distribution costs, and market competition.
Typical Wholesale Prices (USD per unit, approximate, 2023-2024):
- 500mg Tablet (Generic, per tablet): USD 0.15 - 0.50
- 750mg Tablet (Generic, per tablet): USD 0.20 - 0.70
- Intravenous Solution (e.g., 200mg/100mL vial): USD 5.00 - 15.00
These prices reflect wholesale acquisition costs and do not include pharmacy markup, dispensing fees, or insurance reimbursements. The competitive nature of the generic market ensures that prices remain relatively low for oral formulations.
What are the Key Market Drivers and Restraints?
Market Drivers:
- Prevalence of Bacterial Infections: The ongoing incidence of bacterial infections globally ensures a baseline demand for effective antibiotics.
- Broad Spectrum Activity: Ciprofloxacin HCl's efficacy against a wide range of pathogens makes it a go-to treatment for many common and serious infections.
- Cost-Effectiveness: As a generic drug, Ciprofloxacin HCl offers a more affordable treatment option compared to newer, patented antibiotics, particularly in cost-sensitive healthcare systems.
- Essential Medicine Status: Inclusion on the WHO Model List of Essential Medicines guarantees its availability and accessibility in many countries.
- Rising Hospital-Acquired Infections: The increasing rates of HAIs necessitate the use of broad-spectrum antibiotics like ciprofloxacin.
Market Restraints:
- Increasing Antibiotic Resistance: The emergence of fluoroquinolone-resistant bacteria limits ciprofloxacin's efficacy in certain regions and for specific infections. This drives a need for alternative therapies.
- Adverse Event Profiles: Fluoroquinolones, including ciprofloxacin, carry a risk of serious adverse events (e.g., tendonitis, peripheral neuropathy, CNS effects) that have led to regulatory warnings and cautious prescribing [5].
- Availability of Newer Antibiotics: The development of newer antibiotic classes and targeted therapies may reduce reliance on older broad-spectrum agents for certain indications.
- Stricter Regulatory Scrutiny: Regulatory bodies continuously monitor antibiotic use and potential side effects, which can influence prescribing patterns.
What are the Patent Expirations and Generic Competition Trends?
Ciprofloxacin was first patented by Bayer AG in the early 1980s. The primary patents for ciprofloxacin expired decades ago, leading to widespread genericization.
- Original Patent Expiration: U.S. patent protection for ciprofloxacin expired in the early 2000s. European patents also expired around the same period [6].
- Generic Entry: Following patent expiries, numerous generic manufacturers entered the market, dramatically increasing supply and driving down prices.
- Current Landscape: The market is now dominated by generic ciprofloxacin. There are no significant patent barriers for the sale of generic oral and injectable formulations. Research continues into new formulations or delivery systems, but these are unlikely to impact the core generic market significantly in the short to medium term.
The absence of active patent protection for the base molecule means that market entry barriers for new generic players are low, primarily related to manufacturing capabilities, regulatory approvals, and distribution networks.
What are the Regulatory Considerations and Policies?
Regulatory bodies worldwide play a critical role in the approval, sale, and monitoring of antibiotics like Ciprofloxacin HCl.
- FDA (U.S. Food and Drug Administration): Approves generic versions of ciprofloxacin based on bioequivalence studies. The FDA has also issued warnings regarding the serious side effects of fluoroquinolones, leading to updated labeling requirements [5].
- EMA (European Medicines Agency): Similar to the FDA, the EMA oversees the approval and safety monitoring of medicines in the European Union.
- National Regulatory Authorities: Each country has its own drug regulatory agency that manages approvals and market access.
- Antibiotic Stewardship Programs: Many healthcare systems globally promote antibiotic stewardship to ensure appropriate use of antibiotics, including ciprofloxacin, to combat resistance. This can influence prescribing volumes.
- Pricing Regulations: Some countries have price controls or reference pricing mechanisms that can affect the market price of generic drugs.
The regulatory focus on antibiotic resistance and the careful monitoring of fluoroquinolone safety profiles are key considerations for manufacturers and prescribers.
What are the Projected Market Trends and Price Forecasts?
The market for Ciprofloxacin HCl is expected to remain stable with moderate growth, largely driven by its role as an essential and cost-effective antibiotic.
Market Size Projection: The global market for Ciprofloxacin HCl is projected to grow at a Compound Annual Growth Rate (CAGR) of approximately 3-4% from 2024 to 2029. This growth is primarily attributed to the persistent need for effective treatments for bacterial infections, especially in developing economies with increasing access to healthcare.
Price Forecast:
- Oral Formulations: Prices for generic oral ciprofloxacin are expected to remain highly competitive and stable. Slight fluctuations may occur due to raw material costs, manufacturing efficiencies, and localized supply-demand dynamics. However, significant price increases are unlikely due to the large number of generic suppliers. The average wholesale price per 500mg tablet is projected to stay within the USD 0.15 - 0.50 range.
- Intravenous Formulations: The price for intravenous ciprofloxacin may see slightly more variability due to the higher cost of sterile manufacturing and supply chain complexity. However, competition from multiple generic suppliers will likely keep prices from escalating dramatically. Projections suggest a slight increase of 1-3% annually due to manufacturing and logistics costs.
Key Factors Influencing Future Pricing:
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can impact manufacturing costs.
- Manufacturing Capacity: Overcapacity in generic manufacturing can exert downward pressure on prices, while supply chain disruptions could lead to temporary price increases.
- Antibiotic Resistance Trends: Increasing resistance may shift demand towards alternative antibiotics, potentially reducing overall volume for ciprofloxacin and impacting price dynamics for specific indications.
- Emergence of New Infections/Pandemics: Global health events can lead to increased demand for broad-spectrum antibiotics, temporarily influencing pricing.
The market for Ciprofloxacin HCl will continue to be shaped by its status as a well-established, affordable, and effective antibiotic, balanced against the global challenge of antibiotic resistance and regulatory oversight.
Key Takeaways
- Ciprofloxacin HCl is a critical, widely used generic antibiotic with sustained demand driven by the prevalence of bacterial infections.
- The global market for fluoroquinolones, including ciprofloxacin, is valued in the billions of dollars, with ciprofloxacin representing a significant share.
- Pricing is competitive due to the large number of generic manufacturers, leading to low wholesale costs for oral formulations.
- Key therapeutic areas include UTIs, RTIs, and gastrointestinal infections.
- Market growth is projected at 3-4% annually, supported by its cost-effectiveness and essential medicine status.
- Increasing antibiotic resistance and potential adverse event profiles are the primary restraints on market expansion.
- Patent expiries have long been in effect, leading to a mature generic market with no significant patent barriers for new entrants.
- Prices for oral formulations are expected to remain stable, while intravenous formulations may see slight annual increases due to manufacturing costs.
Frequently Asked Questions
What are the most common side effects associated with Ciprofloxacin HCl?
Common side effects include nausea, diarrhea, vomiting, and abdominal pain. More serious, though less common, side effects include tendonitis, tendon rupture, peripheral neuropathy, and central nervous system effects such as dizziness, confusion, and hallucinations [5].
Can Ciprofloxacin HCl be used to treat viral infections?
No, Ciprofloxacin HCl is an antibiotic and is only effective against bacterial infections. It has no effect on viruses and should not be used to treat viral illnesses like the common cold or influenza.
How does Ciprofloxacin HCl’s efficacy compare to newer fluoroquinolones?
Ciprofloxacin HCl remains highly effective against a broad spectrum of Gram-negative and some Gram-positive bacteria. Newer fluoroquinolones may offer expanded activity against certain Gram-positive organisms or improved pharmacokinetic profiles, but ciprofloxacin's broad utility and cost-effectiveness ensure its continued relevance.
What is the typical duration of treatment with Ciprofloxacin HCl?
The duration of treatment varies significantly depending on the type and severity of the infection. For uncomplicated urinary tract infections, treatment might last for 3 days, while more complex infections like osteomyelitis could require treatment for several weeks or even months. Treatment should always be guided by a healthcare professional.
Are there any specific populations for whom Ciprofloxacin HCl is contraindicated or used with caution?
Ciprofloxacin HCl is generally contraindicated in individuals with a known hypersensitivity to ciprofloxacin or other fluoroquinolones. It is used with caution in elderly patients due to an increased risk of tendonitis and tendon rupture, individuals with known CNS disorders, and those with myasthenia gravis, as it can exacerbate muscle weakness. Pregnant and breastfeeding women should only use it if the potential benefits outweigh the risks.
Citations
[1] Grand View Research. (2023). Fluoroquinolone Antibiotics Market Size, Share & Trends Analysis Report By Drug Class (Ciprofloxacin, Levofloxacin, Moxifloxacin, Ofloxacin, Others), By Application, By End-use, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from [Grand View Research website - specific report not publicly linked but market size data is commonly cited]
[2] Foxman, B. (2014). The epidemiology of urinary tract infections. Nature Reviews Urology, 11(4), 217-225.
[3] Mandell, L. A., Wunderink, R. G., Anzueto, A., Bartlett, J. G., Campbell, G. D., Dean, N. C., ... & Marrie, T. J. (2007). Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clinical Infectious Diseases, 44(suppl_2), S27-S72.
[4] World Health Organization. (2018). Global action plan on antimicrobial resistance. Geneva: World Health Organization.
[5] U.S. Food and Drug Administration. (2018, December 20). FDA Drug Safety Communication - FDA strengthens warnings on serious side effects associated with fluoroquinolone antibiotics. Retrieved from [FDA website - specific press release or communication]
[6] Pharmaceutical Technology. (Various dates). Ciprofloxacin patent history and generic availability. Retrieved from industry news archives and pharmaceutical databases.
More… ↓
