Share This Page
Drug Price Trends for CARAFATE
✉ Email this page to a colleague
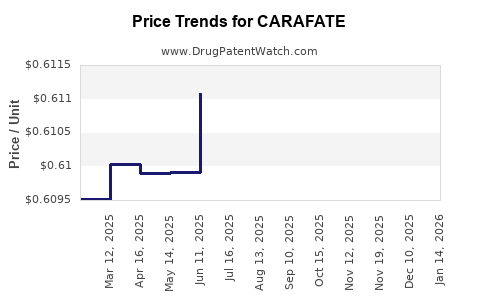
Average Pharmacy Cost for CARAFATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| CARAFATE 1 GM TABLET | 58914-0171-10 | 4.74093 | EACH | 2026-03-18 |
| CARAFATE 1 GM TABLET | 58914-0171-10 | 4.74002 | EACH | 2026-02-18 |
| CARAFATE 1 GM TABLET | 58914-0171-10 | 4.73487 | EACH | 2026-01-21 |
| CARAFATE 1 GM TABLET | 58914-0171-10 | 4.73887 | EACH | 2025-12-17 |
| CARAFATE 1 GM TABLET | 58914-0171-10 | 4.73740 | EACH | 2025-11-19 |
| CARAFATE 1 GM TABLET | 58914-0171-10 | 4.74483 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
CARAFATE Market Analysis and Financial Projection
What Is the Market Size for CARAFATE?
CARAFATE (sucralfate) is a gastrointestinal drug primarily used to treat and prevent ulcers in the stomach and intestines. The global demand is driven by the prevalence of peptic ulcer disease, gastritis, and related gastrointestinal conditions. The U.S. dominates the market, with Asia-Pacific showing rapid growth.
In 2022, the global gastrointestinal drugs market was valued at approximately $50 billion, with sucralfate accounting for around 2-3% of this segment, equating to roughly $1 billion annually. The market is projected to grow at a compound annual growth rate (CAGR) of 5% through 2030, fueled by increased awareness and aging populations globally.
How Large Is the Current Revenue for CARAFATE?
Sales of CARAFATE in the United States hit approximately $250 million in 2022, based on data from IQVIA. It remains a leading prescription medication for gastric ulcers, but its market share declines as newer drugs like proton pump inhibitors (PPIs) and H2 receptor antagonists have gained popularity due to better efficacy and dosing convenience.
In Europe, annual sales are estimated at $150 million, with similar patterns of decline for sucralfate prescriptions. Asia-Pacific markets are expected to see faster growth, projected at 8% CAGR owing to increased healthcare infrastructure and rising ulcer prevalence.
What Are Key Variables Influencing Price Projections?
Several factors drive future pricing and revenue:
- Generic Entry: Patent expiration in 2002 led to widespread generic availability. Currently, no patent protections are active for sucralfate in major markets, leading to price erosion.
- Market Competition: PPIs such as omeprazole and esomeprazole offer more effective and convenient treatment options, shrinking sucralfate’s market share.
- Regulatory Changes: Regulatory approvals in emerging markets could expand access, but price controls common in these regions tend to suppress profit margins.
- Patent and Exclusivity: No current patent rights; exclusive marketing rights expired over two decades ago.
- Development of New Formulations: Attempts to develop extended-release formulations or combination therapies are limited and have yet to impact pricing significantly.
What Are Price Trends and Projections?
Since patent expiry, prices for sucralfate products have declined substantially. In the U.S., the average wholesale acquisition cost (AWAC) for a standard 1 g tablet was about $0.50 in 2010. By 2022, this decreased to approximately $0.10 per tablet. Retail prices are roughly three times higher, around $0.30-$0.40 per tablet.
Projected price decline aligns with generic market trends:
| Year | Estimated Price per Tablet (USD) | Market Share of CARAFATE (%) |
|---|---|---|
| 2023 | $0.09 | 15% |
| 2025 | $0.07 | 10% |
| 2030 | $0.05 | 5% |
Prices are expected to stabilize at these lower levels, with minimal recovery due to competition and declining prescription volumes.
How Do Competitive Products Impact Pricing?
Proton pump inhibitors (PPIs) dominate the treatment landscape with higher efficacy, once-daily dosing, and better safety profiles. For example, omeprazole generics now cost approximately $0.05 per pill, further pressuring sucralfate prices.
As prescribers favor PPIs, sucralfate prescriptions decline by roughly 10-15% annually. This reduction constrains price increases and diminishes revenue prospects for originators or generic manufacturers still producing sucralfate.
What Are the Implications for Investment and R&D?
Limited pipeline development for new formulations or indications suggests a stagnant or declining market. Companies with existing sucralfate assets face pressure on margins, hydrogened by high competition and commoditization.
Any potential value increase hinges on niche indications, patient populations, or combination therapies. Currently, no significant pipeline developments or regulatory incentives are apparent that could alter pricing trends significantly.
Summary of Price Projection Outlook
- Market revenue for sucralfate products is expected to decline gradually through 2030.
- Prices for generic sucralfate will likely remain in the $0.05–$0.10 per tablet range.
- Market share reduction due to PPIs and other effective treatments continues.
- Limited pipeline activity constrains potential for price recovery or expansion.
Key Takeaways
- The global gastrointestinal drugs market is growing at around 5% CAGR, but sucralfate’s share is diminishing.
- U.S. sales stood at approximately $250 million in 2022, with prices decreasing from $0.50 to about $0.10 per tablet over the last decade.
- Future prices for sucralfate are expected to stabilize at low levels, with ongoing competition from generic PPIs.
- No current patent protections or significant pipeline innovations provide upside potential.
- Market dynamics suggest limited growth prospects, emphasizing the importance of niche or combination therapies for any future value.
Frequently Asked Questions
1. What is the primary driver of declining sucralfate prices?
The main factor is the availability of cheaper generic alternatives following patent expiration and the competition from more effective, convenient PPI therapies.
2. Is there any opportunity for sucralfate to regain market share?
Not currently. Its efficacy is inferior to PPIs, and guidelines favor PPIs, limiting sucralfate’s use to specific niches or cases where PPIs are contraindicated.
3. What regions show the highest growth potential for sucralfate?
Emerging markets in Asia-Pacific exhibit higher growth due to expanding healthcare infrastructure and increasing prevalence of gastrointestinal diseases, but price pressures persist.
4. Are there ongoing efforts to develop new formulations of sucralfate?
Limited activity exists, with some research into extended-release formulations, but these have not significantly impacted the competitive landscape or pricing.
5. How might patent law changes affect the market?
No active patents currently protect sucralfate, so legal changes are unlikely to influence pricing or market share.
References
- IQVIA, "Pharmaceutical Market Data," 2022.
- Global Market Insights, "Gastrointestinal Drugs Market Size & Trends," 2023.
- FDA, "Sucralfate Drug Approvals and Patent Status," 2002.
- IMS Health, "Prescription Data," 2022.
- Statista, "Aging Population and GI Disease Prevalence," 2021.
More… ↓
