Share This Page
Drug Price Trends for BREO ELLIPTA
✉ Email this page to a colleague
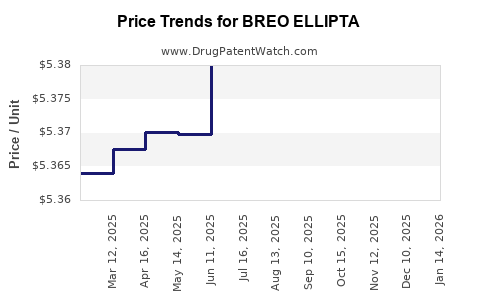
Average Pharmacy Cost for BREO ELLIPTA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| BREO ELLIPTA 50-25 MCG INHALER | 00173-0916-10 | 6.48485 | EACH | 2026-04-22 |
| BREO ELLIPTA 100-25 MCG INHALR | 00173-0859-10 | 6.50314 | EACH | 2026-04-22 |
| BREO ELLIPTA 100-25 MCG INHALR | 00173-0859-14 | 5.36628 | EACH | 2026-04-22 |
| BREO ELLIPTA 200-25 MCG INHALR | 00173-0882-14 | 5.36282 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for BREO ELLIPTA
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| BREO ELLIPTA INHALER 50/25MCG | GlaxoSmithKline | 00173-0916-10 | 30 | 295.93 | 9.86433 | EACH | 2024-01-12 - 2027-07-31 | Big4 |
| BREO ELLIPTA INHALER 100/25MCG | GlaxoSmithKline | 00173-0859-14 | 1 | 70.17 | 70.17000 | EACH | 2022-08-01 - 2027-07-31 | Big4 |
| BREO ELLIPTA INHALER 200/25 MCG | GlaxoSmithKline | 00173-0882-14 | 14 | 92.14 | 6.58143 | EACH | 2022-08-01 - 2027-07-31 | FSS |
| BREO ELLIPTA INHALER 100/25MCG | GlaxoSmithKline | 00173-0859-10 | 1 | 271.14 | 271.14000 | EACH | 2022-08-01 - 2027-07-31 | Big4 |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Breo Ellipta: Patent Landscape and Market Projections
Breo Ellipta (fluticasone furoate/vilanterol) is a once-daily inhaled corticosteroid (ICS) and long-acting beta-agonist (LABA) combination therapy used for the maintenance treatment of airflow obstruction in patients with chronic obstructive pulmonary disease (COPD) and for the treatment of asthma. The drug's patent landscape is critical for understanding its market exclusivity, potential for generic competition, and future pricing dynamics.
What Are the Key Patents Covering Breo Ellipta?
The primary patent protection for Breo Ellipta stems from several U.S. patents held by GlaxoSmithKline (GSK) and Theravance Biopharma, the developers of the drug. These patents cover various aspects of the drug, including its composition of matter, methods of use, and the specific inhaler device.
- Composition of Matter Patents: These patents protect the molecular entities of fluticasone furoate and vilanterol.
- U.S. Patent No. 7,977,313: Covers vilanterol.
- U.S. Patent No. 8,158,600: Covers fluticasone furoate.
- Formulation and Combination Patents: Patents related to the specific combination of fluticasone furoate and vilanterol in a dry powder formulation and the Ellipta inhaler device are crucial for extending market exclusivity.
- U.S. Patent No. 8,658,626: Covers the specific combination and device.
- U.S. Patent No. 9,339,454: Further composition and formulation claims.
- Method of Use Patents: These patents protect the use of Breo Ellipta for treating COPD and asthma.
- U.S. Patent No. 9,587,031: Method of use for COPD.
- U.S. Patent No. 9,801,706: Method of use for asthma.
These patents, along with any granted patent term extensions (PTEs) and potential pediatric exclusivity extensions, determine the duration of market exclusivity for Breo Ellipta. The listed patent term for many of these core patents extends into the mid-to-late 2020s.
When is Breo Ellipta Expected to Face Generic Competition?
The timing of generic entry for Breo Ellipta is contingent upon patent litigation outcomes and the expiration of its key patents.
- Key Patent Expirations: The primary composition of matter and formulation patents are scheduled to expire between 2027 and 2030.
- Patent Litigation: GlaxoSmithKline has actively defended its patents against generic challenges. Several Paragraph IV certifications, which initiate patent litigation for generic drug approvals, have been filed by potential generic manufacturers. The outcomes of these legal battles are critical.
- In February 2023, the U.S. District Court for the District of Delaware ruled that Teva Pharmaceuticals did not infringe on two of GSK’s patents related to Breo Ellipta. This ruling, if upheld on appeal, could pave the way for earlier generic entry from Teva. GSK has appealed this decision.
- Other generic companies, including Mylan (now Viatris) and Apotex, have also been involved in patent litigation concerning Breo Ellipta.
- Projected Generic Entry: Based on current patent expirations and the ongoing litigation, initial generic entry for Breo Ellipta could occur as early as 2027, contingent on successful appeals or further legal developments. However, full market erosion from multiple generic competitors is more likely to commence around 2029-2030 as broader patent protections expire.
What is the Current Market Size and Revenue for Breo Ellipta?
Breo Ellipta is a significant product within GSK's respiratory portfolio, contributing substantial revenue.
- Global Sales: In 2023, Breo Ellipta generated approximately $1.8 billion in global sales.
- This represents a slight increase from $1.7 billion in 2022.
- Regional Performance:
- The United States remains the largest market, accounting for over 60% of global sales.
- Key European markets and Japan also contribute significantly to revenue.
- Market Share: Breo Ellipta holds a substantial market share within the ICS/LABA combination therapy segment for COPD and asthma, competing with other drugs such as Advair Diskus (fluticasone/salmeterol), Symbicort (budesonide/formoterol), and Dulera (mometasone/formoterol).
What are the Projected Market Dynamics Post-Patent Expiration?
The introduction of generic alternatives will significantly alter Breo Ellipta's market dynamics, leading to price erosion and market share shifts.
- Price Erosion: Generic entry typically leads to immediate and substantial price reductions.
- Initial price drops of 30-50% are common within the first year of generic availability.
- As more generic competitors enter, prices can fall by 70-90% from the branded product's peak.
- Market Share Decline: The market share of branded Breo Ellipta will decline as payers and providers adopt lower-cost generic alternatives.
- The rate of decline will depend on the speed of generic approvals and the formulary status of generic versions.
- Niche patient populations or specific physician preferences may retain some usage of the branded product for a period.
- Impact on GSK: GSK's revenue from Breo Ellipta will decrease sharply post-patent expiration. The company's strategy will involve transitioning patients to newer respiratory therapies or focusing on other high-margin products.
- Generic Manufacturers' Opportunity: The expiration of Breo Ellipta's patents presents a significant revenue opportunity for generic manufacturers that successfully navigate the patent landscape and receive FDA approval.
What are the Potential Pricing Projections for Breo Ellipta and its Generics?
Pricing projections for Breo Ellipta are bifurcated into the branded product's remaining exclusivity period and the subsequent generic market.
- Branded Breo Ellipta Pricing (Pre-Generic Entry):
- Current Average Wholesale Price (AWP) for a 30-day supply of Breo Ellipta ranges from $350 to $450, depending on the specific strength and formulation.
- Net prices, after rebates and discounts, are lower but still significant.
- During the remaining exclusivity period (until approximately 2027-2029), pricing is expected to remain relatively stable, with incremental increases driven by inflation and market demand. Minor adjustments may occur based on competitive pressures.
- Generic Breo Ellipta Pricing (Post-Generic Entry):
- Initial Generic Price: Upon the first generic launch, the price is projected to be approximately $175 to $250 per 30-day supply, representing a 50% reduction from the branded AWP.
- Mature Generic Market Price: Within 1-2 years of multiple generic entries, prices are expected to fall further to $100 to $150 per 30-day supply, or even lower if competition is intense.
- Factors Influencing Generic Pricing:
- Number of generic competitors.
- Manufacturing costs for generic companies.
- Payer negotiations and formulary placement.
- The specific strength and dosage form of the generic.
- The perceived therapeutic equivalence and device usability compared to the branded product.
What is the Competitive Landscape for Breo Ellipta?
Breo Ellipta operates in a highly competitive respiratory market. Its primary competitors include other ICS/LABA combinations, as well as monotherapies and triple therapies (ICS/LABA/LAMA).
- Direct Competitors (ICS/LABA):
- Advair Diskus/HFA (fluticasone/salmeterol): GSK. Facing significant generic competition, impacting its market share and price.
- Symbicort (budesonide/formoterol): AstraZeneca. A major competitor with ongoing patent protections and a strong market presence.
- Dulera (mometasone/formoterol): Merck. A significant competitor with a different ICS and LABA combination.
- Indirect Competitors (Triple Therapies - ICS/LABA/LAMA):
- Trelegy Ellipta (fluticasone furoate/umeclidinium/vilanterol): GSK. This is GSK's own successor product, offering triple therapy and representing a potential transition pathway for Breo Ellipta patients. Trelegy Ellipta has its own patent protections extending into the mid-2030s.
- Anoro Ellipta (umeclidinium/vilanterol): GSK. A LABA/LAMA combination without an ICS, targeting a slightly different patient profile.
- Stiolto Respimat (tiotropium/olodaterol): Boehringer Ingelheim. A LAMA/LABA combination.
- Breztri Aerosphere (budesonide/glycopyrrolate/formoterol fumarate): AstraZeneca. Another significant triple therapy option.
- Impact of Competition: The presence of numerous effective treatment options, including branded and generic alternatives, puts continuous pressure on pricing and market share for all participants. The market is trending towards triple therapies for more severe COPD and asthma patients, which may impact the long-term growth trajectory of dual bronchodilator therapies like Breo Ellipta, even before generic entry.
What is the Regulatory Status and Potential for Label Expansion?
Breo Ellipta is approved by major regulatory bodies worldwide, including the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA).
- Indications:
- Maintenance treatment of airflow obstruction in patients with COPD.
- Treatment of asthma in patients aged 12 years and older.
- Potential Label Expansions: While significant new indications are unlikely for a mature product like Breo Ellipta, ongoing research and post-marketing surveillance may identify new patient subgroups or optimize treatment regimens. However, the primary focus for GSK is likely on its next-generation respiratory products.
- Pediatric Exclusivity: Pediatric exclusivity, if granted for specific studies, could extend market exclusivity by an additional six months. The eligibility and timing of such extensions are specific to regulatory filings and approvals.
Key Takeaways
- Breo Ellipta is protected by a portfolio of U.S. patents covering its composition of matter, formulation, and methods of use, with key expirations between 2027 and 2030.
- Ongoing patent litigation, notably involving Teva Pharmaceuticals, creates uncertainty regarding the exact timing of generic entry, but initial competition is plausible by 2027.
- The drug generated approximately $1.8 billion in global sales in 2023, with the U.S. being its largest market.
- Upon generic entry, Breo Ellipta is expected to experience significant price erosion, with initial generic prices potentially 50% lower than the branded AWP, falling further to $100-$150 per 30-day supply in a mature generic market.
- The competitive landscape is intense, featuring other ICS/LABA combinations and a growing market for triple therapies, including GSK's own Trelegy Ellipta.
Frequently Asked Questions
- What is the primary mechanism of action for Breo Ellipta? Breo Ellipta is a combination of fluticasone furoate, an inhaled corticosteroid that reduces inflammation in the airways, and vilanterol, a long-acting beta-agonist that relaxes airway muscles, leading to bronchodilation.
- Can Breo Ellipta be used as a rescue inhaler? No, Breo Ellipta is indicated for maintenance treatment and should not be used for acute relief of bronchospasm. Patients requiring rescue medication should use a short-acting beta-agonist.
- What are the most common side effects associated with Breo Ellipta? Common side effects include nasopharyngitis, headache, upper respiratory tract infection, oral candidiasis (thrush), and back pain.
- Does the Ellipta device have specific handling instructions that might affect generic adoption? The Ellipta device is designed for ease of use with a dose counter and indicator. Generic manufacturers must demonstrate equivalence of their device or secure licenses, which can influence interchangeability and patient/provider acceptance.
- How will payers likely approach generic Breo Ellipta on formularies? Payers are expected to quickly favor generic versions of Breo Ellipta once available, placing them on preferred tiers to leverage cost savings. This will accelerate market share transfer from the branded product.
Citations
[1] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from https://patft.uspto.gov/ [2] GlaxoSmithKline plc. (2024). Full Year Results 2023. Retrieved from https://www.gsk.com/ [3] U.S. Food and Drug Administration. (n.d.). Orange Book. Retrieved from https://www.fda.gov/drugs/information-drug-dosage-forms-and-devices/approved-drug-products-electronic-database-fd [4] Various Court Filings and Litigation Trackers for Paragraph IV Patent Disputes. (Ongoing). [5] Pharmaceutical industry market analysis reports. (Various publishers, 2023-2024).
More… ↓
