Share This Page
Drug Price Trends for ATOMOXETINE HCL
✉ Email this page to a colleague
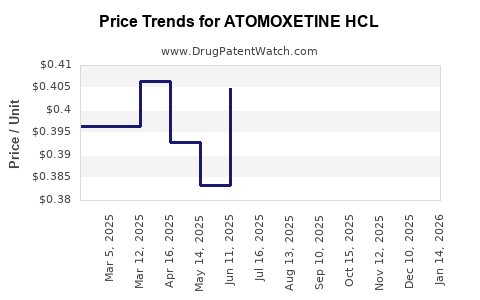
Average Pharmacy Cost for ATOMOXETINE HCL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ATOMOXETINE HCL 10 MG CAPSULE | 31722-0714-30 | 0.37944 | EACH | 2026-05-20 |
| ATOMOXETINE HCL 10 MG CAPSULE | 16714-0755-01 | 0.37944 | EACH | 2026-05-20 |
| ATOMOXETINE HCL 80 MG CAPSULE | 68462-0270-30 | 0.55352 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ATOMOXETINE HCL: Patent Landscape and Market Projections
Atomoxetine hydrochloride (HCl) is a selective norepinephrine reuptake inhibitor used for the treatment of Attention-Deficit/Hyperactivity Disorder (ADHD). This analysis examines the current patent landscape, key market players, and projected market performance for atomoxetine HCl.
What is the Current Patent Status of Atomoxetine HCl?
The primary patent for atomoxetine HCl, held by Eli Lilly and Company, has expired. The U.S. Patent No. 5,702,967, covering atomoxetine and its use, expired on September 16, 2013. However, secondary patents and formulation patents may still be in effect, influencing market dynamics.
- Key Patent Expiration: U.S. Patent 5,702,967 expired September 16, 2013.
- Pediatric Exclusivity: The drug benefited from pediatric exclusivity, extending market protection. This period also expired.
- Formulation Patents: Eli Lilly holds and has held various formulation patents that may offer continued, albeit limited, market exclusivity for specific delivery mechanisms or compositions. For instance, U.S. Patent 6,815,442, concerning improved oral dosage forms, expired in 2022.
- Method of Use Patents: While the original method of use patents have expired, new patents for novel indications or specific patient populations could emerge, though such developments are less common for older, established drugs.
Who are the Key Market Players for Atomoxetine HCl?
The patent expiration has led to the entry of generic manufacturers, significantly increasing competition and impacting pricing.
- Originator: Eli Lilly and Company (Strattera®).
- Generic Manufacturers: A substantial number of generic pharmaceutical companies market atomoxetine HCl. Notable players include:
- Teva Pharmaceuticals Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Pristiq Pharmaceuticals, Inc.
- Torrent Pharmaceuticals Ltd.
- Sun Pharmaceutical Industries Ltd.
- Aurobindo Pharma Ltd.
- Dr. Reddy's Laboratories Ltd.
These companies compete based on manufacturing scale, supply chain efficiency, and pricing strategies to gain market share.
What is the Historical Market Performance of Atomoxetine HCl?
Atomoxetine HCl, marketed as Strattera®, achieved significant sales for Eli Lilly following its FDA approval in 2002.
- Peak Sales: Strattera® generated over $1 billion in annual sales at its peak, driven by its non-stimulant profile, which appealed to parents and patients concerned about stimulant side effects.
- Impact of Generic Entry: Following patent expiry and the subsequent introduction of generics, sales for the branded product declined sharply. Generic competition typically drives down prices by 40-80% compared to branded products within the first two years of launch.
- Market Size Evolution: The overall market for atomoxetine HCl, considering both branded and generic sales, has stabilized but at a lower price point than during its period of exclusivity.
What are the Current Market Dynamics for Atomoxetine HCl?
The market for atomoxetine HCl is characterized by robust generic competition, price sensitivity, and ongoing demand driven by ADHD prevalence.
- Pricing: The average wholesale price (AWP) for atomoxetine HCl has decreased by an estimated 70% since the peak of generic entry. Generic manufacturers now compete primarily on price. The price of a 30-day supply can range from $15 to $50, depending on the dosage and generic manufacturer.
- Demand Drivers:
- Prevalence of ADHD: The diagnosis rate for ADHD remains high globally, particularly in children and adolescents. Global prevalence estimates suggest ADHD affects 5-13% of children and adolescents [1].
- Non-Stimulant Alternative: Atomoxetine HCl remains a critical non-stimulant treatment option for patients who do not tolerate or respond well to stimulant medications, or for those where stimulant use is contraindicated or discouraged.
- Physician Prescribing Habits: Prescribing patterns are influenced by factors such as patient age, co-morbid conditions, and physician familiarity with the drug's efficacy and side effect profile.
- Market Segmentation: The market is segmented by dosage strength (e.g., 10 mg, 25 mg, 40 mg, 60 mg, 80 mg, 100 mg) and capsule form.
- Supply Chain: The generic market relies on efficient manufacturing and distribution networks. Supply chain disruptions, raw material costs, and regulatory compliance are critical factors for generic producers.
What are the Projected Market Trends for Atomoxetine HCl?
The market for atomoxetine HCl is expected to exhibit modest growth, primarily driven by consistent demand and the competitive pricing of generic alternatives.
- Market Size Projection: The global atomoxetine HCl market, encompassing all generic and branded sales, is projected to grow at a compound annual growth rate (CAGR) of 2-4% over the next five years (2024-2029). This projection is based on stable ADHD prevalence and consistent demand for non-stimulant treatments.
- 2024 Estimated Market Size: Approximately $700 million - $800 million globally.
- 2029 Projected Market Size: Approximately $800 million - $950 million globally.
- Price Trends: Prices are expected to remain relatively stable within the generic range. Significant price increases are unlikely due to intense competition. Minor fluctuations may occur based on supply-demand dynamics and manufacturing costs.
- Competition Landscape: The generic market will likely remain fragmented, with established players maintaining their positions. Consolidation or new market entrants are less probable for a mature generic drug.
- Therapeutic Landscape Evolution: While new ADHD treatments, including novel stimulants and non-stimulants, may emerge, atomoxetine HCl's established efficacy, safety profile, and cost-effectiveness as a non-stimulant will likely secure its place in the treatment paradigm. Research into extended-release formulations or combination therapies could represent niche growth opportunities.
- Geographic Variations: Market growth will vary by region. Developed markets like North America and Europe will continue to be primary consumers, while emerging markets may see slightly higher growth rates due to increasing diagnosis rates and access to healthcare.
What are the Key Factors Influencing Future Market Performance?
Several factors will shape the future market trajectory of atomoxetine HCl.
- ADHD Diagnosis and Treatment Rates: Changes in diagnostic criteria or increased awareness of ADHD could positively impact demand. Conversely, shifts towards alternative treatment modalities could dampen growth.
- Competitive Pressure from New ADHD Drugs: The development and market penetration of novel ADHD therapies, particularly those offering improved efficacy, novel mechanisms of action, or more convenient dosing, could draw market share away from atomoxetine HCl.
- Generic Manufacturer Strategies: Aggressive pricing, supply chain optimization, and effective distribution by generic manufacturers will be crucial for maintaining and growing market share.
- Regulatory Environment: Changes in regulatory policies regarding pharmaceutical pricing, generic drug approvals, or manufacturing standards could affect market dynamics.
- Healthcare Reimbursement Policies: Payer policies and formulary decisions can influence physician prescribing patterns and patient access to atomoxetine HCl.
Key Takeaways
The atomoxetine HCl market is a mature generic drug segment characterized by intense price competition and stable demand driven by the prevalence of ADHD. Eli Lilly's patent exclusivity has long expired, leading to a diverse base of generic manufacturers. The market is projected for modest growth, with prices expected to remain competitive. Key influencers on future performance include ADHD diagnosis rates, the emergence of new ADHD therapies, and the strategic actions of generic producers.
FAQs
- Are there any new patents for atomoxetine HCl that could impact its market exclusivity? While the core patent for atomoxetine has expired, manufacturers may hold patents on specific formulations, delivery systems, or manufacturing processes. However, these are unlikely to grant broad market exclusivity comparable to the original patent.
- What is the primary competitive advantage of atomoxetine HCl in the ADHD market? Atomoxetine HCl's primary advantage is its status as a non-stimulant medication, offering an alternative for patients who do not tolerate stimulant drugs or for whom stimulants are contraindicated.
- How has the entry of generic manufacturers affected the price of atomoxetine HCl? The entry of generic manufacturers has led to a significant decrease in the price of atomoxetine HCl, with average wholesale prices dropping by an estimated 70% from their pre-generic levels.
- What is the projected growth rate for the global atomoxetine HCl market in the next five years? The global atomoxetine HCl market is projected to grow at a CAGR of 2-4% between 2024 and 2029.
- Which geographical regions represent the largest markets for atomoxetine HCl? North America and Europe are currently the largest markets for atomoxetine HCl, with emerging markets expected to show higher growth rates.
Citations
[1] Polanczyk, G. V., de Lima, M. S., Horta, B. L., Biedr, L., & Rohde, L. A. P. (2007). The worldwide prevalence of attention deficit hyperactivity disorder: a systematic review and meta-analysis. The American Journal of Psychiatry, 164(10), 1480–1489. doi:10.1176/appi.ajp.2007.07030300
More… ↓
