Share This Page
Drug Price Trends for AMANTADINE
✉ Email this page to a colleague
Average Pharmacy Cost for AMANTADINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
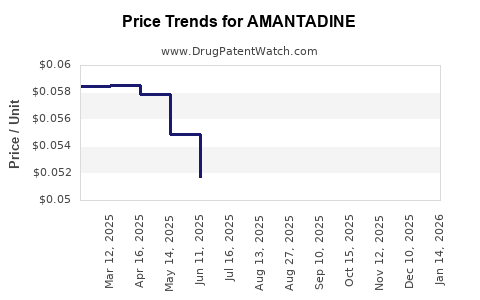
| AMANTADINE 50 MG/5 ML SOLUTION | 62135-0010-47 | 0.05084 | ML | 2026-03-18 |
| AMANTADINE 100 MG CAPSULE | 00832-1015-00 | 0.14606 | EACH | 2026-03-18 |
| AMANTADINE 100 MG CAPSULE | 00832-1015-50 | 0.14606 | EACH | 2026-03-18 |
| AMANTADINE 100 MG CAPSULE | 00904-7042-40 | 0.14606 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for AMANTADINE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| AMANTADINE HCL 50MG/5ML (SF) SYRUP,10ML | Golden State Medical Supply, Inc. | 00121-0646-10 | 100X10ML | 148.24 | 2023-06-15 - 2028-06-14 | FSS | ||
| AMANTADINE HCL 100MG TAB | Golden State Medical Supply, Inc. | 42543-0497-05 | 500 | 314.95 | 0.62990 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| AMANTADINE HCL 100MG CAP | Golden State Medical Supply, Inc. | 42543-0493-01 | 100 | 35.16 | 0.35160 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| AMANTADINE HCL 50MG/5ML (SF) SYRUP,10ML | Golden State Medical Supply, Inc. | 00121-0646-10 | 100X10ML | 158.29 | 2023-06-23 - 2028-06-14 | FSS | ||
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Amantadine: Patent Expiration and Market Dynamics Analysis
Amantadine, an antiviral and antiparkinsonian medication, faces a complex market landscape driven by expiring patents, generic competition, and evolving therapeutic applications. The drug's primary utility has been in treating Parkinson's disease and influenza A. However, recent advancements and potential new indications, alongside ongoing patent expirations, are reshaping its commercial trajectory. This analysis projects market dynamics and potential price movements through 2030.
What are the Key Patent Expirations for Amantadine?
Amantadine hydrochloride, first patented in the late 1950s by DuPont, has seen its foundational patents expire decades ago. The original U.S. patent for amantadine hydrochloride (U.S. Patent 2,875,195) was granted in 1959 and expired in 1976 [1]. This early patent covered the chemical compound itself.
Subsequent patents have focused on specific formulations, delivery methods, and new therapeutic uses. For instance, patents related to extended-release formulations and its use in treating conditions beyond Parkinson's and influenza have been more recent.
- Original Compound Patents: Expired mid-1970s.
- Formulation Patents (e.g., extended-release): Many expired or are nearing expiration between 2015 and 2025. Specific expiration dates vary by patent and jurisdiction. For example, patents protecting extended-release amantadine formulations (like Gocovri, approved for Parkinson's-related dyskinesia) had effective patent terms expiring in the mid-2020s. The U.S. Patent for Gocovri, U.S. Patent 9,238,138, protecting a specific extended-release formulation, has an expiration date of August 14, 2027 [2].
- Method of Use Patents: Patents claiming amantadine for specific indications, such as levodopa-induced dyskinesia, also face or have faced expiration. The patent for Gocovri's use in dyskinesia, U.S. Patent 9,795,535, is set to expire on November 21, 2029 [2].
The expiration of these later-stage patents is critical as it opens the door for broader generic market entry for more advanced amantadine products, not just the immediate-release versions.
How Has Amantadine's Therapeutic Landscape Evolved?
Amantadine was initially approved by the U.S. Food and Drug Administration (FDA) in 1968 for the prophylaxis and treatment of influenza A [3]. Its antiviral mechanism involves inhibiting viral uncoating. However, its efficacy against circulating influenza strains diminished significantly over time due to viral resistance, leading to its withdrawal as a primary influenza treatment by many public health agencies.
Its most enduring application is in the management of Parkinson's disease (PD). Amantadine is used to treat symptoms such as rigidity, tremors, and bradykinesia. It also plays a crucial role in managing levodopa-induced dyskinesia, involuntary movements that can develop in PD patients undergoing long-term levodopa therapy.
Recent research and development efforts have explored amantadine and its derivatives for novel indications:
- Neurological Disorders: Beyond PD, amantadine has been investigated for its potential in treating other neurological conditions, including traumatic brain injury (TBI), multiple sclerosis (MS) fatigue, and even certain psychiatric disorders. Clinical trials have shown promise for amantadine in improving motor function and reducing fatigue in these populations [4].
- Cognitive Enhancement: Some studies suggest amantadine may have cognitive-enhancing properties, leading to its investigation in conditions associated with cognitive impairment.
- COVID-19: During the early stages of the COVID-19 pandemic, amantadine was investigated as a potential antiviral agent, though it did not emerge as a primary treatment [5].
These expanded therapeutic explorations, even if some do not lead to blockbuster drugs, can extend the market relevance of the amantadine molecule and its derivatives.
What is the Current Market Size and Structure for Amantadine Products?
The global market for amantadine is fragmented, comprising both immediate-release (IR) formulations and extended-release (ER) formulations. The market size is difficult to pinpoint precisely due to the generic nature of many products and the inclusion of amantadine within combination therapies or its use off-label.
However, estimates place the global amantadine market in the range of $200 million to $500 million annually, with significant variations depending on the inclusion of specialized formulations and regional markets.
Key Market Segments:
- Immediate-Release (IR) Amantadine Hydrochloride: This is the most established and lowest-cost segment. It is primarily prescribed for Parkinson's disease symptoms and, historically, for influenza. Generic competition is intense in this segment.
- Extended-Release (ER) Amantadine Hydrochloride: Formulations like Gocovri (oseltamivir phosphate, formerly amantadine sulfate, re-branded to Adamantix extended release capsule in 2023 by Osmotica Pharmaceutical) offer improved patient convenience and potentially more stable therapeutic levels, particularly for managing dyskinesia. These branded ER products command higher prices and have faced patent challenges and generic alternatives as their patents expire.
- Investigational/Niche Indications: Products for TBI, MS fatigue, or other emerging uses represent a smaller but potentially growing segment.
Major Market Players:
The market includes both innovator companies and numerous generic manufacturers.
- Innovator Companies: Historically, companies like DuPont (now part of Corteva Agriscience) held initial patents. More recently, Osmotica Pharmaceutical (now part of Acutus Life Sciences) has been prominent with its extended-release formulations for dyskinesia.
- Generic Manufacturers: A substantial number of generic pharmaceutical companies produce IR amantadine hydrochloride globally. These include companies like Teva Pharmaceutical Industries, Mylan (now Viatris), Aurobindo Pharma, and various regional players.
The market is characterized by high volume, low-cost IR generics and lower volume, higher-cost branded ER products.
What are the Projected Price Trends for Amantadine Through 2030?
The price trajectory for amantadine products will be influenced by patent expirations, the introduction of generics, and the success of new indications.
Immediate-Release (IR) Amantadine Hydrochloride:
- Current: Prices for generic IR amantadine hydrochloride are extremely low, often cents per dose, due to intense competition and the absence of patent protection for the molecule.
- 2025-2030 Projection: Prices are expected to remain stable to slightly declining. The market is saturated with generics. Any price increases would likely be driven by inflation or supply chain disruptions, not market demand or innovation.
Extended-Release (ER) Amantadine Hydrochloride (e.g., Gocovri/Adamantix):
- Current: Branded ER formulations like Gocovri/Adamantix command premium pricing, often hundreds of dollars per month, reflecting R&D investment, clinical trial costs for specific indications (like dyskinesia), and the exclusivity period.
- 2025-2030 Projection:
- Pre-Patent Expiration (up to ~2027-2029): Prices for existing branded ER products will likely remain high.
- Post-Patent Expiration (post-2027/2029): The introduction of generic ER amantadine products is anticipated to cause a significant price decrease, potentially 30-60% or more, over a 2-3 year period following generic entry. This decline will be driven by direct price competition from multiple generic manufacturers.
- Niche/New Indications: If amantadine or its derivatives are approved for new, high-value indications (e.g., severe TBI recovery), branded products for these specific uses could maintain premium pricing, at least until their own patent protections expire. However, the base amantadine molecule's broad patent expiry limits the overall long-term pricing power for the core compound.
Overall Market Value Projection:
The total market value is projected to see moderate growth in the short term driven by the continued use of ER formulations and potential new niche indications. However, the broad expiry of patents protecting ER formulations is expected to lead to a flattening or slight decline in overall market value from 2027 onwards, as the significant price erosion in the ER segment outweighs any growth from generics or niche applications.
- 2024-2025: $250M - $550M (slight growth)
- 2026-2028: $300M - $600M (peak, before significant generic ER entry)
- 2029-2030: $250M - $500M (potential decline due to ER genericization)
What are the Key Risks and Opportunities for Amantadine Stakeholders?
Opportunities:
- Repurposing and New Indications: Successful clinical development and FDA approval for new therapeutic areas (e.g., TBI, MS fatigue, specific neurological subtypes) could create new revenue streams and extend product lifecycles.
- Formulation Innovation: Development of novel delivery systems or combination therapies involving amantadine could capture niche markets or improve patient compliance, potentially commanding premium pricing for a limited period.
- Emerging Markets: Increased access to healthcare in developing nations could drive demand for cost-effective IR amantadine generics for Parkinson's disease.
- Generic ER Development: Companies with the capability to develop and market bioequivalent generic ER amantadine products have a significant opportunity to capture market share post-patent expiry.
Risks:
- Generic Erosion: The most significant risk for branded ER amantadine products is the rapid price decline following generic entry, as seen with many other off-patent drugs.
- Competition from Newer Therapies: In Parkinson's disease and other neurological conditions, amantadine faces competition from newer, potentially more effective or better-tolerated treatments.
- Viral Resistance: The diminishing efficacy of amantadine against influenza strains limits its market potential in this original indication.
- Regulatory Hurdles: Obtaining approval for new indications requires extensive and costly clinical trials, with no guarantee of success.
- Supply Chain Vulnerabilities: As a mature product with many generic manufacturers, amantadine supply chains can be susceptible to disruptions.
- Reimbursement Pressures: Payers are increasingly scrutinizing the cost-effectiveness of all medications, including older drugs and their newer formulations.
Key Takeaways
Amantadine's market is transitioning from patent-protected, higher-priced formulations to a predominantly generic landscape. While original compound patents expired decades ago, patents on extended-release formulations and specific methods of use have provided extended market exclusivity for certain products. The expiration of these later patents, particularly for extended-release amantadine used to treat dyskinesia, will trigger significant price erosion and increased generic competition. Opportunities exist in exploring and securing patents for novel therapeutic indications, which could provide new, albeit potentially temporary, revenue streams. However, the overarching trend points towards market value stabilization or decline post-2027 due to genericization of the more lucrative extended-release segment.
Frequently Asked Questions
- When did the original amantadine patents expire? The original U.S. patent for amantadine hydrochloride (U.S. Patent 2,875,195) expired in 1976.
- What is the primary driver for potential price increases in amantadine products? Beyond standard inflation, potential price increases would be driven by successful FDA approval of amantadine for novel, high-value therapeutic indications for which it is not currently approved.
- How will generic competition affect the price of extended-release amantadine? Generic competition is projected to cause a significant price decrease for extended-release amantadine formulations, likely in the range of 30-60% or more, within two to three years of initial generic market entry.
- Are there any active patents that could significantly extend amantadine's market exclusivity for its current primary indications? Patents protecting the extended-release formulations and their specific use in treating dyskinesia are set to expire around 2027-2029. Beyond these, significant patent protection for amantadine for its established uses in Parkinson's disease and influenza is largely absent.
- What is the estimated current annual global market value for amantadine products? The estimated global amantadine market size is between $200 million and $500 million annually, with this figure influenced by the inclusion of branded extended-release formulations and regional market dynamics.
Citations
[1] U.S. Patent 2,875,195. (1959). Alpha-adamantanamine and salts thereof. [2] U.S. Patent 9,238,138. (2016). Controlled release compositions comprising amantadine. [3] U.S. Food & Drug Administration. (1968). FDA approves amantadine hydrochloride (Symmetrel) for influenza A and Parkinson's disease. [4] T. M. Itoh et al. (2021). Amantadine for fatigue in multiple sclerosis: A systematic review and meta-analysis. Journal of Neuroimmunology, 354, 157539. [5] G. L. Wang et al. (2021). Amantadine and hydroxychloroquine as a potential treatment for COVID-19. International Journal of Antimicrobial Agents, 57(1), 106241.
More… ↓
