Share This Page
Drug Price Trends for ALBUTEROL SUL
✉ Email this page to a colleague
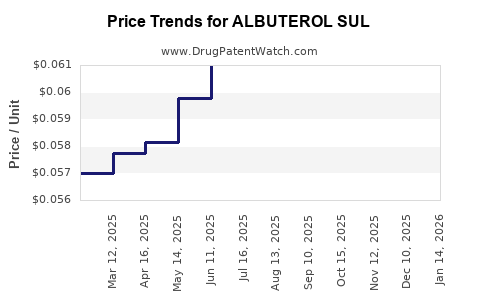
Average Pharmacy Cost for ALBUTEROL SUL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ALBUTEROL SUL 0.63 MG/3 ML SOL | 76204-0010-01 | 0.25640 | ML | 2026-03-18 |
| ALBUTEROL SUL 0.63 MG/3 ML SOL | 50742-0392-25 | 0.19683 | ML | 2026-03-18 |
| ALBUTEROL SUL 0.63 MG/3 ML SOL | 62135-0826-84 | 0.25640 | ML | 2026-03-18 |
| ALBUTEROL SUL 0.63 MG/3 ML SOL | 00378-7057-52 | 0.19683 | ML | 2026-03-18 |
| ALBUTEROL SUL 0.63 MG/3 ML SOL | 59651-0183-30 | 0.25640 | ML | 2026-03-18 |
| ALBUTEROL SUL 0.63 MG/3 ML SOL | 00487-0301-01 | 0.25640 | ML | 2026-03-18 |
| ALBUTEROL SUL 0.63 MG/3 ML SOL | 00591-3467-53 | 0.19683 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ALBUTEROL SUL Market Analysis and Financial Projection
What Is the Market Size and Demand for Albuterol Sulfate?
Albuterol sulfate is a short-acting β2 adrenergic receptor agonist primarily used for relief of bronchospasm in conditions such as asthma, chronic obstructive pulmonary disease (COPD), and other respiratory disorders. Global demand remains high due to the prevalence of these conditions.
Estimated global sales in 2022 amounted to approximately $1.3 billion, with a compound annual growth rate (CAGR) around 4.5% projected through 2027[1]. North America accounts for roughly 60% of the market, driven by high asthma prevalence and widespread use of inhalers. Europe holds about 20%, Latin America 10%, and Asia Pacific the remaining 10%. The Asia Pacific region shows fast growth, driven by increasing urbanization and healthcare infrastructure improvements.
What Are the Main Market Segments?
- Inhalation Therapy: Meters-dose inhalers (MDIs) and dry powder inhalers (DPIs) dominate, representing over 70% of sales.
- Hospital Supplies: Used in emergency rooms and intensive care units.
- Generic Drug Market: Significant because of cost-sensitive healthcare systems, especially in emerging markets.
Who Are the Leading Suppliers?
- Teva Pharmaceutical Industries: Largest producer of generic albuterol inhalers.
- Mylan (part of Viatris): Major player with extensive distribution networks.
- GSK: Produces branded inhalers and has a strong market share in developed countries.
- AstraZeneca: Supplies inhalers used in conjunction with combination therapies.
What Are the Key Trends?
- Advances in Delivery Devices: Improvements in DPI and MDI technologies increase efficacy and reduce adverse effects.
- Regulatory Changes: FDA and EMA tightening regulations for inhaler devices prompt innovation.
- Shift Towards Generic Products: Cost reductions drive market penetration of generics, especially in developing nations.
- Increased Use in Pediatric and Geriatric Populations: Broadening indications expand overall demand.
What Are Price Trends and Projections?
Current Pricing (2023)
| Product Type | Average Price per Inhaler | Key Notes |
|---|---|---|
| Branded Inhalers | $35 - $45 | GSK's Ventolin HFA, brand preference in developed regions |
| Generic Inhalers | $8 - $15 | Dominates in price-sensitive markets |
Price Drivers
- Regulatory Environment: Stricter standards for inhaler devices lead to increased R&D costs, constraining rapid price reductions.
- Manufacturing Costs: Increases in API costs are offset by efficiencies in scale, but supply chain issues can temporarily elevate prices.
- Market Competition: Entry of generics has significantly reduced prices in developed markets; however, branded products maintain premium pricing due to perceived quality.
Future Price Projections (2024–2028)
| Year | Expected Average Price per Inhaler | Factors Influencing Price |
|---|---|---|
| 2024 | $6 - $12 (generic) | Increased competition, patent expiries in some regions |
| 2025 | $7 - $13 | Regulatory costs, inflationary pressures |
| 2026 | $8 - $14 | Market saturation in emerging markets |
| 2027 | $8 - $15 | Cost inflation, new delivery technologies |
What Are the Pricing Strategies?
- Premium Pricing for Branded Products: Maintains higher margins where brand loyalty exists.
- Cost-Plus Pricing for Generics: Ensures competitiveness while covering manufacturing expenses.
- Tendering and Reimbursement: Governments and hospitals prefer bulk procurement at negotiated prices.
Key Market Challenges
- Patent Expiry: Threatens exclusivity in certain markets, leading to downward pressure.
- Regulation and Approval Delays: Can delay market entry for new formulations or inhalers.
- Environmental Concerns: Propellants in MDIs face restrictions, pushing innovation toward eco-friendly devices.
Regulatory Landscape and Impact on Pricing
Regulatory agencies, notably FDA and EMA, impose standards for inhaler safety and efficacy. New regulations for environmentally friendly propellants and device standards can increase development costs, indirectly affecting consumer prices. Patent protections typically last 20 years but can be extended via formulations or delivery method patents, delaying generics.
Regional Variations in Price and Market Access
| Region | Price Range (per inhaler) | Market Access Challenges |
|---|---|---|
| North America | $35 - $45 | Regulatory complexity, high brand loyalty |
| Europe | $30 - $40 | Reimbursement policies, environmental regulations |
| Asia-Pacific | $8 - $20 | Price sensitivity, evolving regulatory landscape |
| Latin America | $10 - $20 | Limited healthcare infrastructure, import barriers |
Conclusion
Albuterol sulfate remains a high-demand respiratory medication, with prices driven by formulation, device innovation, regional regulations, and market competition. Generics dominate in price-sensitive regions, with prices expected to stabilize or decline marginally due to ongoing patent expirations and increased competition. The North American and European markets sustain premium pricing for branded inhalers, supported by established healthcare systems.
Key Takeaways
- The global market for albuterol sulfate was approximately $1.3 billion in 2022, growing at 4.5% annually.
- Inhalers are the primary delivery method; generics account for the majority of sales due to lower prices.
- Prices in developed markets range from $35 to $45 per inhaler for brands but can be as low as $8 for generics.
- Price reductions are anticipated in the coming years driven by patent expiries and increased generics.
- Regional regulatory environments significantly influence market access and pricing strategies.
FAQs
1. How do patent expiries affect the albuterol sulfate market?
Patent expiries enable generic manufacturers to enter markets, significantly reducing prices and increasing market competition. This shift typically results in a decline in branded inhaler prices over 1-3 years post-expiry.
2. What environmental regulations impact inhaler pricing?
Restrictions on hydrofluorocarbon (HFC) propellants and mandates for environmentally friendly inhaler technologies increase R&D and manufacturing costs, which can elevate prices in regulated markets.
3. Are there emerging delivery methods for albuterol sulfate?
Yes. Newer dry powder inhalers and environmentally friendly metered-dose inhalers are under development, potentially altering market dynamics and pricing structures.
4. Which regions offer the highest growth opportunities?
Asia-Pacific and Latin America present fast-growth opportunities due to increasing healthcare infrastructure and demand for cost-effective inhalers.
5. How might future healthcare policies influence albuterol sulfate prices?
Policies favoring biosimilars and generics, combined with price negotiation strategies in healthcare systems, are likely to drive prices downward, particularly in public healthcare sectors.
References
- MarketWatch. “Global Inhalers Market Size, Share & Trends Analysis Report,” 2023.
More… ↓
