Share This Page
HUMULIN N Drug Profile
✉ Email this page to a colleague
Summary for Tradename: HUMULIN N
| High Confidence Patents: | 0 |
| Applicants: | 1 |
| BLAs: | 1 |
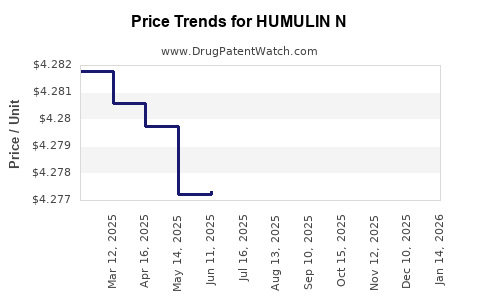
| Drug Prices: | Drug price information for HUMULIN N |
| Recent Clinical Trials: | See clinical trials for HUMULIN N |
Recent Clinical Trials for HUMULIN N
Identify potential brand extensions & biosimilar entrants
| Sponsor | Phase |
|---|---|
| Portal Diabetes, Inc. | PHASE1 |
| Stanford University | PHASE1 |
| Laboratorios Pisa S.A. de C.V. | PHASE1 |
Note on Biologic Patents
Matching patents to biologic drugs is far more complicated than for small-molecule drugs.
DrugPatentWatch employs three methods to identify biologic patents:
- Brand-side disclosures in response to biosimilar applications
- DrugPatentWatch analysis and company disclosures
- Patents from broad patent text search
These patents were identified from disclosures by the brand-side company, in response to a potential biosimilar seeking to launch. They have a high certainty of blocking biosimilar entry. The expiration dates listed are not estimates — they're expiration dates as indicated by the brand-side company.
These patents were identified from searching various sources, including drug labels and other general disclosures from the brand-side company. This list may exclude some of the patents which block biosimilar launch, and some of these patents listed may not actually block biosimilar launch. The expiration dates listed for these patents are estimates, based on the grant date of the patent.
For completeness, these patents were identified by searching the patent literature for mentions of the branded or ingredient name of the drug. Some of these patents protect the original drug, whereas others may protect follow-on inventions or even inventions casually mentioning the drug. The expiration dates listed for these patents are estimates, based on the grant date of the patent.
1) High Certainty: US Patents for HUMULIN N Derived from Brand-Side Litigation
No patents found based on brand-side litigation
2) High Certainty: US Patents for HUMULIN N Derived from DrugPatentWatch Analysis and Company Disclosures
No patents found based on company disclosures
3) Low Certainty: US Patents for HUMULIN N Derived from Patent Text Search
| Applicant | Tradename | Biologic Ingredient | Dosage Form | BLA | Patent No. | Estimated Patent Expiration | Source |
|---|---|---|---|---|---|---|---|
| Eli Lilly And Company | HUMULIN N | insulin isophane human | Injectable Suspension | 018781 | ⤷ Start Trial | 2037-02-17 | Patent claims search |
| >Applicant | >Tradename | >Biologic Ingredient | >Dosage Form | >BLA | >Patent No. | >Estimated Patent Expiration | >Source |
HUMULIN N: Market Dynamics and Financial Trajectory
Humulin N, a recombinant human insulin analog, holds a significant position in the diabetes management market. Its long-acting profile and established safety record contribute to its sustained demand. This analysis details its market positioning, patent landscape, competitive environment, and financial performance to inform strategic R&D and investment decisions.
What is the Current Market Position of Humulin N?
Humulin N is a basal insulin used for the management of type 1 and type 2 diabetes mellitus. Its primary function is to provide a relatively steady blood glucose lowering effect over a 24-hour period, complementing mealtime insulin or oral antidiabetic medications.
- Therapeutic Class: Intermediate-acting insulin (NPH insulin).
- Mechanism of Action: Humulin N is a suspension of human insulin that is absorbed more slowly than regular insulin, providing a longer duration of action. It works by facilitating the uptake of glucose into cells, reducing hepatic glucose production, and promoting fat and protein synthesis.
- Indications: Management of diabetes mellitus.
- Dosage Forms and Strengths: Available as a suspension for subcutaneous injection. Common strengths include U-100.
The global diabetes market is substantial and projected to grow, driven by increasing prevalence of obesity, sedentary lifestyles, and an aging population [1]. Humulin N occupies a segment of this market as a reliable and cost-effective basal insulin option.
- Market Size for Insulin: The global insulin market was valued at approximately USD 56.1 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 5.1% from 2023 to 2030 [1].
- Basal Insulin Share: While specific figures for NPH insulin within the basal insulin segment are not always disaggregated, NPH insulins like Humulin N represent a significant portion of the basal insulin market due to their historical presence and lower cost compared to some newer insulin analogs [2].
What is the Patent Landscape for Humulin N?
Humulin N is a mature product. Its original composition of matter patents have long expired. However, ongoing patent filings and intellectual property strategies for biologic drugs often involve formulations, manufacturing processes, and methods of use.
- Original Patent Expiration: The foundational patents for recombinant human insulin, including Humulin N, were filed in the late 1970s and early 1980s. These have long since expired, opening the door for generic and biosimilar competition. For example, U.S. Patent 4,215,037, related to the production of human insulin, was granted in 1980 [3].
- Formulation and Delivery Patents: While the active pharmaceutical ingredient (API) is off-patent, manufacturers may hold patents related to specific formulations, stabilizers, or novel drug delivery devices that enhance the product's performance or patient convenience. Such patents, if they exist for Humulin N, would likely focus on aspects like extended shelf-life, improved injection devices, or combination therapies.
- Manufacturing Process Patents: Biologic drugs, including recombinant insulins, involve complex manufacturing processes. Patents can be secured for specific fermentation techniques, purification methods, or quality control procedures that improve yield, purity, or reduce costs.
- Biosimilar Competition: The expiration of core patents has led to the development and introduction of biosimilar versions of recombinant human insulin. These biosimilars offer a competitive threat by providing similar therapeutic profiles at potentially lower price points. Regulatory pathways for biosimilars vary by region, with significant markets including the U.S. (under the Biologics Price Competition and Innovation Act, BPCIA) and Europe.
The lack of active composition of matter patents for Humulin N itself means that its market defense relies on brand loyalty, manufacturing efficiency, cost competitiveness, and potentially ongoing process or formulation patents that extend its commercial life or create barriers to biosimilar entry.
Who are the Key Competitors for Humulin N?
The competitive landscape for Humulin N is characterized by both traditional pharmaceutical companies and emerging biosimilar manufacturers. Competition exists across different classes of insulin and diabetes management therapies.
Direct Competitors (Other NPH Insulins):
- Novolin N (Novo Nordisk): This is a direct competitor, also an NPH insulin formulation. It is marketed by Novo Nordisk, a major player in diabetes care. Novolin N shares a similar pharmacokinetic profile and therapeutic indication with Humulin N.
- Basaglar (Eli Lilly and Company): While not a direct NPH competitor, Basaglar is a long-acting insulin analog (basal insulin) that competes for the same patient population seeking 24-hour glucose control. It is often considered a more advanced and less variable option than NPH insulins.
Broader Competitive Landscape (Other Basal Insulins and Diabetes Therapies):
- Insulin Glargine (e.g., Lantus, Toujeo - Sanofi): These are long-acting insulin analogs offering a flatter pharmacokinetic profile and potentially fewer nocturnal hypoglycemia events compared to NPH insulins.
- Insulin Detemir (e.g., Levemir - Novo Nordisk): Another long-acting basal insulin with a duration of action that can be adjusted by dose.
- Insulin Degludec (e.g., Tresiba - Novo Nordisk): Ultra-long-acting basal insulin with an extended duration of action, allowing for greater flexibility in dosing times.
- Oral Antidiabetic Medications: For type 2 diabetes, Humulin N competes with a wide array of oral agents, including metformin, sulfonylureas, DPP-4 inhibitors, SGLT2 inhibitors, and GLP-1 receptor agonists, some of which are used in combination with basal insulin.
- Combination Therapies: Fixed-dose combination pens containing both basal and rapid-acting insulins compete for patients requiring multiple insulin types.
Biosimilar Competition:
The advent of biosimilars for recombinant human insulin represents a significant competitive pressure. Companies like Biocon, Wockhardt, and others have developed and marketed biosimilar NPH insulins.
- Pricing: Biosimilars typically enter the market at a lower price point than the reference product, driving down overall market prices for NPH insulins.
- Market Access: Reimbursement policies and formulary placement by payers can significantly influence the uptake of both Humulin N and its biosimilar alternatives.
The competitive advantage for Humulin N increasingly relies on its established manufacturing base, supply chain reliability, and a brand name that may engender patient and physician trust, particularly in regions where cost is a primary driver or where newer analogs are less accessible.
What is the Financial Trajectory and Market Performance of Humulin N?
Humulin N's financial trajectory is influenced by its status as a mature product facing biosimilar competition and evolving treatment paradigms. Its performance is typically reported within broader insulin portfolios by its manufacturer, Eli Lilly and Company.
- Sales Performance (Eli Lilly): Humulin N's sales have generally shown a declining trend or modest growth in recent years due to the aforementioned competitive pressures. For example, in recent reporting periods, Eli Lilly has noted pressures on its older insulins due to biosimilar entry and a shift towards newer insulin analogs. Specific quarterly or annual revenue figures for Humulin N are often aggregated into "older insulins" or "human insulin" categories by Eli Lilly in their financial reports.
- In 2022, Eli Lilly reported that its U.S. insulin sales experienced headwinds due to increased competition from biosimilars for its older insulins, including Humulin [4]. This dynamic has persisted.
- While specific revenue figures for Humulin N are not always disclosed separately, the overall category of human insulins has seen pressure. For instance, Lilly's "Insulin" segment revenue can be impacted by the performance of Humulin and Novolin [5].
- Pricing Dynamics: The price of Humulin N has been subject to negotiation with payers and pharmacy benefit managers (PBMs). The introduction of biosimilars has exerted downward pressure on pricing, particularly in developed markets. In some regions, list prices may appear higher, but net prices after rebates and discounts are significantly lower.
- Gross Margins: As a mature product with established manufacturing processes, Humulin N likely possesses healthy gross margins when produced efficiently. However, the gross-to-net deduction (rebates and discounts) can significantly impact the realized net revenue and profitability.
- Investment in Manufacturing and Supply Chain: Eli Lilly continues to invest in its insulin manufacturing capabilities to ensure a reliable supply of Humulin N and other insulins, which is a critical factor in maintaining market share, especially in the face of supply chain disruptions that have affected other drug classes.
- Geographic Variations: The market dynamics and financial performance of Humulin N can vary significantly by region. In emerging markets, Humulin N may continue to hold a stronger position due to its affordability and established track record, while in highly developed markets, newer insulin analogs and biosimilars often dominate.
The financial outlook for Humulin N is one of a mature product managing its market share against newer therapeutic options and cost-effective biosimilars. Its continued financial contribution will depend on Eli Lilly's ability to optimize manufacturing costs, negotiate favorable payer contracts, and maintain strong supply chain reliability.
What are the Regulatory Considerations for Humulin N?
The regulatory landscape for Humulin N is primarily shaped by its status as a well-established biologic and the evolving regulatory pathways for biosimilars.
- FDA Approval and Post-Market Surveillance: Humulin N was originally approved by the U.S. Food and Drug Administration (FDA) for marketing. Like all approved drugs, it is subject to ongoing post-market surveillance to monitor for adverse events and ensure continued product safety and efficacy.
- Original approval dates for Humulin formulations are in the early 1980s.
- Biosimilar Pathways (FDA): The FDA's pathway for approving biosimilars, established by the Biologics Price Competition and Innovation Act (BPCIA) of 2010, allows for the approval of highly similar versions of reference biologics like Humulin N. A biosimilar must demonstrate no clinically meaningful differences in safety, purity, and potency from its reference product [6].
- The FDA has approved several biosimilars for recombinant human insulin. For instance, Semglee (insulin glargine-yfgn) and Admelog (insulin lispro-afrg) are examples of biosimilars approved for insulin products, highlighting the regulatory framework in place [7]. While specific biosimilars for Humulin N (as NPH insulin) have been approved, their market penetration varies.
- European Medicines Agency (EMA) Regulations: In Europe, the EMA has a similar, well-established pathway for the approval of biosimilar medicinal products. These products undergo rigorous scientific and regulatory evaluation to ensure they meet the same standards of quality, safety, and efficacy as the reference product [8].
- Labeling and Prescribing Information: Regulatory agencies ensure that labeling and prescribing information for Humulin N and its biosimilars accurately reflect their indications, contraindications, warnings, precautions, and adverse reactions. Changes to labeling may be required based on new safety data or comparative studies.
- Manufacturing Standards (cGMP): All manufacturers of Humulin N and its biosimilars must adhere to current Good Manufacturing Practices (cGMP) regulations. These regulations ensure the quality, safety, and efficacy of manufactured biological products through stringent control over manufacturing processes, facilities, and quality systems [9]. Eli Lilly's manufacturing sites for Humulin N are subject to regular FDA inspections.
- Pharmacovigilance: Ongoing pharmacovigilance activities are crucial for monitoring the safety profile of Humulin N post-approval. This includes reporting and analyzing adverse event data from healthcare providers, patients, and clinical studies. Regulatory agencies may mandate post-marketing studies to further assess specific safety aspects.
The regulatory environment for Humulin N is stable in terms of its established approval status. The primary regulatory dynamic influencing its market is the continuous evaluation and approval of biosimilars, which directly impacts competition and market access.
What are the Future Outlooks and Opportunities for Humulin N?
The future outlook for Humulin N is characterized by its role as a cost-effective basal insulin in a market increasingly dominated by newer analogs and advanced delivery systems, but with persistent demand in specific segments.
- Sustained Demand in Cost-Sensitive Markets: Humulin N is likely to retain a significant market share in emerging economies and among patient populations with limited financial resources due to its lower cost compared to insulin analogs. This segment represents an ongoing opportunity for stable, albeit lower-margin, revenue.
- Role in Combination Therapies: Humulin N can continue to be used effectively in combination with rapid-acting insulins or oral antidiabetic agents. Opportunities exist to promote its use as part of cost-effective treatment regimens, particularly for patients who do not achieve adequate glycemic control with oral medications alone.
- Biosimilar Competition Management: Eli Lilly's strategy will involve managing the impact of biosimilars. This includes focusing on manufacturing efficiency to remain cost-competitive and leveraging its established brand reputation and supply chain reliability.
- Potential for Formulation or Delivery Improvements: While core patents have expired, there may be incremental opportunities to patent novel formulations, stabilizers, or combination products that enhance Humulin N's characteristics, such as improved storage stability or ease of administration. However, the R&D investment required for such innovations must be weighed against the market potential of a mature product.
- Focus on Supply Chain Resilience: In an era of global supply chain vulnerability, Humulin N's consistent availability, backed by Eli Lilly's robust manufacturing infrastructure, can be a key differentiator and an opportunity to reinforce its value proposition to healthcare providers and patients.
- Integration with Digital Health Tools: While newer insulins are often paired with advanced pens and continuous glucose monitoring (CGM) systems, there may be opportunities to integrate Humulin N with more basic digital health tools for adherence tracking and glucose logging, enhancing its utility for a broader patient base.
The primary challenge remains the increasing therapeutic sophistication and patient preference for insulin analogs with more predictable pharmacokinetic profiles and reduced hypoglycemia risk. Therefore, Humulin N's future opportunities are more likely to be in solidifying its position in value-based healthcare settings and specific patient segments rather than leading market innovation.
Key Takeaways
- Humulin N is an established NPH insulin with a significant, albeit mature, market position in diabetes management.
- Original composition of matter patents have expired, leading to the presence of biosimilar competitors.
- Key competitors include other NPH insulins, insulin analogs, and a broad range of oral antidiabetic medications.
- Financial performance is characterized by declining or modest growth in developed markets due to biosimilar pressure and a shift to newer analogs, with sustained demand in cost-sensitive regions.
- Regulatory focus is on the ongoing approval and market access of biosimilar versions.
- Future opportunities lie in cost-sensitive markets, cost-effective combination therapies, and leveraging supply chain reliability.
Frequently Asked Questions
-
What is the primary difference between Humulin N and newer insulin analogs like glargine or detemir? Humulin N is an intermediate-acting NPH insulin with a more variable absorption profile and a peak of action. Newer insulin analogs (glargine, detemir, degludec) are long-acting, offering a flatter pharmacokinetic curve, more predictable glucose lowering, and often a reduced risk of hypoglycemia, particularly nocturnal hypoglycemia.
-
How has the introduction of biosimilars impacted the pricing of Humulin N? The introduction of biosimilar NPH insulins has exerted significant downward pressure on the net price of Humulin N, especially in markets with established biosimilar pathways and payer incentives for their uptake.
-
Does Eli Lilly hold any active patents that protect Humulin N from biosimilar competition? While the core patents for recombinant human insulin have expired, Eli Lilly may hold patents related to specific manufacturing processes, formulations, or drug delivery devices for Humulin N that could create some barriers to biosimilar entry or differentiation. However, these are typically not as strong as composition of matter patents.
-
In which geographical regions is Humulin N expected to maintain the strongest market share? Humulin N is expected to maintain its strongest market share in emerging markets and developing countries where cost is a primary determinant of drug selection, and where access to newer, more expensive insulin analogs is limited.
-
What is the typical dosing frequency for Humulin N? Humulin N is an intermediate-acting insulin and is typically administered once or twice daily by subcutaneous injection to provide basal (background) insulin coverage.
Citations
[1] Grand View Research. (2023). Diabetes Market Size, Share & Trends Analysis Report By Type (Type 1, Type 2), By Product (Insulin, Oral Hypoglycemic Drugs, Blood Glucose Monitoring Devices), By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/diabetes-market
[2] Global Market Insights. (2023). Insulin Market Report: By Type (Basal, Bolus, Pre-mixed), By Product (Insulin Pens, Insulin Syringes, Vials & Cartridges), By Indication (Type 1 Diabetes, Type 2 Diabetes), By Region, Forecast 2023-2032. Retrieved from https://www.gminsights.com/industry-analysis/insulin-market
[3] U.S. Patent 4,215,037. (1980). Process for the production of human insulin. Inventors: Goeddel, D. V., Heyneker, H. L., Hozumi, T., Arentzen, R., Itakura, K., Yansura, D. G., ... & Riggs, A. D.
[4] Eli Lilly and Company. (2023). Eli Lilly and Company Reports Strong Second Quarter 2023 Results. [Press Release].
[5] Eli Lilly and Company. (2022). Eli Lilly and Company Reports 2022 Fourth Quarter and Full-Year Results. [Press Release].
[6] U.S. Food and Drug Administration. (2020, December 1). What is a Biosimilar? Retrieved from https://www.fda.gov/drugs/biosimilars/what-biosimilar
[7] U.S. Food and Drug Administration. (2021, July 27). FDA Approves First Interchangeable Biosimilar For Insulin Glargine. [Press Release].
[8] European Medicines Agency. (2021, April). Biosimilar medicines. Retrieved from https://www.ema.europa.eu/en/human-regulatory-information/medicines/browse-all-medicines/biosimilar-medicines
[9] U.S. Food and Drug Administration. (2018, May 31). Current Good Manufacturing Practice (CGMP) Regulations. Retrieved from https://www.fda.gov/drugs/pharmaceutical-manufacturing/current-good-manufacturing-practice-cgmp-regulations
More… ↓