Strongbridge Company Profile
✉ Email this page to a colleague
What is the competitive landscape for STRONGBRIDGE
STRONGBRIDGE has one approved drug.
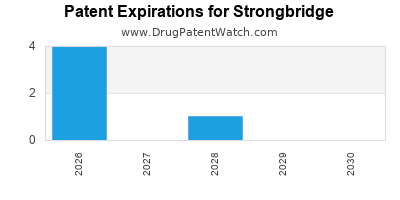
There are nine US patents protecting STRONGBRIDGE drugs.
There are thirty-three patent family members on STRONGBRIDGE drugs in twenty-one countries.
Drugs and US Patents for Strongbridge
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | RX | Yes | Yes | 11,903,940 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | RX | Yes | Yes | 12,377,096 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | RX | Yes | Yes | 11,020,393 | ⤷ Start Trial | ⤷ Start Trial | ||||
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | RX | Yes | Yes | ⤷ Start Trial | ⤷ Start Trial | |||||
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | RX | Yes | Yes | 10,835,530 | ⤷ Start Trial | ⤷ Start Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Strongbridge
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | 10,098,877 | ⤷ Start Trial |
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | 10,517,868 | ⤷ Start Trial |
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | 11,478,471 | ⤷ Start Trial |
| Strongbridge | RECORLEV | levoketoconazole | TABLET;ORAL | 214133-001 | Dec 30, 2021 | 10,835,530 | ⤷ Start Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for Strongbridge Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Norway | 20074117 | ⤷ Start Trial |
| South Korea | 20210135278 | ⤷ Start Trial |
| Israel | 286099 | ⤷ Start Trial |
| South Korea | 101387910 | ⤷ Start Trial |
| Cyprus | 1112519 | ⤷ Start Trial |
| >Country | >Patent Number | >Estimated Expiration |
Similar Applicant Names
Here is a list of applicants with similar names.